62.99.192.174 (talk) No edit summary |
31.52.113.206 (talk) No edit summary |
||
| Line 11: | Line 11: | ||
| ImageAlt2 = Sample of compound |
| ImageAlt2 = Sample of compound |
||
| PIN = (2''E'')-3-Phenylprop-2-enoic acid |
| PIN = (2''E'')-3-Phenylprop-2-enoic acid |
||
| OtherNames = Cinnamic acid<br>''trans''-Cinnamic acid<br>Phenylacrylic acid<ref>{{cite EB1911|wstitle=Cinnamic Acid|volume=6|page=376|short=1}}</ref><br>Cinnamylic acid<br>3-Phenylacrylic acid<br />(E)-Cinnamic acid<br>Benzenepropenoic acid<br>Isocinnamic acid |
| OtherNames = Cinnamic acid<br>''trans''-Cinnamic acid<br>Phenylacrylic acid<ref>{{cite EB1911|wstitle=Cinnamic Acid|volume=6|page=376|short=1}}</ref><br>Cinnamylic acid<br>3-Phenylacrylic acid<br />(''E'')-Cinnamic acid<br>Benzenepropenoic acid<br>Isocinnamic acid |
||
|Section1={{Chembox Identifiers |
|Section1={{Chembox Identifiers |
||
| IUPHAR_ligand = 3203 |
| IUPHAR_ligand = 3203 |
||
| Line 45: | Line 45: | ||
| BoilingPt_ref = <ref name=GESTIS/> |
| BoilingPt_ref = <ref name=GESTIS/> |
||
| pKa = 4.44 |
| pKa = 4.44 |
||
| MagSus = - |
| MagSus = {{val|-7.836e-5|u=cm<sup>3</sup>/mol}} |
||
}} |
}} |
||
|Section7={{Chembox Hazards |
|Section7={{Chembox Hazards |
||
| Line 64: | Line 64: | ||
}} |
}} |
||
'''Cinnamic acid''' is an [[organic compound]] with the formula C<sub>6</sub>H<sub>5</sub> |
'''Cinnamic acid''' is an [[organic compound]] with the formula [[phenyl|C<sub>6</sub>H<sub>5</sub>]]CH=CHCOOH. It is a white crystalline compound that is slightly [[soluble]] in water, and freely soluble in many organic solvents.<ref name=Merck/> Classified as an unsaturated [[carboxylic acid]], it occurs naturally in a number of plants. It exists as both a [[Cis–trans isomerism|''cis'' and a ''trans'' isomer]], although the latter is more common.<ref name=Ullmann/> |
||
==Occurrence and production== |
==Occurrence and production== |
||
===Biosynthesis=== |
===Biosynthesis=== |
||
Cinnamic acid is a central intermediate in the biosynthesis of myriad natural products include lignols (precursors to [[lignin]] and [[lignocellulose]]), [[flavonoids]], [[isoflavonoids]], [[coumarin]]s, [[aurones]], [[stilbenes]], [[catechin]], and [[phenylpropanoid]]s. Its [[biosynthesis]] is performed by action of the [[enzyme]] [[phenylalanine ammonia-lyase]] (PAL) on [[phenylalanine]].<ref>{{cite journal|journal=Molecular Plant|year=2010|pages=2-20|doi=10.1093/mp/ssp106|title=Phenylpropanoid Biosynthesis| |
Cinnamic acid is a central intermediate in the biosynthesis of myriad natural products include lignols (precursors to [[lignin]] and [[lignocellulose]]), [[flavonoids]], [[isoflavonoids]], [[coumarin]]s, [[aurones]], [[stilbenes]], [[catechin]], and [[phenylpropanoid]]s. Its [[biosynthesis]] is performed by action of the [[enzyme]] [[phenylalanine ammonia-lyase]] (PAL) on [[phenylalanine]].<ref>{{cite journal|journal=Molecular Plant|year=2010|pages=2-20|doi=10.1093/mp/ssp106|title=Phenylpropanoid Biosynthesis|last=Vogt|first=T.}}</ref> |
||
===Natural occurrence=== |
===Natural occurrence=== |
||
It is obtained from oil of [[cinnamon]], or from [[balsam]]s such as [[storax]].<ref name=Merck>{{Merck12th}}</ref> It is also found in [[shea butter]]. Cinnamic acid has a honey-like odor;<ref> |
It is obtained from oil of [[cinnamon]], or from [[balsam]]s such as [[storax]].<ref name=Merck>{{Merck12th}}</ref> It is also found in [[shea butter]]. Cinnamic acid has a [[honey]]-like odor;<ref>{{cite web|url=http://www.flavornet.org/info/140-10-3.html|title=Cinnamic acid|website=flavornet.org}}</ref> it and its more volatile ethyl ester ([[ethyl cinnamate]]) are flavor components in the essential oil of [[cinnamon]], in which related [[cinnamaldehyde]] is the major constituent. |
||
===Production=== |
===Production=== |
||
The original synthesis of cinnamic acid involves the [[Perkin reaction]], which entails the base-catalysed condensation of [[acetic anhydride]] and benzaldehyde. [[Rainer Ludwig Claisen]] (1851–1930) described the synthesis of cinnamate |
The original synthesis of cinnamic acid involves the [[Perkin reaction]], which entails the base-catalysed [[condensation reaction|condensation]] of [[acetic anhydride]] and [[benzaldehyde]]. [[Rainer Ludwig Claisen]] (1851–1930) described the synthesis of cinnamate [[ester]]s by the reaction of benzaldehyde and esters.<ref>{{cite journal|last=Claisen|first=L.|date=1890|url=https://babel.hathitrust.org/cgi/pt?id=uc1.b3481786;view=1up;seq=988|title=Zur Darstellung der Zimmtsäure und ihrer Homologen|trans-title=On the preparation of cinnamic acid and its homologues|journal=Berichte der deutschen chemischen Gesellschaft|volume=23|page=976–978}}</ref> The reaction is known as the [[aldol condensation]] (with accompanying [[hydrolysis]] of the anhydride). |
||
:[[File:Zimtsäure nach Perkin.svg|450px|center|thumb|Synthesis of cinnamic acid via Perkin reaction]] |
:[[File:Zimtsäure nach Perkin.svg|450px|center|thumb|Synthesis of cinnamic acid via the [[Perkin reaction]]]] |
||
It can also be prepared from cinnamaldehyde and benzal chloride.<ref name=Ullmann>Dorothea |
It can also be prepared from [[cinnamaldehyde]] and [[benzal chloride]].<ref name=Ullmann>{{Ullmann|first=Dorothea|last=Garbe|title=Cinnamic Acid|doi=10.1002/14356007.a07_099}}</ref> |
||
Another way of preparing |
Another way of preparing cinnamic acid is by the Knövenaegel–Hans condensation reaction.<ref>{{cite book|first=L.|last=Tieze|title=Reactions and Synthesis in the Organic Chemistry Laboratory|location=Mill Vall, CA|date=1988|page=1988}}</ref> The reactants for this are the corresponding benzaldehyde and [[malonic acid]] in the presence of a weak base, followed by acid hydrolysis. |
||
==Uses== |
==Uses== |
||
Cinnamic acid is used in |
Cinnamic acid is used in flavorings, [[Indigo dye#Chemical synthesis|synthetic indigo]], and certain [[pharmaceuticals]]. A major use is in the manufacturing of the methyl, ethyl, and benzyl [[ester]]s for the perfume industry.<ref name=Merck/> Cinnamic acid is a precursor to the sweetener [[aspartame]] via enzyme-catalysed amination to [[phenylalanine]].<ref name=Ullmann/> Cinnamic acid can [[Dimer_(chemistry)|dimerize]] in non-polar solvents resulting in different [[Free-energy_relationship|linear free energy relationships]].<ref>{{cite journal | doi = 10.1186/s13065-015-0080-9 | pmid = 25798191 | pmc = 4369286 | title = Determination of Abraham model solute descriptors for the monomeric and dimeric forms of ''trans''-cinnamic acid using measured solubilities from the Open Notebook Science Challenge | journal = Chemistry Central Journal | volume = 9 | pages = 11 | year = 2015 | last1 = Bradley | first1 = J.-C. | last2 = Abraham | first2 = M. H. | last3 = Acree | first3 = W. E. | last4 = Lang | first4 = A. | last5 = Beck | first5 = S. N. | last6 = Bulger | first6 = D. A. | last7 = Clark | first7 = E. A. | last8 = Condron | first8 = L. N. | last9 = Costa | first9 = S. T. | last10 = Curtin | first10 = E. M. | last11 = Kurtu | first11 = S. B. | last12 = Mangir | first12 = M. I. | last13 = McBride | first13 = M. J. }}</ref> |
||
==References== |
==References== |
||
{{Reflist}} |
{{Reflist|30em}} |
||
{{Hydroxycinnamic acid}} |
{{Hydroxycinnamic acid}} |
||
Revision as of 13:24, 16 August 2018

| |

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
(2E)-3-Phenylprop-2-enoic acid | |
| Other names
Cinnamic acid
trans-Cinnamic acid Phenylacrylic acid[1] Cinnamylic acid 3-Phenylacrylic acid (E)-Cinnamic acid Benzenepropenoic acid Isocinnamic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.908 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H8O2 | |
| Molar mass | 148.161 g·mol−1 |
| Appearance | White monoclinic crystals |
| Density | 1.2475 g/cm3[2] |
| Melting point | 133 °C (271 °F; 406 K)[2] |
| Boiling point | 300 °C (572 °F; 573 K)[2] |
| 500 mg/L[2] | |
| Acidity (pKa) | 4.44 |
| −7.836×10−5 cm3/mol | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | > 100 °C (212 °F; 373 K)[2] |
| Related compounds | |
Related compounds
|
Benzoic acid, Phenylacetic acid, Phenylpropanoic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cinnamic acid is an organic compound with the formula C6H5CH=CHCOOH. It is a white crystalline compound that is slightly soluble in water, and freely soluble in many organic solvents.[3] Classified as an unsaturated carboxylic acid, it occurs naturally in a number of plants. It exists as both a cis and a trans isomer, although the latter is more common.[4]
Occurrence and production
Biosynthesis
Cinnamic acid is a central intermediate in the biosynthesis of myriad natural products include lignols (precursors to lignin and lignocellulose), flavonoids, isoflavonoids, coumarins, aurones, stilbenes, catechin, and phenylpropanoids. Its biosynthesis is performed by action of the enzyme phenylalanine ammonia-lyase (PAL) on phenylalanine.[5]
Natural occurrence
It is obtained from oil of cinnamon, or from balsams such as storax.[3] It is also found in shea butter. Cinnamic acid has a honey-like odor;[6] it and its more volatile ethyl ester (ethyl cinnamate) are flavor components in the essential oil of cinnamon, in which related cinnamaldehyde is the major constituent.
Production
The original synthesis of cinnamic acid involves the Perkin reaction, which entails the base-catalysed condensation of acetic anhydride and benzaldehyde. Rainer Ludwig Claisen (1851–1930) described the synthesis of cinnamate esters by the reaction of benzaldehyde and esters.[7] The reaction is known as the aldol condensation (with accompanying hydrolysis of the anhydride).
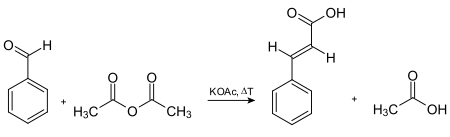
Synthesis of cinnamic acid via the Perkin reaction
It can also be prepared from cinnamaldehyde and benzal chloride.[4]
Another way of preparing cinnamic acid is by the Knövenaegel–Hans condensation reaction.[8] The reactants for this are the corresponding benzaldehyde and malonic acid in the presence of a weak base, followed by acid hydrolysis.
Uses
Cinnamic acid is used in flavorings, synthetic indigo, and certain pharmaceuticals. A major use is in the manufacturing of the methyl, ethyl, and benzyl esters for the perfume industry.[3] Cinnamic acid is a precursor to the sweetener aspartame via enzyme-catalysed amination to phenylalanine.[4] Cinnamic acid can dimerize in non-polar solvents resulting in different linear free energy relationships.[9]
References
- ^ . Encyclopædia Britannica. Vol. 6 (11th ed.). 1911. p. 376.
- ^ a b c d e Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ a b c Budavari, Susan, ed. (1996), The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (12th ed.), Merck, ISBN 0911910123
- ^ a b c Garbe, Dorothea. "Cinnamic Acid". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a07_099. ISBN 978-3527306732.
- ^ Vogt, T. (2010). "Phenylpropanoid Biosynthesis". Molecular Plant: 2–20. doi:10.1093/mp/ssp106.
- ^ "Cinnamic acid". flavornet.org.
- ^ Claisen, L. (1890). "Zur Darstellung der Zimmtsäure und ihrer Homologen" [On the preparation of cinnamic acid and its homologues]. Berichte der deutschen chemischen Gesellschaft. 23: 976–978.
- ^ Tieze, L. (1988). Reactions and Synthesis in the Organic Chemistry Laboratory. Mill Vall, CA. p. 1988.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Bradley, J.-C.; Abraham, M. H.; Acree, W. E.; Lang, A.; Beck, S. N.; Bulger, D. A.; Clark, E. A.; Condron, L. N.; Costa, S. T.; Curtin, E. M.; Kurtu, S. B.; Mangir, M. I.; McBride, M. J. (2015). "Determination of Abraham model solute descriptors for the monomeric and dimeric forms of trans-cinnamic acid using measured solubilities from the Open Notebook Science Challenge". Chemistry Central Journal. 9: 11. doi:10.1186/s13065-015-0080-9. PMC 4369286. PMID 25798191.
{{cite journal}}: CS1 maint: unflagged free DOI (link)
