Content deleted Content added
OrganoMetallurgy (talk | contribs) removed Category:Alkenes using HotCat |
Shinkolobwe (talk | contribs) Adding short description: "Organosulfur compound", overriding automatically generated description Tag: Shortdesc helper |
||
| (6 intermediate revisions by 5 users not shown) | |||
| Line 1: | Line 1: | ||
{{Short description|Organosulfur compound}} |
|||
{{Chembox |
{{Chembox |
||
| Verifiedfields = changed |
| Verifiedfields = changed |
||
| Watchedfields = changed |
| Watchedfields = changed |
||
| verifiedrevid = 477314172 |
| verifiedrevid = 477314172 |
||
| ImageFile = |
| ImageFile = Allylmethyl sulfide Structural Formula V1.svg |
||
| ImageFile_Ref = {{chemboximage|correct|??}} |
| ImageFile_Ref = {{chemboximage|correct|??}} |
||
| ImageSize = 244 |
| ImageSize = 244 |
||
| ImageName = XYZ |
| ImageName = XYZ |
||
| PIN = 3- |
| PIN = 3-(Methylsulfanyl)prop-1-ene |
||
| OtherNames = Methyl propenyl sulfide<br /> |
| OtherNames = Methyl propenyl sulfide<br /> |
||
3-Methylthio-1-propene |
3-Methylthio-1-propene |
||
| Line 47: | Line 48: | ||
}} |
}} |
||
'''Allyl methyl sulfide''' |
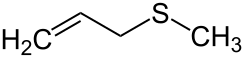
'''Allyl methyl sulfide''' is an [[organosulfur compound]] with the [[chemical formula]] CH<sub>2</sub>=CHCH<sub>2</sub>SCH<sub>3</sub>. The molecule features two [[functional group]]s, an [[allyl]] (CH<sub>2</sub>=CHCH<sub>2</sub>) and a [[thioether|sulfide]]. It is a colourless liquid with a strong odor characteristic of alkyl sulfides. It is a metabolite of [[garlic]], and "[[garlic breath]]" is attributed to its presence.<ref>{{cite book| author = Eric Block| title = Garlic and Other Alliums: The Lore and the Science| date = 2010-01-04| publisher = Royal Society of Chemistry| isbn = 978-0-85404-190-9 }}</ref> |
||
It is prepared by the reaction of [[allyl chloride]] with [[sodium hydroxide]] and [[methanethiol]]. |
It is prepared by the reaction of [[allyl chloride]] with [[sodium hydroxide]] and [[methanethiol]]. |
||
:CH<sub>2</sub>=CHCH<sub>2</sub>Cl + NaOH(aq) + CH<sub>3</sub>SH → CH<sub>2</sub>=CHCH<sub>2</sub>SCH<sub>3</sub> + NaCl + |
:CH<sub>2</sub>=CHCH<sub>2</sub>Cl + NaOH (aq) + CH<sub>3</sub>SH → CH<sub>2</sub>=CHCH<sub>2</sub>SCH<sub>3</sub> + NaCl + H<sub>2</sub>O |
||
==References== |
==References== |
||
Latest revision as of 20:49, 2 August 2022
| |
| Names | |
|---|---|
| Preferred IUPAC name
3-(Methylsulfanyl)prop-1-ene | |
| Other names
Methyl propenyl sulfide
3-Methylthio-1-propene | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.030.371 |
| EC Number |
|
| MeSH | allyl+methyl+sulfide |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1993 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H8S | |
| Molar mass | 88.17 g·mol−1 |
| Odor | Garlic |
| Density | 0.803 g cm−3 |
| Boiling point | 92 °C; 197 °F; 365 K |
| Hazards | |
| GHS labelling: | |

| |
| Danger | |
| H225 | |
| P210 | |
| Flash point | 18.0 °C (64.4 °F; 291.1 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Allyl methyl sulfide is an organosulfur compound with the chemical formula CH2=CHCH2SCH3. The molecule features two functional groups, an allyl (CH2=CHCH2) and a sulfide. It is a colourless liquid with a strong odor characteristic of alkyl sulfides. It is a metabolite of garlic, and "garlic breath" is attributed to its presence.[1]
It is prepared by the reaction of allyl chloride with sodium hydroxide and methanethiol.
- CH2=CHCH2Cl + NaOH (aq) + CH3SH → CH2=CHCH2SCH3 + NaCl + H2O