Content deleted Content added
Script assisted update of identifiers from ChemSpider, CommonChemistry and FDA for the Chem/Drugbox validation project - Updated: StdInChI StdInChIKey. |
Updating {{chembox}} (no changed fields - added verified revid - updated 'UNII_Ref', 'ChemSpiderID_Ref', 'StdInChI_Ref', 'StdInChIKey_Ref') per Chem/Drugbox validation (report [[Wikipedia_talk:Wi |
||
| Line 1: | Line 1: | ||
{{chembox |
{{chembox |
||
| verifiedrevid = 399528513 |
|||
| OtherNames = |
| OtherNames = |
||
| ImageFile= BSbBr3structure.jpg |
| ImageFile= BSbBr3structure.jpg |
||
| Section1 = {{Chembox Identifiers |
| Section1 = {{Chembox Identifiers |
||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
|||
| |
| ChemSpiderID = 23017 |
||
| InChI = 1/3BrH.Sb.3H/h3*1H;;;;/q;;;+3;;;/p-3/r3BrH.H3Sb/h3*1H;1H3/q;;;+3/p-3 |
| InChI = 1/3BrH.Sb.3H/h3*1H;;;;/q;;;+3;;;/p-3/r3BrH.H3Sb/h3*1H;1H3/q;;;+3/p-3 |
||
| SMILES = [Br-].[Br-].[Br-].[SbH3+3] |
| SMILES = [Br-].[Br-].[Br-].[SbH3+3] |
||
| Line 10: | Line 12: | ||
| InChIKey1 = RPJGYLSSECYURW-DFZHHIFOAI |
| InChIKey1 = RPJGYLSSECYURW-DFZHHIFOAI |
||
| SMILES1 = Br[Sb](Br)Br |
| SMILES1 = Br[Sb](Br)Br |
||
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
|||
| StdInChI = 1S/3BrH.Sb/h3*1H;/q;;;+3/p-3 |
| StdInChI = 1S/3BrH.Sb/h3*1H;/q;;;+3/p-3 |
||
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
|||
| StdInChIKey = RPJGYLSSECYURW-UHFFFAOYSA-K |
| StdInChIKey = RPJGYLSSECYURW-UHFFFAOYSA-K |
||
| CASNo = 7789-61-9 |
| CASNo = 7789-61-9 |
||
Revision as of 15:28, 29 November 2010
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.254 |
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| SbBr3 | |
| Molar mass | 361.472 g/mol |
| Appearance | colorless hygroscopic crystals |
| Density | 4.35 g/cm3, solid |
| Melting point | 96.6°C |
| Boiling point | 280°C |
| reacts | |
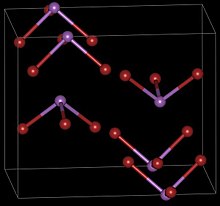
| Structure | |
| Orthorhombic, oP16, SpaceGroup = Pnma, No. 62 (β form) | |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
7000 mg/kg |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Antimony tribromide (SbBr3) is a chemical compound containing antimony in its +3 oxidation state. It may be made by the reaction of antimony with elemental bromine or the reaction of antimony trioxide with hydrobromic acid.It can be added to polymers such as polyethylene as a fire retardant.[1] It is also used in the production of other antimony compounds, in chemical analysis, and in dyeing. [2]
Antimony tribromide has two crystalline forms, both having orthorhombic symmetries. When a warm carbon disulfide solution of SbBr3 is rapidly cooled, it crystallizes into the needle-like α-SbBr3, which then slowly converts to the more stable β form.[3]
Antimony tribromide hydrolyzes in water to form hydrobromic acid and antimony trioxide:
- 2 SbBr3 + 3 H2O → Sb2O3 + 6 HBr
References
- ^ Yang, Y. P. (1991). "A study of the synergistic action of antimony oxide in fire-retardant polyethylene". Fire and Materials. 15: 37. doi:10.1002/fam.810150107.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ "Antimony tribromide" http://cameochemicals.noaa.gov/chemical/2501
- ^ Okuda, Tsutomu; Terao, Hiromitsu; Ege, Osamu; Negita, Hisao (1970). "Structural Studies of Antimony Tribromide and Its Molecular Complex with Benzene by Means of the 81Br Nuclear Quadrupole Resonance". Bulletin of the Chemical Society of Japan. 43: 2398. doi:10.1246/bcsj.43.2398.