Content deleted Content added
Replacing image (higher technical standard) |
Updating {{chembox}} (no changed fields - updated 'ChemSpiderID_Ref', 'StdInChI_Ref', 'StdInChIKey_Ref') per Chem/Drugbox validation (report errors or [[u |
||
| Line 11: | Line 11: | ||
| Section1 = {{Chembox Identifiers |
| Section1 = {{Chembox Identifiers |
||
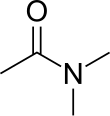
| SMILES = O=C(N(C)C)C |
| SMILES = O=C(N(C)C)C |
||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
|||
| |
| ChemSpiderID = 29107 |
||
| PubChem = 31374 |
| PubChem = 31374 |
||
| UNII_Ref = {{fdacite}} |
| UNII_Ref = {{fdacite|correct|FDA}} |
||
| UNII = JCV5VDB3HY |
| UNII = JCV5VDB3HY |
||
| InChI = 1/C4H9NO/c1-4(6)5(2)3/h1-3H3 |
| InChI = 1/C4H9NO/c1-4(6)5(2)3/h1-3H3 |
||
Revision as of 15:18, 29 November 2010
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Dimethylacetamide
| |||
| Other names
DMAc, DMA, acetic acid-dimethylamide, N,N-dimethylacetamide, acetyldimethylamine
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.004.389 | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H9NO | |||
| Molar mass | 87.122 g·mol−1 | ||
| Appearance | Colorless liquid with faint ammonia odor | ||
| Density | 0.94 g/cm3 | ||
| Melting point | −20 °C (−4 °F; 253 K) | ||
| Boiling point | 164-166 °C | ||
| Viscosity | 1.956 cP @ 25 °C 1.279 cP @ 50 °C | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Toxic (T) | ||
| NFPA 704 (fire diamond) | |||
| Flash point | 70 °C | ||
| Related compounds | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Dimethylacetamide is the organic compound with the formula CH3C(O)N(CH3)2. This colorless, water miscible, high boiling liquid is commonly used as a polar solvent in organic chemistry. DMAc is miscible with most other solvents, although it is poorly soluble in aliphatic hydrocarbons.
The chemical reactions of dimethylacetamide are typical of N,N-disubstituted amides. It will hydrolyze in the presence of acids:
- CH3CON(CH3)2 + H2O + HCl → CH3COOH + (CH3)2NH2+Cl-
Dimethylacetamide is useful as a medium for strong bases such as sodium hydroxide.[1] Dimethylacetamide is commonly used as a solvent for fibers or in the adhesive industry. It is also employed in the production of pharmaceuticals and plasticizers as a reaction medium.
References
- ^ S. Zen and E. Kaji (1988). "Dimethyl nitrosuccinate". Organic Syntheses; Collected Volumes, vol. 6, p. 503.