Content deleted Content added
m clean up, References after punctuation per WP:REFPUNC and WP:PAIC using AWB (8434) |
m Open access bot: doi updated in citation with #oabot. |
||
| (16 intermediate revisions by 13 users not shown) | |||
| Line 1: | Line 1: | ||
{{ |
{{Chembox |
||
| Watchedfields = changed |
|||
| verifiedrevid = 443408352 |
| verifiedrevid = 443408352 |
||
| Name = Ayanin |
| Name = Ayanin |
||
| ImageFile = Ayanin. |
| ImageFile = Ayanin.svg |
||
| ImageSize = 200px |
|||
| ImageName = Chemical structure of ayanin |
| ImageName = Chemical structure of ayanin |
||
| IUPACName = 5- |
| IUPACName = 3′,5-Dihydroxy-3,4′,7-trimethoxyflavone |
||
| SystematicName = 5-Hydroxy-2-(3-hydroxy-4-methoxyphenyl)-3,7-dimethoxy-4''H''-1-benzopyran-4-one |
|||
| OtherNames = 3,7,4'-Tri-O-methylquercetin<br>3,7,4'-trimethylquercetin<br>5,3'-dihydroxy-3,7,4'-trimethoxyflavone |
|||
| OtherNames = {{ubl|3,7,4'-Tri-''O''-methylquercetin|3,7,4'-Trimethylquercetin}} |
|||
|Section1= |
|Section1={{Chembox Identifiers |
||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
||
| ChemSpiderID = 4444274 |
| ChemSpiderID = 4444274 |
||
| Line 20: | Line 21: | ||
| StdInChIKey = KPCRYSMUMBNTCK-UHFFFAOYSA-N |
| StdInChIKey = KPCRYSMUMBNTCK-UHFFFAOYSA-N |
||
| CASNo = 572-32-7 |
| CASNo = 572-32-7 |
||
| CASNo_Ref = {{cascite|correct| |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| CASOther = |
|||
| UNII = YA465UF3LK |
|||
| CASNoOther = |
|||
| PubChem = 5280682 |
| PubChem = 5280682 |
||
| ChEBI_Ref = {{ebicite|correct|EBI}} |
| ChEBI_Ref = {{ebicite|correct|EBI}} |
||
| ChEBI = 27825 |
| ChEBI = 27825 |
||
| SMILES = COC1=C(C=C(C=C1)C2=C(C(=O)C3=C(C=C(C=C3O2)OC)O)OC)O |
| SMILES = COC1=C(C=C(C=C1)C2=C(C(=O)C3=C(C=C(C=C3O2)OC)O)OC)O |
||
| InChI = |
|||
| MeSHName = |
| MeSHName = |
||
}} |
}} |
||
|Section2= |
|Section2={{Chembox Properties |
||
| C=18 | H=16 | O=7 |
|||
| Formula = C<sub>18</sub>H<sub>16</sub>O<sub>7</sub> |
|||
| MolarMass = 344.31 g/mol |
|||
| ExactMass = 344.089603 u |
|||
| Appearance = |
| Appearance = |
||
| Density = |
| Density = 1.454 g/mL |
||
| MeltingPtC = |
|||
| MeltingPt = <!-- °C --> |
|||
| BoilingPtC = |
|||
| BoilingPt = <!-- °C --> |
|||
| Solubility = |
| Solubility = |
||
| ⚫ | |||
}} |
}} |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | It can be found in ''[[Croton schiedeanus]]''. It can also be |
||
| ⚫ | It can be found in ''[[Croton schiedeanus]]''. It can also be synthesized.<ref>{{cite journal | doi = 10.1002/jhet.5570130629| title = Partial methylation of quercetin: Direct synthesis of tamarixetin, ombuin and ayanin| journal = Journal of Heterocyclic Chemistry| volume = 13| issue = 6| pages = 1293–1295| year = 1976| last1 = Rao| first1 = Koppaka V.| last2 = Owoyale| first2 = Jacob A.| doi-access = free}}</ref> |
||
==Biosynthetis== |
|||
| ⚫ | |||
== |
== Biosynthesis == |
||
| ⚫ | |||
== References == |
|||
{{reflist}} |
{{reflist}} |
||
{{flavonol}} |
{{flavonol}} |
||
[[Category: |
[[Category:O-methylated flavonols]] |
||
[[Category:Phenol ethers]] |
|||
{{Natural-phenol-stub}} |
|||
{{aromatic-stub}} |
|||
[[fa:آیانین]] |
|||
[[ja:アヤニン]] |
|||
[[pl:Ajanina]] |
|||
Latest revision as of 00:08, 9 November 2023
| |
| Names | |
|---|---|
| IUPAC name
3′,5-Dihydroxy-3,4′,7-trimethoxyflavone
| |
| Systematic IUPAC name
5-Hydroxy-2-(3-hydroxy-4-methoxyphenyl)-3,7-dimethoxy-4H-1-benzopyran-4-one | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H16O7 | |
| Molar mass | 344.319 g·mol−1 |
| Density | 1.454 g/mL |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
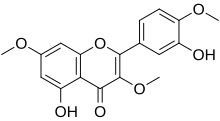
Ayanin is an O-methylated flavonol, a type of flavonoid. It is the 3,7,4'-tri-O-methylated derivative of quercetin.
It can be found in Croton schiedeanus. It can also be synthesized.[1]
Biosynthesis[edit]
The enzyme 3,7-dimethylquercetin 4'-O-methyltransferase uses S-adenosyl methionine and rhamnazin to produce S-adenosylhomocysteine and ayanin.
References[edit]
- ^ Rao, Koppaka V.; Owoyale, Jacob A. (1976). "Partial methylation of quercetin: Direct synthesis of tamarixetin, ombuin and ayanin". Journal of Heterocyclic Chemistry. 13 (6): 1293–1295. doi:10.1002/jhet.5570130629.