| |
| Names | |
|---|---|
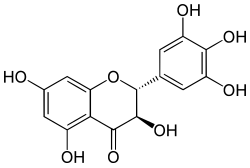
| IUPAC name
(2R,3R)-3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-2,3-dihydrochromen-4-one
| |
| Other names
Dihydromyricetin, Ampeloptin,(+)-Ampelopsin,(+)-Dihydromyricetin
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H12O8 | |
| Molar mass | 320.253 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ampelopsin, sometimes referred to as dihydromyricetin or DHM, is a flavan-on-ol, a type of flavonoid. It shares it's hydroxy-group pattern with myricetin, but since ampelopsin is missing the double-bond in the C-ring, it has stereoisomers that are not possible in myricetin. In plant synthesis pathways, ampelopsin is upstream of myricetin; myricetin may be formed from ampelopsin by the plant enzyme flavonol synthase (FLS)[1], or ampelopsin may enter other pathways. When glycosylated, it commonly happens at the C-ring 3-position. It is found in the Ampelopsis species japonica, megalophylla, and grossedentata; Cercidiphyllum japonicum; Hovenia dulcis; Rhododendron cinnabarinum; some Pinus species; and some Cedrus species,[2] as well as in Salix sachalinensis.[3]
Hovenia dulcis has been used in traditional Japanese, Chinese, and Korean medicines to treat fever, parasitic infection, as a laxative, and a treatment of liver diseases, and as a hangover treatment.[4] Methods have been developed to extract ampelopsin from it at large scales, and laboratory research has been conducted with the compound to see if it might be useful as a drug in any of the conditions for which the parent plant has been traditionally used.[4]
In a trial of sixty patients with fatty liver disease dihydromyricetin improved glucose and lipid metabolism and exerted anti-inflammatory effects which were beneficial.[5]
References
- ^ "The Biochemistry of the Grape Berry" https://books.google.com/books?isbn=1608053601
- ^ Zhou, Jiaju; Xie, Guirong; Yan, Xinjian (2011-02-21). Encyclopedia of Traditional Chinese Medicines – Molecular Structures, Pharmacological Activities, Natural Sources and Applications: Vol. 1: Isolated Compounds A-C. Springer Science & Business Media. ISBN 978-3-642-16735-5.
- ^ Tahara, Satoshi (June 23, 2007). "A Journey of Twenty-Five Years through the Ecological Biochemistry of Flavonoids". Bioscience, Biotechnology, and Biochemistry. 71 (6): 1387–1404. doi:10.1271/bbb.70028. ISSN 0916-8451. PMID 17587669.
- ^ a b Hyun, Tae; Eom, Seung; Yu, Chang; Roitsch, Thomas (2010). "Hovenia dulcis– An Asian Traditional Herb". Planta Medica. 76 (10): 943–949. doi:10.1055/s-0030-1249776. ISSN 0032-0943. PMID 20379955.
- ^ Chen, Shihui; Zhao, Xiaolan; Wan, Jing; Ran, Li; Qin, Yu; Wang, Xiaofang; Gao, Yanxiang; Shu, Furong; Zhang, Yong; Liu, Peng; Zhang, Qianyong; Zhu, Jundong; Mi, Mantian (2015). "Dihydromyricetin improves glucose and lipid metabolism and exerts anti-inflammatory effects in nonalcoholic fatty liver disease: A randomized controlled trial". Pharmacological Research. 99: 74–81. doi:10.1016/j.phrs.2015.05.009. ISSN 1043-6618.