Script assisted update of identifiers from ChemSpider, CommonChemistry and FDA for the Chem/Drugbox validation project - Updated: StdInChI StdInChIKey. |
Updating {{chembox}} (no changed fields - added verified revid - updated 'UNII_Ref', 'ChemSpiderID_Ref', 'StdInChI_Ref', 'StdInChIKey_Ref') per Chem/Drugbox validation (report [[Wikipedia_talk:Wi |
||
| Line 1: | Line 1: | ||
{{chembox |
{{chembox |
||
| verifiedrevid = 399507923 |
|||
| Name = Ammonia borane |
| Name = Ammonia borane |
||
| ImageFile = Ammonia-borane-from-xtal-3D-balls.png |
| ImageFile = Ammonia-borane-from-xtal-3D-balls.png |
||
| Line 8: | Line 9: | ||
| Section1 = {{Chembox Identifiers |
| Section1 = {{Chembox Identifiers |
||
| SMILES = [NH3+][BH3-] |
| SMILES = [NH3+][BH3-] |
||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
|||
| |
| ChemSpiderID = 371215 |
||
| InChI = 1/BH3.H3N/h2*1H3 |
| InChI = 1/BH3.H3N/h2*1H3 |
||
| InChIKey = WZMUUWMLOCZETI-UHFFFAOYAV |
| InChIKey = WZMUUWMLOCZETI-UHFFFAOYAV |
||
| SMILES1 = B.N |
| SMILES1 = B.N |
||
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
|||
| StdInChI = 1S/BH3.H3N/h2*1H3 |
| StdInChI = 1S/BH3.H3N/h2*1H3 |
||
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
|||
| StdInChIKey = WZMUUWMLOCZETI-UHFFFAOYSA-N |
| StdInChIKey = WZMUUWMLOCZETI-UHFFFAOYSA-N |
||
| CASNo = 13774-81-7 |
| CASNo = 13774-81-7 |
||
Revision as of 12:42, 29 November 2010

| |
| Names | |
|---|---|
| IUPAC name
Ammonia borane
| |
| Other names
Borazane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.170.890 |
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| H6NB | |
| Molar mass | 30.86534 g/mol |
| Appearance | colorless, waxy solid |
| Density | 0.780 g/cm3 |
| Melting point | 104 °C |
| Boiling point | decomposes |
| good | |
| Solubility in other solvents | MeOH, Et2O |
| Structure | |
| I4mm (orthorhombic, < 200K)(tetragonal, >200K) | |
| ethane-like | |
| 5.2 D | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
flammable |
| Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ammonia borane (or borazane) is the chemical compound with the formula H3NBH3. The colourless solid is the simplest molecular boron-nitrogen-hydride compound and has attracted attention as a source of hydrogen fuel.
Synthesis
Reaction of diborane with ammonia mainly gives the diammoniate salt [H2B(NH3)2]+, (BH4)−. Ammonia borane is the main product when an adduct of borane is employed in place of diborane:[1]
- BH3(THF) + NH3 → BH3NH3 + THF
Properties and structure
The molecule adopts a structure like ethane. The B-N distance is 1.58(2) Å. The B-H and N-H distances are 1.15 and 0.96 Å, respectively. Its similarity to ethane is tenuous since borane-ammonia is a solid and ethane is a gas: their melting points differing by 284 °C. This difference is consistent with the highly polar nature of ammonia borane. The H atoms attached to boron are hydridic and those attached to nitrogen are somewhat acidic.
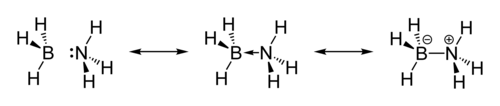
The structure of the solid indicates a close association of the NH and the BH centers.[2][3] The original crystallographic analysis of this compound reversed the assignments of B and N. The updated structure was arrived at with improved data using the technique of neutron diffraction that allowed the hydrogen atoms to be located with greater precision.
![Part of the crystal structure of ammonia borane[2]](https://upload.wikimedia.org/wikipedia/commons/thumb/9/93/Ammonia-borane-xtal-3D-balls.png/300px-Ammonia-borane-xtal-3D-balls.png)
Uses
Ammonia borane has been suggested as a storage medium for hydrogen when the gas is used to fuel motor vehicles. It can be made to release hydrogen on heating, being polymerized first to (NH2BH2)n, and then to (NHBH)n.[4] It is more hydrogen-dense than liquid hydrogen and also able to exist in normal temperatures and pressures.[5]
Borane-ammonia finds some use in organic synthesis as an air-stable derivative of diborane.[6]
References
- ^ Shore, S.G. (1964), "Large Scale Synthesis of H2B(NH3)2+BH4− and H3NBH3", Inorganic Chemistry, 3: pp. 914–15, doi:10.1021/ic50016a038.
{{citation}}:|pages=has extra text (help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b Klooster, W.T. (1999), "Study of the N-H...H-B Dihydrogen Bond Including the Crystal Structure of BH3NH3 by Neutron Diffraction", Journal of the American Chemical Society, 121: pp. 6337–6343, doi:10.1021/ja9825332
{{citation}}:|pages=has extra text (help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Boese, R (1992), Maksic, Z.B., Eckert-Masic, M. (ed.), Molecules in Natural Science and Medicine, Chichester, England: E. Horwood
{{citation}}: Unknown parameter|coauthors=ignored (|author=suggested) (help)CS1 maint: multiple names: editors list (link) - ^ "Hydrogen gets on board", Maciej Gutowski and Tom Autrey, Royal Society of Chemistry
- ^ Frances H. Stephens, Vincent Pons, R. Tom Baker "Ammonia–borane: the hydrogen source par excellence?" Dalton Transactions, 2007, pp. 2613-2626 doi:10.1039/b703053c
- ^ Andrews, G.C. (2004), "Borane–Ammonia", in Paquette, L. (ed.), Encyclopedia of Reagents for Organic Synthesis, New York: John Wiley & Sons, doi:10.1002/047084289