2600:1700:ce10:3700:a945:27df:56d7:2c03 (talk) →Bio-medical considerations: expanded citation |
not a polyphenol |
||
| (35 intermediate revisions by 16 users not shown) | |||
| Line 1: | Line 1: | ||
{{chembox |
{{chembox |
||
| ⚫ | |||
| Watchedfields = changed |
|||
| ⚫ | |||
| verifiedrevid = 443818652 |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |OtherNames = 2-propenoic acid, 3-(4-hydroxy-3-methoxyphenyl)-<br>ferulic acid<br>3-(4-hydroxy-3-methoxyphenyl)-2-propenoic acid<br>3-(4-hydroxy-3-methoxyphenyl)acrylic acid<br>3-methoxy-4-hydroxycinnamic acid<br>4-hydroxy-3-methoxycinnamic acid<br>(2''E'')-3-(4-hydroxy-3-methoxyphenyl)-2-propenoic acid<br>Coniferic acid<br>trans-ferulic acid<br>(''E'')-ferulic acid |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | | |
||
|Section1={{Chembox Identifiers |
|Section1={{Chembox Identifiers |
||
| |
|ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
||
| |
|ChemSpiderID = 393368 |
||
| |
|InChI = 1/C10H10O4/c1-14-9-6-7(2-4-8(9)11)3-5-10(12)13/h2-6,11H,1H3,(H,12,13)/b5-3+ |
||
| |
|InChIKey = KSEBMYQBYZTDHS-HWKANZROBE |
||
| |
|StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
||
| |
|StdInChI = 1S/C10H10O4/c1-14-9-6-7(2-4-8(9)11)3-5-10(12)13/h2-6,11H,1H3,(H,12,13)/b5-3+ |
||
| |
|StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
||
| |
|StdInChIKey = KSEBMYQBYZTDHS-HWKANZROSA-N |
||
| |
|CASNo_Ref = {{cascite|correct|CAS}} |
||
| |
|CASNo = 537-98-4 |
||
<!-- note CASNo for compound with undefined stereochemistry is 1135-24-6. Do not use this! --> |
|||
| |
|UNII_Ref = {{fdacite|correct|FDA}} |
||
| UNII = AVM951ZWST |
|||
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
|UNII = AVM951ZWST |
||
|ChEMBL_Ref = {{ebicite|correct|EBI}} |
|||
| |
|ChEMBL = 32749 |
||
| |
|PubChem = 445858 |
||
| |
|DrugBank_Ref = {{drugbankcite|correct|drugbank}} |
||
| |
|DrugBank = DB07767 |
||
| |
|ChEBI_Ref = {{ebicite|correct|EBI}} |
||
| |
|ChEBI = 17620 |
||
| |
|SMILES = COc1cc(ccc1O)/C=C/C(=O)O |
||
}} |
}} |
||
|Section2={{Chembox Properties |
|Section2={{Chembox Properties |
||
| |
|Formula = C<sub>10</sub>H<sub>10</sub>O<sub>4</sub> |
||
| |
|MolarMass = 194.18 g/mol |
||
| |
|Appearance = Crystalline powder |
||
| ⚫ | |||
| Density = |
|||
| ⚫ | |||
| MeltingPt_notes = |
|||
| BoilingPt = |
|||
| pKa = 4.61<ref name=mota>{{cite journal|last1=Mota|first1=Fátima L.|last2=Queimada|first2=António J.|last3=Pinho|first3=Simão P.|last4=Macedo|first4=Eugénia A.|title=Aqueous Solubility of Some Natural Phenolic Compounds|journal=Industrial & Engineering Chemistry Research|date=August 2008|volume=47|issue=15|pages=5182–5189|doi=10.1021/ie071452o|hdl=10198/1506|hdl-access=free}}</ref> |
| pKa = 4.61<ref name=mota>{{cite journal|last1=Mota|first1=Fátima L.|last2=Queimada|first2=António J.|last3=Pinho|first3=Simão P.|last4=Macedo|first4=Eugénia A.|title=Aqueous Solubility of Some Natural Phenolic Compounds|journal=Industrial & Engineering Chemistry Research|date=August 2008|volume=47|issue=15|pages=5182–5189|doi=10.1021/ie071452o|hdl=10198/1506|hdl-access=free}}</ref> |
||
| Solubility = 0.78 g/L<ref name=mota /> |
| Solubility = 0.78 g/L<ref name=mota /> |
||
}} |
|||
|Section3={{Chembox Hazards |
|Section3={{Chembox Hazards |
||
| NFPA-H = 2 |
| NFPA-H = 2 |
||
| NFPA-F = 1 |
| NFPA-F = 1 |
||
| NFPA-R = 0 |
| NFPA-R = 0 |
||
| MainHazards = |
|||
| FlashPt = |
|||
| AutoignitionPt = |
|||
| ⚫ | |||
}} |
}} |
||
| ⚫ | |||
'''Ferulic acid''' is a [[hydroxycinnamic acid]], an organic compound. It is an abundant phenolic [[phytochemical]] found in plant [[cell wall|cell walls]], covalently bonded as side chains to molecules such as [[arabinoxylan]]s. As a component of lignin, ferulic acid is a precursor in the manufacture of other [[aromatic]] compounds. The name is derived from the genus ''[[Ferula]]'', referring to the giant fennel (''Ferula communis''). |
|||
'''Ferulic acid''' is a [[hydroxycinnamic acid]]; it is an organic compound with the formula (CH<sub>3</sub>O)HOC<sub>6</sub>H<sub>3</sub>CH=CHCO<sub>2</sub>H. The name is derived from the genus ''[[Ferula]]'', referring to the giant fennel (''[[Ferula communis]]''). Classified as a [[phenols|phenolic]] [[phytochemical]], ferulic acid is an amber colored solid. Esters of ferulic acid are found in plant [[cell wall|cell walls]], covalently bonded to [[hemicellulose]] such as [[arabinoxylan]]s.<ref name="De Oliveira">{{cite journal |doi=10.1111/pbi.12292|title=Ferulic acid: A Key Component in Grass Lignocellulose Recalcitrance to Hydrolysis|year=2015|last1=De Oliveira|first1=Dyoni Matias|last2=Finger-Teixeira|first2=Aline|last3=Rodrigues Mota|first3=Thatiane|last4=Salvador|first4=Victor Hugo|last5=Moreira-Vilar|first5=Flávia Carolina|last6=Correa Molinari|first6=Hugo Bruno|last7=Craig Mitchell|first7=Rowan Andrew|last8=Marchiosi|first8=Rogério|last9=Ferrarese-Filho|first9=Osvaldo|last10=Dantas Dos Santos|first10=Wanderley|journal=Plant Biotechnology Journal|volume=13|issue=9|pages=1224–1232|pmid=25417596|doi-access=free}}</ref> Salts and esters derived from ferulic acid are called '''ferulates'''. |
|||
== Occurrence in nature == |
== Occurrence in nature == |
||
| ⚫ | As a building block of [[lignocellulose]]s, such as [[pectin]] and [[lignin]], ferulic acid is ubiquitous in the plant kingdom, including a number of vegetable sources. It occurs in particularly high concentrations in [[popcorn]] and [[bamboo shoot]]s.<ref>{{cite journal|last1=Zhao|first1=Zhaohui|last2=Moghadasian|first2=Mohammed H.|title=Chemistry, natural sources, dietary intake and pharmacokinetic properties of ferulic acid: A review|journal=Food Chemistry|date=August 2008|volume=109|issue=4|pages=691–702|doi=10.1016/j.foodchem.2008.02.039|pmid=26049981}}</ref><ref>{{cite journal|last1=Kumar|first1=Naresh|last2=Pruthi|first2=Vikas|title=Potential applications of ferulic acid from natural sources|journal=Biotechnology Reports|date=December 2014|volume=4|pages=86–93|doi=10.1016/j.btre.2014.09.002|pmid=28626667|pmc=5466124}}</ref> It is a major metabolite of [[chlorogenic acid]]s in humans along with [[caffeic acid|caffeic]] and [[isoferulic acid]], and is absorbed in the [[small intestine]], whereas other metabolites such as [[dihydroferulic acid]], [[feruloylglycine]] and dihydroferulic acid sulfate are produced from chlorogenic acid in the [[large intestine]] by the action of [[gut flora]].<ref>{{cite book|last1=Bagchi|first1=Debasis|last2=Moriyama|first2=Hiroyoshi|last3=Swaroop|first3=Anand|title=Green Coffee Bean Extract in Human Health|date=2016|publisher=CRC Press|isbn=9781315353982|page=92|url=https://books.google.com/books?id=S5WKDQAAQBAJ&pg=PT92|access-date=23 September 2017|language=en}}</ref> |
||
As a building block of [[lignocellulose]]s, such as [[pectin]]<ref>{{cite journal|last1=Saulnier|first1=Luc|last2=Thibault|first2=Jean-François|title=Ferulic acid and diferulic acids as components of sugar-beet pectins and maize bran heteroxylans|journal=Journal of the Science of Food and Agriculture|date=1 March 1999|volume=79|issue=3|pages=396–402|doi=10.1002/(SICI)1097-0010(19990301)79:3<396::AID-JSFA262>3.0.CO;2-B}}</ref> and [[lignin]], ferulic acid is ubiquitous in the plant kingdom. |
|||
| ⚫ | In cereals, ferulic acid is localized in the [[bran]] – the hard outer layer of grain. In [[wheat]], phenolic compounds are mainly found in the form of insoluble bound ferulic acid and may be relevant to resistance to wheat fungal diseases.<ref>{{cite journal | doi = 10.1111/j.1365-2621.2005.01057.x | title = Effect of wheat variety, farming site, and bread-baking on total phenolics | date = 2006 | last1 = Gelinas | first1 = Pierre | last2 = McKinnon | first2 = Carole M. | journal = International Journal of Food Science and Technology | volume = 41 | issue = 3 | pages = 329–332}}</ref> The highest known concentration of ferulic acid [[glucoside]] has been found in [[flaxseed]] ({{val|4.1|0.2|u=g/kg}}).<ref>{{cite journal | doi = 10.1002/pca.973 | pmid = 17623361 | title = Microwave-assisted extraction of the main phenolic compounds in flaxseed | date = 2007 | last1 = Beejmohun | first1 = Vickram | last2 = Fliniaux | first2 = Ophélie | journal = Phytochemical Analysis | volume = 18 | issue = 4 | pages = 275–285}}</ref> It is also found in [[barley]] grain.<ref>{{cite journal|title=Phenolic Compounds of Barley Grain and Their Implication in Food Product Discoloration|first1=Zory|last1=Quinde-Axtell|first2=Byung-Kee|last2=Baik|journal=J. Agric. Food Chem.|date=2006|volume=54|issue=26|pages=9978–84|doi=10.1021/jf060974w|pmid=17177530}}</ref> |
||
=== In food === |
|||
| ⚫ | |||
| ⚫ | [[Asterid]] [[eudicot]] plants can also produce ferulic acid. The tea brewed from the leaves of [[yacón]] (''Smallanthus sonchifolius''), a plant traditionally grown in the northern and central [[Andes]], contains quantities of ferulic acid. In [[legume]]s, the white bean variety [[navy bean]] is the richest source of ferulic acid among the common bean (''[[Phaseolus vulgaris]]'') varieties.<ref>{{cite journal | last1 = Luthria | first1 = Devanand L. | last2 = Pastor-Corrales | first2 = Marcial A. | date = 2006 | title = Phenolic acids content of fifteen dry edible bean (''Phaseolus vulgaris'' L.) varieties | journal = Journal of Food Composition and Analysis | volume = 19 | issue = 2–3| pages = 205–211 | doi=10.1016/j.jfca.2005.09.003}}</ref> It is also found in [[horse gram]]s (''Macrotyloma uniflorum'').{{Citation needed|date=December 2019|reason=removed citation to predatory publisher content}} |
||
| ⚫ | In cereals, ferulic acid is localized in the [[bran]] – the hard outer layer of grain. In [[wheat]], phenolic compounds are mainly found in the form of insoluble bound ferulic acid and may be relevant to resistance to wheat fungal diseases.<ref>{{cite journal | doi = 10.1111/j.1365-2621.2005.01057.x | title = Effect of wheat variety, farming site, and bread-baking on total phenolics | date = 2006 | last1 = Gelinas | first1 = Pierre | last2 = McKinnon | first2 = Carole M. | journal = International Journal of Food Science and Technology | volume = 41 | issue = 3 | pages = 329–332}}</ref> The highest known concentration of ferulic acid [[glucoside]] has been found in [[flaxseed]] {{ |
||
| ⚫ | Although there are many sources of ferulic acid in nature, its [[bioavailability]] depends on the form in which it is present: free ferulic acid has limited solubility in water, and hence poor bioavailability. In wheat grain, ferulic acid is found bound to [[cell wall]] [[polysaccharide]]s, allowing it to be released and absorbed in the small intestine.<ref>{{cite journal | doi = 10.1016/j.jcs.2008.12.001 | title =Bioavailability of ferulic acid is determined by its bioaccessibility| date = 2009 | last1 = Anson | first1 = Nuria Mateo | last2 = van den Berg | first2 = Robin | last3 = Bast | first3 = Aalt | last4 = Haenen | first4 = Guido R. M. M. | journal = Journal of Cereal Science | volume = 49 | issue = 2 | pages = 296–300}}</ref> |
||
| ⚫ | [[Asterid]] [[eudicot]] plants can also produce ferulic acid. The tea brewed from the leaves of [[yacón]] (''Smallanthus sonchifolius''), a plant traditionally grown in the northern and central [[Andes]], contains quantities of ferulic acid. In [[legume]]s, the white bean variety [[navy bean]] is the richest source of ferulic acid among the common bean (''[[Phaseolus vulgaris]]'') varieties.<ref>{{cite journal | last1 = Luthria | first1 = Devanand L. | last2 = Pastor-Corrales | first2 = Marcial A. | date = 2006 | title = Phenolic acids content of fifteen dry edible bean (''Phaseolus vulgaris'' L.) varieties |
||
| ⚫ | Although there are many sources of ferulic acid in nature, its [[bioavailability]] depends on the form in which it is present: free ferulic acid has limited solubility in water, and hence poor bioavailability. In wheat grain, ferulic acid is found bound to cell wall |
||
===In herbal medicines=== |
===In herbal medicines=== |
||
Ferulic acid has been identified in [[Chinese medicine]] herbs such as ''[[Angelica sinensis]]'' (female ginseng), ''[[Cimicifuga|Cimicifuga heracleifolia]]''<ref>{{cite journal|last1=Sakai|first1=S.|last2=Kawamata|first2=H.|last3=Kogure|first3=T.|last4=Mantani|first4=N.|last5=Terasawa|first5=K.|last6=Umatake|first6=M.|last7=Ochiai|first7=H.|title=Inhibitory effect of ferulic acid and isoferulic acid on the production of macrophage inflammatory protein-2 in response to respiratory syncytial virus infection in RAW264.7 cells |
Ferulic acid has been identified in [[Chinese medicine]] herbs such as ''[[Angelica sinensis]]'' (female ginseng), ''[[Cimicifuga|Cimicifuga heracleifolia]]''<ref>{{cite journal|last1=Sakai|first1=S.|last2=Kawamata|first2=H.|last3=Kogure|first3=T.|last4=Mantani|first4=N.|last5=Terasawa|first5=K.|last6=Umatake|first6=M.|last7=Ochiai|first7=H.|title=Inhibitory effect of ferulic acid and isoferulic acid on the production of macrophage inflammatory protein-2 in response to respiratory syncytial virus infection in RAW264.7 cells|journal=Mediators of Inflammation|date=1999|volume=8|issue=3|pages=173–175|pmid=10704056|doi=10.1080/09629359990513|pmc=1781798}}</ref> and ''[[Ligusticum|Ligusticum chuangxiong]]''. It is also found in the tea brewed from the European centaury (''[[Centaurium erythraea]]''), a plant used as a medical herb in many parts of Europe.<ref>{{cite journal | doi = 10.1021/jf001145s | title = Antioxidant Activity of ''Centaurium erythraea'' Infusion Evidenced by Its Superoxide Radical Scavenging and Xanthine Oxidase Inhibitory Activity | date = 2001 | last1 = Valentão | first1 = P. | last2 = Fernandes | first2 = E. | last3 = Carvalho | first3 = F. | last4 = Andrade | first4 = P. B. | last5 = Seabra | first5 = R. M. | last6 = Bastos | first6 = M. L. | journal = Journal of Agricultural and Food Chemistry | volume = 49 | issue = 7 | pages = 3476–3479 | pmid = 11453794}}</ref> |
||
=== In processed foods === |
=== In processed foods === |
||
Cooked [[sweetcorn]] releases increased levels of ferulic acid.<ref>{{cite web| url = http://www.news.cornell.edu/releases/Aug02/CornLiu.bpf.html | title = Cooking sweet corn boosts its ability to fight cancer and heart disease by freeing healthful compounds, Cornell scientists find | publisher = Cornell News | |
Cooked [[sweetcorn]] releases increased levels of ferulic acid.<ref>{{cite web| url = http://www.news.cornell.edu/releases/Aug02/CornLiu.bpf.html | title = Cooking sweet corn boosts its ability to fight cancer and heart disease by freeing healthful compounds, Cornell scientists find | publisher = Cornell News | access-date = 2009-09-07}}</ref> As [[plant sterol]] [[ester]]s, this compound is naturally found in [[rice bran oil]], a popular cooking oil in several Asian countries.<ref>{{cite book |title=Bailey's Industrial Oil and Fat Products |last=Orthoefer |first=F. T. |edition=6th |volume=2 |editor1-first=F. |editor1-last=Shahidi |date=2005 |publisher=John Wiley & Sons, Inc. |isbn=978-0-471-38552-3 |page=465 |chapter=Chapter 10: Rice Bran Oil |chapter-url=https://books.google.com/books?id=wG-0QgAACAAJ |access-date=2012-03-01}}</ref> |
||
Ferulic acid [[glycoside|glucoside]] can be found in commercial [[bread]]s containing [[flaxseed]].<ref>{{cite journal | doi = 10.1016/j.foodchem.2008.02.088 | pmid = 26047292 | title = Phenolic glucosides in bread containing flaxseed | date = 2008 | last1 = Strandås | first1 = C. | last2 = Kamal-Eldin | first2 = A. | last3 = Andersson | first3 = R. | last4 = Åman | first4 = P. | journal = Food Chemistry | volume = 110 | issue = 4 | pages = 997–999}}</ref> [[Rye bread]] contains [[ferulic acid dehydrodimer]]s.<ref |
Ferulic acid [[glycoside|glucoside]] can be found in commercial [[bread]]s containing [[flaxseed]].<ref>{{cite journal | doi = 10.1016/j.foodchem.2008.02.088 | pmid = 26047292 | title = Phenolic glucosides in bread containing flaxseed | date = 2008 | last1 = Strandås | first1 = C. | last2 = Kamal-Eldin | first2 = A. | last3 = Andersson | first3 = R. | last4 = Åman | first4 = P. | journal = Food Chemistry | volume = 110 | issue = 4 | pages = 997–999}}</ref> [[Rye bread]] contains [[ferulic acid dehydrodimer]]s.<ref>{{cite journal|last1=Boskov Hansen|first1=H.|last2=Andreasen|first2=M.|last3=Nielsen|first3=M.|last4=Larsen|first4=L.|last5=Knudsen|first5=Bach K.|last6=Meyer|first6=A.|last7=Christensen|first7=L.|last8=Hansen|first8=Å.|title=Changes in dietary fibre, phenolic acids and activity of endogenous enzymes during rye bread-making|journal=European Food Research and Technology|volume=214|issue=1|date=2014|pages=33–42|issn=1438-2377|doi=10.1007/s00217-001-0417-6|s2cid=85239461}}</ref> |
||
== Metabolism == |
== Metabolism == |
||
[[File:CaffeicAcIn.png|thumb|300px|left|In plants, ferulic acid (right) is derived from phenylalanine, which is converted to 4-hydroxycinnamic acid (left) and then [[caffeic acid]].]] |
[[File:CaffeicAcIn.png|thumb|300px|left|In plants, ferulic acid (right) is derived from [[phenylalanine]] (not shown), which is converted to [[4-hydroxycinnamic acid]] (left) and then [[caffeic acid]] (center).]] |
||
=== Biosynthesis === |
=== Biosynthesis === |
||
Ferulic acid is biosynthesized in plants from [[caffeic acid]] by the action of the enzyme [[caffeate O-methyltransferase|caffeate ''O''-methyltransferase]].<ref>{{cite book |last1= Shahadi |first1=Fereidoon|last2=Naczk |first2=Marian|title= Phenolics in Food and Nutraceuticals|url= https://archive.org/details/phenolicsfoodnut00shah_938 |url-access= limited |publisher= CRC Press|location= Florida|isbn= 978-1-58716-138-4|page= [https://archive.org/details/phenolicsfoodnut00shah_938/page/n12 4] |date= 2004}}</ref> |
Ferulic acid is biosynthesized in plants from [[caffeic acid]] by the action of the enzyme [[caffeate O-methyltransferase|caffeate ''O''-methyltransferase]].<ref>{{cite book |last1= Shahadi |first1=Fereidoon|last2=Naczk |first2=Marian|title= Phenolics in Food and Nutraceuticals|url= https://archive.org/details/phenolicsfoodnut00shah_938 |url-access= limited |publisher= CRC Press|location= Florida|isbn= 978-1-58716-138-4|page= [https://archive.org/details/phenolicsfoodnut00shah_938/page/n12 4] |date= 2004}}</ref><ref name="De Oliveira"/> |
||
In a proposed ferulic acid biosynthetic pathway for ''[[Escherichia coli]]'', <small>L</small>-tyrosine is converted to [[4-coumaric acid]] by [[tyrosine ammonia lyase]], which is converted to caffeic acid by Sam5, which is then converted to ferulic acid by caffeic acid methyltransferase.<ref>{{cite journal |last1=Kang |first1=Sun-Young |last2=Choi |first2=Oksik |last3=Lee |first3=Jae Kyung |last4=Hwang |first4=Bang Yeon |last5=Uhm |first5=Tai-Boong |last6=Hong |first6=Young-Soo |title=Artificial biosynthesis of phenylpropanoic acids in a tyrosine overproducing Escherichia coli strain |journal=Microbial Cell Factories |date=December 2012 |volume=11 |issue=1 |pages=153 |doi=10.1186/1475-2859-11-153 |pmid=23206756 |pmc=3554431 |doi-access=free }}</ref> |
|||
| ⚫ | Ferulic acid, together with dihydroferulic acid, is a component of [[lignocellulose]], serving to crosslink the lignin and polysaccharides, thereby conferring rigidity to the cell walls.<ref |
||
| ⚫ | Ferulic acid, together with [[dihydroferulic acid]], is a component of [[lignocellulose]], serving to crosslink the lignin and polysaccharides, thereby conferring rigidity to the cell walls.<ref>{{cite journal|last1=Iiyama|first1=K.|last2=Lam|first2=T. B.-T.|last3=Stone|first3=B. A.|title=Covalent Cross-Links in the Cell Wall|journal=Plant Physiology|volume=104|issue=2|date=1994|pages=315–320|issn=0032-0889|doi=10.1104/pp.104.2.315|pmid=12232082|pmc=159201}}</ref> |
||
It is an intermediate in the synthesis of [[monolignol]]s |
It is an intermediate in the synthesis of [[monolignol]]s, the monomers of [[lignin]], and is also used for the synthesis of [[lignan]]s. |
||
=== Biodegradation === |
=== Biodegradation === |
||
Ferulic acid is converted by certain strains of yeast, notably strains used in brewing of [[wheat beer]]s, such as [[Torulaspora delbrueckii|''Saccharomyces delbrueckii'']] (''Torulaspora delbrueckii''), to [[4-vinyl guaiacol]] (2-methoxy-4-vinylphenol) which gives beers such as [[Weissbier]] and Wit their distinctive |
Ferulic acid is converted by certain strains of yeast, notably strains used in brewing of [[wheat beer]]s, such as [[Torulaspora delbrueckii|''Saccharomyces delbrueckii'']] (''Torulaspora delbrueckii''), to [[4-vinyl guaiacol]] (2-methoxy-4-vinylphenol) which gives beers such as [[Weissbier]] and Wit their distinctive clove-like flavour. ''[[Saccharomyces cerevisiae]]'' (dry baker's yeast) and ''[[Pseudomonas fluorescens]]'' are also able to convert ''trans''-ferulic acid into 2-methoxy-4-vinylphenol.<ref>{{Cite journal |
||
| last1 = Huang | first1 = Z. |
| last1 = Huang | first1 = Z. |
||
| last2 = Dostal | first2 = L. |
| last2 = Dostal | first2 = L. |
||
| last3 = Rosazza | first3 = J. P. |
| last3 = Rosazza | first3 = J. P. |
||
| title = Microbial transformations of ferulic acid by Saccharomyces cerevisiae and Pseudomonas fluorescens |
| title = Microbial transformations of ferulic acid by ''Saccharomyces cerevisiae'' and ''Pseudomonas fluorescens '' |
||
| journal = Applied and Environmental Microbiology |
| journal = Applied and Environmental Microbiology |
||
| volume = 59 |
| volume = 59 |
||
| Line 92: | Line 83: | ||
| pages = 2244–2250 |
| pages = 2244–2250 |
||
| date = 1993 |
| date = 1993 |
||
| doi = 10.1128/AEM.59.7.2244-2250.1993 |
|||
| pmid = 8395165 |
| pmid = 8395165 |
||
| pmc = 182264 |
| pmc = 182264 |
||
| Line 98: | Line 90: | ||
| last2 = Dostal | first2 = L. |
| last2 = Dostal | first2 = L. |
||
| last3 = Rosazza | first3 = J. P. |
| last3 = Rosazza | first3 = J. P. |
||
| title = Purification and characterization of a ferulic acid decarboxylase from Pseudomonas fluorescens |
| title = Purification and characterization of a ferulic acid decarboxylase from ''Pseudomonas fluorescens'' |
||
| journal = Journal of Bacteriology |
| journal = Journal of Bacteriology |
||
| volume = 176 |
| volume = 176 |
||
| Line 106: | Line 98: | ||
| pmid = 7928951 |
| pmid = 7928951 |
||
| pmc = 196807 |
| pmc = 196807 |
||
| doi=10.1128/jb.176.19.5912-5918.1994 |
|||
}}</ref> |
}}</ref> |
||
== Ecology == |
== Ecology == |
||
Ferulic acid is one of the compounds that initiate the ''vir'' (virulence) region of ''[[Agrobacterium tumefaciens]]'', inducing it to infect plant cells.<ref |
Ferulic acid is one of the compounds that initiate the ''vir'' (virulence) region of ''[[Agrobacterium tumefaciens]]'', inducing it to infect plant cells.<ref>{{cite journal|last1=Kalogeraki|first1=Virginia S.|last2=Zhu|first2=Jun|last3=Eberhard|first3=Anatol|last4=Madsen|first4=Eugene L.|last5=Winans|first5=Stephen C.|title=The phenolic ''vir'' gene inducer ferulic acid is ''O''-demethylated by the VirH2 protein of an ''Agrobacterium tumefaciens'' Ti plasmid|journal=Molecular Microbiology|date=November 1999|volume=34|issue=3|pages=512–522|doi=10.1046/j.1365-2958.1999.01617.x|pmid=10564493|s2cid=28658847|doi-access=free}}</ref> |
||
== Extraction == |
== Extraction == |
||
It can be extracted from wheat bran and maize bran using concentrated alkali.<ref>{{cite journal| |
It can be extracted from wheat bran and maize bran using concentrated alkali.<ref>{{cite journal|last1=Buranov|first1=Anvar U.|first2=G.|last2=Mazza|title=Extraction and purification of ferulic acid from flax shives, wheat and corn bran by alkaline hydrolysis and pressurised solvents|journal=Food Chemistry|date=2009|volume=115|issue=4|pages=1542–1548|doi=10.1016/j.foodchem.2009.01.059}}</ref> |
||
== Bio-medical considerations == |
|||
[[File:Ferulicacidspectrum.PNG|thumb|right|[[Ultraviolet–visible spectroscopy|UV–visible spectrum]] of ferulic acid, with ''λ''<sub>max</sub> at 321 nm and a shoulder at 278 nm]] |
[[File:Ferulicacidspectrum.PNG|thumb|right|[[Ultraviolet–visible spectroscopy|UV–visible spectrum]] of ferulic acid, with ''λ''<sub>max</sub> at 321 nm and a shoulder at 278 nm]] |
||
Ferulic acid, like many [[natural phenol]]s, is an [[antioxidant effect of natural phenols|antioxidant]] [[in vitro]] in the sense that it is reactive toward [[free radical]]s such as [[reactive oxygen species]] (ROS). ROS and free radicals are implicated in [[DNA repair#DNA damage|DNA damage]], [[cancer]], and accelerated cell [[senescence|aging]]. |
|||
If added to a topical preparation of [[ascorbic acid]] and [[vitamin E]], ferulic acid may reduce [[oxidative stress]] and formation of [[thymine dimer]]s in [[skin]].<ref>{{Cite journal |
|||
| last1 = Lin | first1 = F. H. |
|||
| last2 = Lin | first2 = J. Y. |
|||
| last3 = Gupta | first3 = R. D. |
|||
| last4 = Tournas | first4 = J. A. |
|||
| last5 = Burch | first5 = J. A. |
|||
| last6 = Selim | first6 = M. A. |
|||
| last7 = Monteiro-Riviere | first7 = N. A. |
|||
| last8 = Grichnik | first8 = J. M. |
|||
| last9 = Zielinski | first9 = J. |
|||
| doi = 10.1111/j.0022-202X.2005.23768.x |
|||
| last10 = Pinnell | first10 = S. R. |
|||
| title = Ferulic Acid Stabilizes a Solution of Vitamins C and E and Doubles its Photoprotection of Skin |
|||
| journal = Journal of Investigative Dermatology |
|||
| volume = 125 |
|||
| issue = 4 |
|||
| pages = 826–832 |
|||
| date = 2005 |
|||
| pmid = 16185284 |
|||
| pmc = |
|||
}}</ref> There is also a small amount of research showing oral supplements of ferulic acid can inhibit melanin production in the process of [[skin whitening]].<ref>{{cite journal|work =Experimental Dermatology|date = August 2005| pages= 601-608|title = DeoxyArbutin: a novel reversible tyrosinase inhibitor with effective in vivo skin lightening potency |first1= Raymond E. |last1= Boissy|first2= Marty|last2= Visscher|first3= Mitchell A. |last3=DeLong|volume=14|issue = 8|doi = 10.1111/j.0906-6705.2005.00337.x}}</ref><ref>''Bioscience, Biotechnology, and Biochemistry'', December 2005, pages 2368-2373; ''International Journal of Dermatology'', August 2004, pages 604-607; ''Journal of Drugs in Dermatology'', July–August 2004, pages 377-381; ''Facial and Plastic Surgery'', February 2004, pages 3-9; ''Dermatologic Surgery'', March 2004, pages 385-388; ''Journal of Bioscience and Bioengineering'', March 2005, pages 272-276; ''Journal of Biological Chemistry'', November 7, 2003, pages 44320-44325; ''Journal of Agriculture and Food Chemistry'', February 2003, pages 1201-1207; ''International Journal of Cosmetic Science'', August 2000, pages 291-303; and ''Anti-Cancer Research'', September–October 1999, pages 3769-3774.</ref>{{full citation needed|date=August 2017}} |
|||
== Other applications == |
|||
=== Mass spectrometry === |
|||
It is used as a matrix for [[protein]]s in [[Matrix-assisted laser desorption/ionization|MALDI]] [[mass spectrometry]] analyses.<ref>{{Cite journal |
|||
| last1 = Beavis | first1 = R. C. |
|||
| last2 = Chait | first2 = B. T. |
|||
| last3 = Fales | first3 = H. M. |
|||
| doi = 10.1002/rcm.1290031207 |
|||
| title = Cinnamic acid derivatives as matrices for ultraviolet laser desorption mass spectrometry of proteins |
|||
| journal = Rapid Communications in Mass Spectrometry |
|||
| volume = 3 |
|||
| issue = 12 |
|||
| pages = 432–435 |
|||
| date = 1989 |
|||
| pmid = 2520223 |
|||
| pmc = |
|||
}}</ref> |
|||
== See also == |
== See also == |
||
| Line 168: | Line 118: | ||
== References == |
== References == |
||
{{reflist}} |
{{reflist}} |
||
<!-- orphaned reference |
<!-- orphaned reference<ref>{{cite journal |first1=Catherine |last1=Tomaro-Duchesneau |first2=Shyamali |last2=Saha |first3=Meenakshi |last3=Malhotra |first4=Michael |last4=Coussa-Charley |first5=Imen |last5=Kahouli |first6=Mitchell L. |last6=Jones |first7=Alain |last7=Labbe |first8=Satya |last8=Prakash | date = 2012 | title = Probiotic Ferulic Acid Esterase Active ''Lactobacillus fermentum'' NCIMB 5221 APA Microcapsules for Oral Delivery: Preparation and In Vitro Characterization | journal = Pharmaceuticals | volume = 5 | pages = 236–248 | issue = 2 | doi = 10.3390/ph5020236 | pmid=24288090 | pmc=3763630}}</ref> --> |
||
{{Hydroxycinnamic acid}} |
{{Hydroxycinnamic acid}} |
||
[[Category:Phenol antioxidants]] |
[[Category:Phenol antioxidants]] |
||
[[Category:O- |
[[Category:O-methylated hydroxycinnamic acids]] |
||
[[Category:Bitter-masking compounds]] |
[[Category:Bitter-masking compounds]] |
||
[[Category:Vinylogous carboxylic acids]] |
[[Category:Vinylogous carboxylic acids]] |
||
Latest revision as of 14:21, 2 April 2024
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
(2E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enoic acid | |
| Other names
2-propenoic acid, 3-(4-hydroxy-3-methoxyphenyl)-
ferulic acid 3-(4-hydroxy-3-methoxyphenyl)-2-propenoic acid 3-(4-hydroxy-3-methoxyphenyl)acrylic acid 3-methoxy-4-hydroxycinnamic acid 4-hydroxy-3-methoxycinnamic acid (2E)-3-(4-hydroxy-3-methoxyphenyl)-2-propenoic acid Coniferic acid trans-ferulic acid (E)-ferulic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.013.173 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H10O4 | |
| Molar mass | 194.18 g/mol |
| Appearance | Crystalline powder |
| Melting point | 168 to 172 °C (334 to 342 °F; 441 to 445 K) |
| 0.78 g/L[1] | |
| Acidity (pKa) | 4.61[1] |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
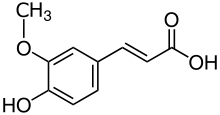
Ferulic acid is a hydroxycinnamic acid; it is an organic compound with the formula (CH3O)HOC6H3CH=CHCO2H. The name is derived from the genus Ferula, referring to the giant fennel (Ferula communis). Classified as a phenolic phytochemical, ferulic acid is an amber colored solid. Esters of ferulic acid are found in plant cell walls, covalently bonded to hemicellulose such as arabinoxylans.[2] Salts and esters derived from ferulic acid are called ferulates.
Occurrence in nature[edit]
As a building block of lignocelluloses, such as pectin and lignin, ferulic acid is ubiquitous in the plant kingdom, including a number of vegetable sources. It occurs in particularly high concentrations in popcorn and bamboo shoots.[3][4] It is a major metabolite of chlorogenic acids in humans along with caffeic and isoferulic acid, and is absorbed in the small intestine, whereas other metabolites such as dihydroferulic acid, feruloylglycine and dihydroferulic acid sulfate are produced from chlorogenic acid in the large intestine by the action of gut flora.[5]
In cereals, ferulic acid is localized in the bran – the hard outer layer of grain. In wheat, phenolic compounds are mainly found in the form of insoluble bound ferulic acid and may be relevant to resistance to wheat fungal diseases.[6] The highest known concentration of ferulic acid glucoside has been found in flaxseed (4.1±0.2 g/kg).[7] It is also found in barley grain.[8]
Asterid eudicot plants can also produce ferulic acid. The tea brewed from the leaves of yacón (Smallanthus sonchifolius), a plant traditionally grown in the northern and central Andes, contains quantities of ferulic acid. In legumes, the white bean variety navy bean is the richest source of ferulic acid among the common bean (Phaseolus vulgaris) varieties.[9] It is also found in horse grams (Macrotyloma uniflorum).[citation needed]
Although there are many sources of ferulic acid in nature, its bioavailability depends on the form in which it is present: free ferulic acid has limited solubility in water, and hence poor bioavailability. In wheat grain, ferulic acid is found bound to cell wall polysaccharides, allowing it to be released and absorbed in the small intestine.[10]
In herbal medicines[edit]
Ferulic acid has been identified in Chinese medicine herbs such as Angelica sinensis (female ginseng), Cimicifuga heracleifolia[11] and Ligusticum chuangxiong. It is also found in the tea brewed from the European centaury (Centaurium erythraea), a plant used as a medical herb in many parts of Europe.[12]
In processed foods[edit]
Cooked sweetcorn releases increased levels of ferulic acid.[13] As plant sterol esters, this compound is naturally found in rice bran oil, a popular cooking oil in several Asian countries.[14]
Ferulic acid glucoside can be found in commercial breads containing flaxseed.[15] Rye bread contains ferulic acid dehydrodimers.[16]
Metabolism[edit]
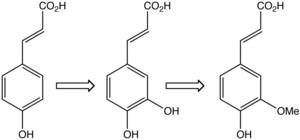
Biosynthesis[edit]
Ferulic acid is biosynthesized in plants from caffeic acid by the action of the enzyme caffeate O-methyltransferase.[17][2]
In a proposed ferulic acid biosynthetic pathway for Escherichia coli, L-tyrosine is converted to 4-coumaric acid by tyrosine ammonia lyase, which is converted to caffeic acid by Sam5, which is then converted to ferulic acid by caffeic acid methyltransferase.[18]
Ferulic acid, together with dihydroferulic acid, is a component of lignocellulose, serving to crosslink the lignin and polysaccharides, thereby conferring rigidity to the cell walls.[19]
It is an intermediate in the synthesis of monolignols, the monomers of lignin, and is also used for the synthesis of lignans.
Biodegradation[edit]
Ferulic acid is converted by certain strains of yeast, notably strains used in brewing of wheat beers, such as Saccharomyces delbrueckii (Torulaspora delbrueckii), to 4-vinyl guaiacol (2-methoxy-4-vinylphenol) which gives beers such as Weissbier and Wit their distinctive clove-like flavour. Saccharomyces cerevisiae (dry baker's yeast) and Pseudomonas fluorescens are also able to convert trans-ferulic acid into 2-methoxy-4-vinylphenol.[20] In P. fluorescens, a ferulic acid decarboxylase has been isolated.[21]
Ecology[edit]
Ferulic acid is one of the compounds that initiate the vir (virulence) region of Agrobacterium tumefaciens, inducing it to infect plant cells.[22]
Extraction[edit]
It can be extracted from wheat bran and maize bran using concentrated alkali.[23]

See also[edit]
- Caffeic acid
- Coumaric acid
- Diferulic acids
- Eugenol
- Isoferulic acid, an isomer of ferulic acid
References[edit]
- ^ a b Mota, Fátima L.; Queimada, António J.; Pinho, Simão P.; Macedo, Eugénia A. (August 2008). "Aqueous Solubility of Some Natural Phenolic Compounds". Industrial & Engineering Chemistry Research. 47 (15): 5182–5189. doi:10.1021/ie071452o. hdl:10198/1506.
- ^ a b De Oliveira, Dyoni Matias; Finger-Teixeira, Aline; Rodrigues Mota, Thatiane; Salvador, Victor Hugo; Moreira-Vilar, Flávia Carolina; Correa Molinari, Hugo Bruno; Craig Mitchell, Rowan Andrew; Marchiosi, Rogério; Ferrarese-Filho, Osvaldo; Dantas Dos Santos, Wanderley (2015). "Ferulic acid: A Key Component in Grass Lignocellulose Recalcitrance to Hydrolysis". Plant Biotechnology Journal. 13 (9): 1224–1232. doi:10.1111/pbi.12292. PMID 25417596.
- ^ Zhao, Zhaohui; Moghadasian, Mohammed H. (August 2008). "Chemistry, natural sources, dietary intake and pharmacokinetic properties of ferulic acid: A review". Food Chemistry. 109 (4): 691–702. doi:10.1016/j.foodchem.2008.02.039. PMID 26049981.
- ^ Kumar, Naresh; Pruthi, Vikas (December 2014). "Potential applications of ferulic acid from natural sources". Biotechnology Reports. 4: 86–93. doi:10.1016/j.btre.2014.09.002. PMC 5466124. PMID 28626667.
- ^ Bagchi, Debasis; Moriyama, Hiroyoshi; Swaroop, Anand (2016). Green Coffee Bean Extract in Human Health. CRC Press. p. 92. ISBN 9781315353982. Retrieved 23 September 2017.
- ^ Gelinas, Pierre; McKinnon, Carole M. (2006). "Effect of wheat variety, farming site, and bread-baking on total phenolics". International Journal of Food Science and Technology. 41 (3): 329–332. doi:10.1111/j.1365-2621.2005.01057.x.
- ^ Beejmohun, Vickram; Fliniaux, Ophélie (2007). "Microwave-assisted extraction of the main phenolic compounds in flaxseed". Phytochemical Analysis. 18 (4): 275–285. doi:10.1002/pca.973. PMID 17623361.
- ^ Quinde-Axtell, Zory; Baik, Byung-Kee (2006). "Phenolic Compounds of Barley Grain and Their Implication in Food Product Discoloration". J. Agric. Food Chem. 54 (26): 9978–84. doi:10.1021/jf060974w. PMID 17177530.
- ^ Luthria, Devanand L.; Pastor-Corrales, Marcial A. (2006). "Phenolic acids content of fifteen dry edible bean (Phaseolus vulgaris L.) varieties". Journal of Food Composition and Analysis. 19 (2–3): 205–211. doi:10.1016/j.jfca.2005.09.003.
- ^ Anson, Nuria Mateo; van den Berg, Robin; Bast, Aalt; Haenen, Guido R. M. M. (2009). "Bioavailability of ferulic acid is determined by its bioaccessibility". Journal of Cereal Science. 49 (2): 296–300. doi:10.1016/j.jcs.2008.12.001.
- ^ Sakai, S.; Kawamata, H.; Kogure, T.; Mantani, N.; Terasawa, K.; Umatake, M.; Ochiai, H. (1999). "Inhibitory effect of ferulic acid and isoferulic acid on the production of macrophage inflammatory protein-2 in response to respiratory syncytial virus infection in RAW264.7 cells". Mediators of Inflammation. 8 (3): 173–175. doi:10.1080/09629359990513. PMC 1781798. PMID 10704056.
- ^ Valentão, P.; Fernandes, E.; Carvalho, F.; Andrade, P. B.; Seabra, R. M.; Bastos, M. L. (2001). "Antioxidant Activity of Centaurium erythraea Infusion Evidenced by Its Superoxide Radical Scavenging and Xanthine Oxidase Inhibitory Activity". Journal of Agricultural and Food Chemistry. 49 (7): 3476–3479. doi:10.1021/jf001145s. PMID 11453794.
- ^ "Cooking sweet corn boosts its ability to fight cancer and heart disease by freeing healthful compounds, Cornell scientists find". Cornell News. Retrieved 2009-09-07.
- ^ Orthoefer, F. T. (2005). "Chapter 10: Rice Bran Oil". In Shahidi, F. (ed.). Bailey's Industrial Oil and Fat Products. Vol. 2 (6th ed.). John Wiley & Sons, Inc. p. 465. ISBN 978-0-471-38552-3. Retrieved 2012-03-01.
- ^ Strandås, C.; Kamal-Eldin, A.; Andersson, R.; Åman, P. (2008). "Phenolic glucosides in bread containing flaxseed". Food Chemistry. 110 (4): 997–999. doi:10.1016/j.foodchem.2008.02.088. PMID 26047292.
- ^ Boskov Hansen, H.; Andreasen, M.; Nielsen, M.; Larsen, L.; Knudsen, Bach K.; Meyer, A.; Christensen, L.; Hansen, Å. (2014). "Changes in dietary fibre, phenolic acids and activity of endogenous enzymes during rye bread-making". European Food Research and Technology. 214 (1): 33–42. doi:10.1007/s00217-001-0417-6. ISSN 1438-2377. S2CID 85239461.
- ^ Shahadi, Fereidoon; Naczk, Marian (2004). Phenolics in Food and Nutraceuticals. Florida: CRC Press. p. 4. ISBN 978-1-58716-138-4.
- ^ Kang, Sun-Young; Choi, Oksik; Lee, Jae Kyung; Hwang, Bang Yeon; Uhm, Tai-Boong; Hong, Young-Soo (December 2012). "Artificial biosynthesis of phenylpropanoic acids in a tyrosine overproducing Escherichia coli strain". Microbial Cell Factories. 11 (1): 153. doi:10.1186/1475-2859-11-153. PMC 3554431. PMID 23206756.
- ^ Iiyama, K.; Lam, T. B.-T.; Stone, B. A. (1994). "Covalent Cross-Links in the Cell Wall". Plant Physiology. 104 (2): 315–320. doi:10.1104/pp.104.2.315. ISSN 0032-0889. PMC 159201. PMID 12232082.
- ^ Huang, Z.; Dostal, L.; Rosazza, J. P. (1993). "Microbial transformations of ferulic acid by Saccharomyces cerevisiae and Pseudomonas fluorescens ". Applied and Environmental Microbiology. 59 (7): 2244–2250. doi:10.1128/AEM.59.7.2244-2250.1993. PMC 182264. PMID 8395165.
- ^ Huang, Z.; Dostal, L.; Rosazza, J. P. (1994). "Purification and characterization of a ferulic acid decarboxylase from Pseudomonas fluorescens". Journal of Bacteriology. 176 (19): 5912–5918. doi:10.1128/jb.176.19.5912-5918.1994. PMC 196807. PMID 7928951.
- ^ Kalogeraki, Virginia S.; Zhu, Jun; Eberhard, Anatol; Madsen, Eugene L.; Winans, Stephen C. (November 1999). "The phenolic vir gene inducer ferulic acid is O-demethylated by the VirH2 protein of an Agrobacterium tumefaciens Ti plasmid". Molecular Microbiology. 34 (3): 512–522. doi:10.1046/j.1365-2958.1999.01617.x. PMID 10564493. S2CID 28658847.
- ^ Buranov, Anvar U.; Mazza, G. (2009). "Extraction and purification of ferulic acid from flax shives, wheat and corn bran by alkaline hydrolysis and pressurised solvents". Food Chemistry. 115 (4): 1542–1548. doi:10.1016/j.foodchem.2009.01.059.
