m r2.7.3) (Robot: Modifying fa:۱،۳-دیهیدروکسیانتراکوینون |
JJMC89 bot III (talk | contribs) m Moving Category:3-hydroxypropenals within hydroxyquinones to Category:3-Hydroxypropenals within hydroxyquinones per Wikipedia:Categories for discussion/Speedy |
||
| (12 intermediate revisions by 10 users not shown) | |||
| Line 8: | Line 8: | ||
| ImageSize1 = 220px |
| ImageSize1 = 220px |
||
| ImageName1 = Ball-and-stick model |
| ImageName1 = Ball-and-stick model |
||
| |
| PIN = 1,3-Dihydroxyanthracene-9,10-dione |
||
| OtherNames = Purpuroxanthin; Xanthopurpurin |
| OtherNames = Purpuroxanthin; Xanthopurpurin |
||
| |
|Section1={{Chembox Identifiers |
||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
||
| ChEBI_Ref = {{ebicite|correct|EBI}} |
| ChEBI_Ref = {{ebicite|correct|EBI}} |
||
| Line 23: | Line 23: | ||
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
||
| StdInChIKey = WPWWKBNOXTZDQJ-UHFFFAOYSA-N |
| StdInChIKey = WPWWKBNOXTZDQJ-UHFFFAOYSA-N |
||
| CASNo_Ref = {{cascite| |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| CASNo = 518-83-2 |
| CASNo = 518-83-2 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| UNII = 9RBB2G1GQB |
|||
| PubChem = 196978 |
| PubChem = 196978 |
||
| SMILES = O=C2c1ccccc1C(=O)c3c2cc(O)cc3O |
| SMILES = O=C2c1ccccc1C(=O)c3c2cc(O)cc3O |
||
}} |
}} |
||
| |
|Section2={{Chembox Properties |
||
| Formula = C<sub>14</sub>H<sub>8</sub>O<sub>4</sub> |
| Formula = C<sub>14</sub>H<sub>8</sub>O<sub>4</sub> |
||
| MolarMass = 240.21 g/mol |
| MolarMass = 240.21 g/mol |
||
| Line 36: | Line 38: | ||
| BoilingPt = |
| BoilingPt = |
||
| Solubility = }} |
| Solubility = }} |
||
| |
|Section3={{Chembox Hazards |
||
| MainHazards = |
| MainHazards = |
||
| FlashPt = |
| FlashPt = |
||
| |
| AutoignitionPt = }} |
||
}} |
}} |
||
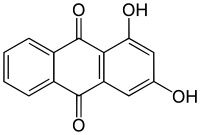
'''1,3-Dihydroxyanthraquinone''', also called '''purpuroxanthin''' or '''xanthopurpurin''' is an [[organic compound]] with formula {{chem|C|14|H|8|O|4}} that occurs in the plant ''[[Rubia cordifolia]]'' (Indian |
'''1,3-Dihydroxyanthraquinone''', also called '''purpuroxanthin''' or '''xanthopurpurin''', is an [[organic compound]] with formula {{chem|C|14|H|8|O|4}} that occurs in the plant ''[[Rubia cordifolia]]'' (Indian madder).<ref name=vankar> |
||
Padma S. Vankar, Rakhi Shanker, Debajit Mahanta and S.C. Tiwari (2008), ''Ecofriendly sonicator dyeing of cotton with ''Rubia cordifolia'' Linn. using biomordant''. Dyes and Pigments, Volume 76, Issue 1, Pages 207-212. {{doi|10.1016/j.dyepig.2006.08.023}} |
Padma S. Vankar, Rakhi Shanker, Debajit Mahanta and S.C. Tiwari (2008), ''Ecofriendly sonicator dyeing of cotton with ''Rubia cordifolia'' Linn. using biomordant''. Dyes and Pigments, Volume 76, Issue 1, Pages 207-212. {{doi|10.1016/j.dyepig.2006.08.023}} |
||
</ref> It is one of ten [[dihydroxyanthraquinone]] isomers. Its molecular structure can be viewed as being derived from [[anthraquinone]] by replacement of two [[hydrogen]] atoms (H) by [[hydroxyl]] groups (-OH). |
</ref> It is one of ten [[dihydroxyanthraquinone]] isomers. Its molecular structure can be viewed as being derived from [[anthraquinone]] by replacement of two [[hydrogen]] atoms (H) by [[hydroxyl]] groups (-OH). |
||
Xanthopurpurin occurs in small amounts (as a [[glycoside]]) in the root of the common madder plant, ''[[Rubia tinctorum]]'', together with [[alizarin]], [[Purpurin (dye)|purpurin]] and other anthraquinone derivatives.<ref name=derk> |
Xanthopurpurin occurs in small amounts (as a [[glycoside]]) in the root of the common madder plant, ''[[Rubia tinctorum]]'', together with [[alizarin]], [[Purpurin (dye)|purpurin]] and other anthraquinone derivatives.<ref name=derk> |
||
| Line 51: | Line 53: | ||
==Properties== |
==Properties== |
||
Xanthopurpurin is insoluble in [[hexane]] but soluble in [[chloroform]]. It can be obtained from solutions in the latter as reddish crystals that melt at 270–273 |
Xanthopurpurin is insoluble in [[hexane]] but soluble in [[chloroform]]. It can be obtained from solutions in the latter as reddish crystals that melt at 270–273 °C.<!-- Vis λ<sub>max</sub> (MeOH) (nm): 425; IR ν<sub>max</sub> (KBr) (cm<sup>−1</sup>): 3310 (–OH), 1660, 1625 (–C=O), 1580 (aromatic –C=C–); MS: ''m''/''z'' 240 (M<sup>+</sup>).--><ref name=vankar/> |
||
Like many [[dihydroxyanthraquinone|dihydroxy]]- and [[trihydroxyanthraquinone]]s, it has a [[purgative]] action, although only 1/6 as effective as [[1,2,7-Trihidroxyanthraquinone|1,2,7-trihidroxyanthraquinone]] (anthrapurpurin).<ref name= guigan> |
Like many [[dihydroxyanthraquinone|dihydroxy]]- and [[trihydroxyanthraquinone]]s, it has a [[purgative]] action, although only 1/6 as effective as [[1,2,7-Trihidroxyanthraquinone|1,2,7-trihidroxyanthraquinone]] (anthrapurpurin).<ref name= guigan> |
||
Hugh Alister McGuigan (1921), ''An introduction to chemical pharmacology; pharmacodynamics in relation to chemistry''. P. Blakiston's son, Philadelphia. [ |
Hugh Alister McGuigan (1921), ''An introduction to chemical pharmacology; pharmacodynamics in relation to chemistry''. P. Blakiston's son, Philadelphia. [https://archive.org/stream/introductiontoch00mcguuoft#page/132 Online version] at archive.org, accessed on 2010-01-30. |
||
</ref> |
</ref> |
||
| Line 64: | Line 66: | ||
{{DEFAULTSORT:Dihydroxyanthraquinone, 1,3-}} |
{{DEFAULTSORT:Dihydroxyanthraquinone, 1,3-}} |
||
[[Category:Anthraquinone dyes]] |
[[Category:Anthraquinone dyes]] |
||
[[Category:Dihydroxyanthraquinones]] |
[[Category:Dihydroxyanthraquinones]] |
||
[[Category:Resorcinols]] |
[[Category:Resorcinols]] |
||
[[Category:3-Hydroxypropenals within hydroxyquinones]] |
|||
[[fa:۱،۳-دیهیدروکسیانتراکوینون]] |
|||
Latest revision as of 07:20, 7 January 2022
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1,3-Dihydroxyanthracene-9,10-dione | |
| Other names
Purpuroxanthin; Xanthopurpurin
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C14H8O4 | |
| Molar mass | 240.21 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
1,3-Dihydroxyanthraquinone, also called purpuroxanthin or xanthopurpurin, is an organic compound with formula C
14H
8O
4 that occurs in the plant Rubia cordifolia (Indian madder).[1] It is one of ten dihydroxyanthraquinone isomers. Its molecular structure can be viewed as being derived from anthraquinone by replacement of two hydrogen atoms (H) by hydroxyl groups (-OH).
Xanthopurpurin occurs in small amounts (as a glycoside) in the root of the common madder plant, Rubia tinctorum, together with alizarin, purpurin and other anthraquinone derivatives.[2]
Properties[edit]
Xanthopurpurin is insoluble in hexane but soluble in chloroform. It can be obtained from solutions in the latter as reddish crystals that melt at 270–273 °C.[1]
Like many dihydroxy- and trihydroxyanthraquinones, it has a purgative action, although only 1/6 as effective as 1,2,7-trihidroxyanthraquinone (anthrapurpurin).[3]
See also[edit]
- alizarin (1,2-dihydroxyanthraquinone)
References[edit]
- ^ a b Padma S. Vankar, Rakhi Shanker, Debajit Mahanta and S.C. Tiwari (2008), Ecofriendly sonicator dyeing of cotton with Rubia cordifolia Linn. using biomordant. Dyes and Pigments, Volume 76, Issue 1, Pages 207-212. doi:10.1016/j.dyepig.2006.08.023
- ^ Goverdina C. H. Derksen, Harm A. G. Niederländer and Teris A. van Beek (2002), Analysis of anthraquinones in Rubia tinctorum L. by liquid chromatography coupled with diode-array UV and mass spectrometric detection. Journal of Chromatography A, Volume 978, Issues 1-2, Pages 119-127, doi:10.1016/S0021-9673(02)01412-7
- ^ Hugh Alister McGuigan (1921), An introduction to chemical pharmacology; pharmacodynamics in relation to chemistry. P. Blakiston's son, Philadelphia. Online version at archive.org, accessed on 2010-01-30.