2600:1700:1d1:dc40:d05c:9530:1fde:795a (talk) No edit summary Tags: Visual edit Mobile edit Mobile web edit |
m source "EncTCM" +page (the rest is only all sources excess blanks removed) |
||
| (12 intermediate revisions by 9 users not shown) | |||
| Line 1: | Line 1: | ||
{{ |
{{Other uses|Ampelopsin (compound)}} |
||
{{chembox |
{{chembox |
||
| Verifiedfields = changed |
| Verifiedfields = changed |
||
| Line 6: | Line 6: | ||
| ImageFile = Ampelopsin.svg |
| ImageFile = Ampelopsin.svg |
||
| ImageSize = 250px |
| ImageSize = 250px |
||
| IUPACName = (2''R'',3''R'')-3, |
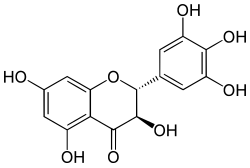
| IUPACName = (2''R'',3''R'')-3,3′,4′,5,5′,7-Hexahydroxyflavan-4-one |
||
| SystematicName = (2''R'',3''R'')-3,5,7-Trihydroxy-2-(3,4,5-trihydroxy)-2,3-dihydro-4''H''-1-benzopyran-4-one |
|||
| OtherNames = Dihydromyricetin, Ampeloptin,(+)-Ampelopsin,(+)-Dihydromyricetin |
| OtherNames = Dihydromyricetin, Ampeloptin,(+)-Ampelopsin,(+)-Dihydromyricetin |
||
|Section1={{Chembox Identifiers |
|Section1={{Chembox Identifiers |
||
| Line 19: | Line 20: | ||
| CASNo_Ref = {{cascite|correct|??}} |
| CASNo_Ref = {{cascite|correct|??}} |
||
| CASNo = 27200-12-0 |
| CASNo = 27200-12-0 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| UNII = KD8QND6427 |
|||
| PubChem = 161557 |
| PubChem = 161557 |
||
| ChEBI_Ref = {{ebicite|changed|EBI}} |
| ChEBI_Ref = {{ebicite|changed|EBI}} |
||
| Line 38: | Line 41: | ||
}} |
}} |
||
}} |
}} |
||
| ⚫ | '''Ampelopsin''', also known as '''dihydromyricetin''' and '''DHM''', when purported as an effective ingredient in supplements and other tonics, is a [[flavanonol]], a type of flavonoid. It is |
||
| ⚫ | '''Ampelopsin''', also known as '''dihydromyricetin''' and '''DHM''', when purported as an effective ingredient in supplements and other tonics, is a [[flavanonol]], a type of flavonoid. It is extracted from the Japanese raisin tree and found in ''[[Ampelopsis]]'' species ''japonica'', ''megalophylla'', and ''grossedentata''; ''[[Cercidiphyllum japonicum]]''; ''[[Hovenia dulcis]]''; ''[[Rhododendron cinnabarinum]]''; some ''[[Pinus]]'' species; and some ''[[Cedrus]]'' species,<ref name="EncTCM">{{cite book |title=Encyclopedia of Traditional Chinese Medicines – Molecular Structures, Pharmacological Activities, Natural Sources and Applications: Vol. 1: Isolated Compounds A-C |url=https://books.google.com/books?id=PMsXJnUYTFkC |publisher=Springer Science & Business Media |date=2011-02-21 |isbn=978-3-642-16735-5 |first1=Jiaju |last1=Zhou |first2=Guirong |last2=Xie |first3=Xinjian |last3=Yan |page=123}}</ref> as well as in ''[[Salix sachalinensis]]''.<ref name="pmid17587669">{{cite journal |vauthors=Tahara S |title=A journey of twenty-five years through the ecological biochemistry of flavonoids |journal=Biosci Biotechnol Biochem |volume=71 |issue=6 |pages=1387–404 |date=June 2007 |pmid=17587669 |doi=10.1271/bbb.70028 |s2cid=35670587|doi-access=free }}</ref> |
||
| ⚫ | ''[[Hovenia dulcis]]'' has been used in traditional [[Kampo|Japanese]], [[Traditional chinese medicine|Chinese]], and [[Traditional Korean medicine|Korean]] medicines to treat fever, parasitic infection, as a laxative, and a treatment of liver diseases, and as a [[hangover]] treatment.<ref name=Hyun>{{cite journal| |
||
| ⚫ | ''[[Hovenia dulcis]]'' has been used in traditional [[Kampo|Japanese]], [[Traditional chinese medicine|Chinese]], and [[Traditional Korean medicine|Korean]] medicines to treat fever, parasitic infection, as a laxative, and a treatment of liver diseases, and as a [[hangover]] treatment.<ref name=Hyun>{{cite journal |vauthors=Hyun TK, Eom SH, Yu CY, Roitsch T |title=Hovenia dulcis--an Asian traditional herb |journal=Planta Med |volume=76 |issue=10 |pages=943–9 |date=July 2010 |pmid=20379955 |doi=10.1055/s-0030-1249776 |doi-access=free}}</ref> Methods have been developed to extract ampelopsin on a larger scale, and laboratory research has been conducted with the compound to see if it might be useful as a drug in any of the conditions for which the parent plant has been traditionally used.<ref name=Hyun /> |
||
<big>'''Scientific peer-reviewed research'''</big> |
|||
== Research == |
|||
| ⚫ | In a trial of 60 patients with "[[Non-alcoholic fatty liver disease|nonalcoholic fatty liver disease]]," dihydromyricetin improved glucose and lipid metabolism and yielded potentially beneficial anti-inflammatory effects.<ref name="ChenZhao2015">{{cite journal| |
||
Research suggests that DHM protects against [[Doxorubicin| DOX-induced cardiotoxicity]] by inhibiting [[NLRP3]] inflammasome activation via stimulation of the [[SIRT1]] pathway.<ref name="pmid33795647">{{cite journal |vauthors=Christidi E, Brunham LR |title=Regulated cell death pathways in doxorubicin-induced cardiotoxicity |journal=Cell Death Dis |volume=12 |issue=4 |pages=339 |date=April 2021 |pmid=33795647 |pmc=8017015 |doi=10.1038/s41419-021-03614-x }}</ref> |
|||
| ⚫ | In a trial of 60 patients with "[[Non-alcoholic fatty liver disease|nonalcoholic fatty liver disease]]," dihydromyricetin improved glucose and lipid metabolism and yielded potentially beneficial anti-inflammatory effects.<ref name="ChenZhao2015">{{cite journal |vauthors=Chen S, Zhao X, Wan J, Ran L, Qin Y, Wang X, Gao Y, Shu F, Zhang Y, Liu P, Zhang Q, Zhu J, Mi M |title=Dihydromyricetin improves glucose and lipid metabolism and exerts anti-inflammatory effects in nonalcoholic fatty liver disease: A randomized controlled trial |journal=Pharmacol Res |volume=99 |issue= |pages=74–81 |date=September 2015 |pmid=26032587 |doi=10.1016/j.phrs.2015.05.009 |url=}}</ref> |
||
In a study wherein the subjects were rats, researchers demonstrated "pharmacological properties of dihydromyricetin consistent with those expected to underlie successful medical treatment of [[alcohol use disorders]]; therefore dihydromyricetin is a therapeutic candidate."<ref>{{Cite journal |
|||
| author = Yi Shen, A. Kerstin Lindemeyer, Claudia Gonzalez, Xuesi M. Shao, Igor Spigelman, Richard W. Olsen & Jing Liang |
|||
| title = Dihydromyricetin as a novel anti-alcohol intoxication medication |
|||
| journal = [[The Journal of Neuroscience]] |
|||
| volume = 32 |
|||
| issue = 1 |
|||
| pages = 390–401 |
|||
| year = 2012 |
|||
| doi = 10.1523/JNEUROSCI.4639-11.2012 |
|||
| pmid = 22219299 |
|||
| pmc = 3292407 |
|||
}}</ref> |
|||
A study of rats demonstrated pharmacological properties of DHM which suggest it would be a therapeutic candidate to treat [[alcohol use disorders]].<ref name="pmid22219299">{{cite journal |vauthors=Shen Y, Lindemeyer AK, Gonzalez C, Shao XM, Spigelman I, Olsen RW, Liang J |title=Dihydromyricetin as a novel anti-alcohol intoxication medication |journal=J Neurosci |volume=32 |issue=1 |pages=390–401 |date=January 2012 |pmid=22219299 |pmc=3292407 |doi=10.1523/JNEUROSCI.4639-11.2012 }}</ref> |
|||
"Structurally, due to the highly hydrophilic character, dihydromyricetin shows poor '''[[bioavailability]]''' and significantly limits its potential medicinal applications."<ref>{{Cite journal|last=Li|first=Hongliang|last2=Li|first2=Qisheng|last3=Liu|first3=Zhaowen|last4=Yang|first4=Kai|last5=Chen|first5=Zhixi|last6=Cheng|first6=Qilai|last7=Wu|first7=Longhuo|date=2017|title=The Versatile Effects of Dihydromyricetin in Health|url=https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5602609/|journal=Evidence-based Complementary and Alternative Medicine : eCAM|volume=2017|doi=10.1155/2017/1053617|issn=1741-427X|pmc=5602609|pmid=28947908}}</ref> "In pharmacology, '''bioavailability''' [...] is a subcategory of absorption and is the fraction (%) of an administered drug that reaches the systemic circulation."<ref>{{Cite web|url=https://en.m.wikipedia.org/wiki/Bioavailability|title=Bioavailability - Wikipedia|website=en.m.wikipedia.org|language=en|access-date=2020-04-28}}</ref> |
|||
Dihydromyricetin shows poor [[bioavailability]] which limits its potential medicinal applications.<ref name="pmid28947908">{{cite journal |vauthors=Li H, Li Q, Liu Z, Yang K, Chen Z, Cheng Q, Wu L |title=The Versatile Effects of Dihydromyricetin in Health |journal=Evid Based Complement Alternat Med |volume=2017 |issue= |pages=1053617 |date=2017 |pmid=28947908 |pmc=5602609 |doi=10.1155/2017/1053617 |doi-access=free }}</ref> |
|||
| ⚫ | |||
| ⚫ | |||
== References == |
== References == |
||
{{Reflist |
{{Reflist}} |
||
{{Flavanonol}} |
|||
{{GABAAR PAMs}} |
{{GABAAR PAMs}} |
||
Latest revision as of 05:59, 4 October 2023
| |
| Names | |
|---|---|
| IUPAC name
(2R,3R)-3,3′,4′,5,5′,7-Hexahydroxyflavan-4-one
| |
| Systematic IUPAC name
(2R,3R)-3,5,7-Trihydroxy-2-(3,4,5-trihydroxy)-2,3-dihydro-4H-1-benzopyran-4-one | |
| Other names
Dihydromyricetin, Ampeloptin,(+)-Ampelopsin,(+)-Dihydromyricetin
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H12O8 | |
| Molar mass | 320.253 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ampelopsin, also known as dihydromyricetin and DHM, when purported as an effective ingredient in supplements and other tonics, is a flavanonol, a type of flavonoid. It is extracted from the Japanese raisin tree and found in Ampelopsis species japonica, megalophylla, and grossedentata; Cercidiphyllum japonicum; Hovenia dulcis; Rhododendron cinnabarinum; some Pinus species; and some Cedrus species,[1] as well as in Salix sachalinensis.[2]
Hovenia dulcis has been used in traditional Japanese, Chinese, and Korean medicines to treat fever, parasitic infection, as a laxative, and a treatment of liver diseases, and as a hangover treatment.[3] Methods have been developed to extract ampelopsin on a larger scale, and laboratory research has been conducted with the compound to see if it might be useful as a drug in any of the conditions for which the parent plant has been traditionally used.[3]
Research[edit]
Research suggests that DHM protects against DOX-induced cardiotoxicity by inhibiting NLRP3 inflammasome activation via stimulation of the SIRT1 pathway.[4]
In a trial of 60 patients with "nonalcoholic fatty liver disease," dihydromyricetin improved glucose and lipid metabolism and yielded potentially beneficial anti-inflammatory effects.[5]
A study of rats demonstrated pharmacological properties of DHM which suggest it would be a therapeutic candidate to treat alcohol use disorders.[6]
Dihydromyricetin shows poor bioavailability which limits its potential medicinal applications.[7]
Additional research is required before claims of human efficacy and application, necessary dosage, and solutions to poor bioavailability, are met with scientific validation.
References[edit]
- ^ Zhou, Jiaju; Xie, Guirong; Yan, Xinjian (2011-02-21). Encyclopedia of Traditional Chinese Medicines – Molecular Structures, Pharmacological Activities, Natural Sources and Applications: Vol. 1: Isolated Compounds A-C. Springer Science & Business Media. p. 123. ISBN 978-3-642-16735-5.
- ^ Tahara S (June 2007). "A journey of twenty-five years through the ecological biochemistry of flavonoids". Biosci Biotechnol Biochem. 71 (6): 1387–404. doi:10.1271/bbb.70028. PMID 17587669. S2CID 35670587.
- ^ a b Hyun TK, Eom SH, Yu CY, Roitsch T (July 2010). "Hovenia dulcis--an Asian traditional herb". Planta Med. 76 (10): 943–9. doi:10.1055/s-0030-1249776. PMID 20379955.
- ^ Christidi E, Brunham LR (April 2021). "Regulated cell death pathways in doxorubicin-induced cardiotoxicity". Cell Death Dis. 12 (4): 339. doi:10.1038/s41419-021-03614-x. PMC 8017015. PMID 33795647.
- ^ Chen S, Zhao X, Wan J, Ran L, Qin Y, Wang X, Gao Y, Shu F, Zhang Y, Liu P, Zhang Q, Zhu J, Mi M (September 2015). "Dihydromyricetin improves glucose and lipid metabolism and exerts anti-inflammatory effects in nonalcoholic fatty liver disease: A randomized controlled trial". Pharmacol Res. 99: 74–81. doi:10.1016/j.phrs.2015.05.009. PMID 26032587.
- ^ Shen Y, Lindemeyer AK, Gonzalez C, Shao XM, Spigelman I, Olsen RW, Liang J (January 2012). "Dihydromyricetin as a novel anti-alcohol intoxication medication". J Neurosci. 32 (1): 390–401. doi:10.1523/JNEUROSCI.4639-11.2012. PMC 3292407. PMID 22219299.
- ^ Li H, Li Q, Liu Z, Yang K, Chen Z, Cheng Q, Wu L (2017). "The Versatile Effects of Dihydromyricetin in Health". Evid Based Complement Alternat Med. 2017: 1053617. doi:10.1155/2017/1053617. PMC 5602609. PMID 28947908.