m Dating maintenance tags: {{Citation needed}} |
The Nth User (talk | contribs) →External links: Added one category |
||
| Line 70: | Line 70: | ||
[[Category:Triols]] |
[[Category:Triols]] |
||
[[Category:Secondary alcohols]] |
[[Category:Secondary alcohols]] |
||
[[Category:Vinylogous carboxylic acid]] |
|||
Revision as of 00:19, 25 May 2019
| |

| |
| Names | |
|---|---|
| IUPAC name
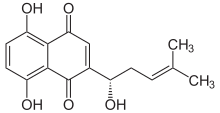
5,8-Dihydroxy-2-[(1S)-1-hydroxy-4-methylpent-3-en-1-yl]naphthalene-1,4-dione
| |
| Other names
C.I. Natural red 20
Alkanet extract Anchusaic acid Anchusin | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.497 |
| E number | E103 (colours) |
| KEGG | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties[1] | |
| C16H16O5 | |
| Molar mass | 288.299 g·mol−1 |
| Appearance | Red-brown crystalline prisms |
| Density | 1.15 g/mL |
| Melting point | 149 °C (300 °F; 422 K) |
| Boiling point | 567 °C (1,053 °F; 840 K) |
| Sparingly soluble | |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
3.0 g/kg (mice) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Alkannin is a natural dye that is obtained from the extracts of plants from the borage family Alkanna tinctoria that are found in the south of France. The dye is used as a food coloring and in cosmetics. It is used as a red-brown food additive in regions such as Australia,[2] and is designated in Europe as the E number E103,[citation needed] but is no longer approved for use.[3] Alkannin has a deep red color in a greasy or oily environment and a violet color in an alkaline environment.[citation needed]
The chemical structure as a naphthoquinone derivative was first determined by Brockmann in 1936.[4] The enantiomer of alkannin is known as shikonin, and the racemic mixture of the two is known as shikalkin.[5][6]
The enzyme 4-hydroxybenzoate geranyltransferase utilizes geranyl diphosphate and 4-hydroxybenzoate to produce 3-geranyl-4-hydroxybenzoate and diphosphate. These compounds are then used to form alkannin.[6]
Alkannin is an antioxidant[7] and has an antimicrobial effect against Staphylococcus aureus and Staphylococcus epidermidis. It is also known to have wound healing, antitumor, and antithrombotic properties.[6]
Shikonin is also found in the Chinese herbal medicine plant Lithospermum erythrorhizon, the red-root gromwell, (紫草 zicao, Pinyin: zǐcǎo).
References
- ^ The Merck Index, 11th Edition, 243
- ^ Additives Archived 2011-04-06 at the Wayback Machine, Food Standards Australia New Zealand
- ^ "Current EU approved additives and their E Numbers", Food Standards Agency website, retrieved 15 Dec 2011
- ^ H. Brockmann (1936). "Die Konstitution des Alkannins, Shikonins und Alkannans". Justus Liebigs Ann. Chem. 521: 1–47. doi:10.1002/jlac.19365210102.
- ^ Shmuel Yannai (2012). Dictionary of Food Compounds. CRC Press. p. 478.
- ^ a b c Vassilios P. Papageorgiou; Andreana N. Assimopoulou; Elias A. Couladouros; David Hepworth; K. C. Nicolaou (1999). "The Chemistry and Biology of Alkannin, Shikonin, and Related Naphthazarin Natural Products". Angew. Chem. Int. Ed. 38 (3): 270–300. doi:10.1002/(SICI)1521-3773(19990201)38:3<270::AID-ANIE270>3.0.CO;2-0.
- ^ A.N. Assimopoulou; D. Boskou; V.P. Papageorgiou (2004). "Antioxidant activities of alkannin, shikonin and Alkanna tinctoria root extracts in oil substrates". Food Chemistry. 87 (3): 433–438. doi:10.1016/j.foodchem.2003.12.017.
- ^ Wiench, B; Eichhorn, T; Paulsen, M; Efferth, T (2012). "Shikonin Directly Targets Mitochondria and Causes Mitochondrial Dysfunction in Cancer Cells". Evidence-Based Complementary and Alternative Medicine. 2012: 726025. doi:10.1155/2012/726025. PMC 3478753. PMID 23118796.
{{cite journal}}: CS1 maint: unflagged free DOI (link)