
| |
| Names | |
|---|---|
| Other names
Methyl L-histidinate, HME
| |
| Identifiers | |
3D model (JSmol)
|
|
| 957974 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.014.645 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H11N3O2 | |
| Molar mass | 169.184 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Histidine methyl ester (HME) is an irreversible histidine decarboxylase inhibitor.[1][2][3] It is the methyl ester of histidine.
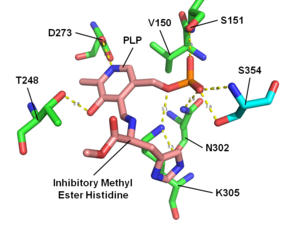
HME bound to histidine decarboxylase
See also[edit]
References[edit]
- ^ Lane, Roger S; Manning, James M; Snell, Esmond E (2002). "Histidine decarboxylase of lactobacillus 30a: inactivation and active-site labeling by L-histidine methyl ester". Biochemistry. 15 (19): 4180–5. doi:10.1021/bi00664a008. PMID 963031.
- ^ Alston, Theodore A; Abeles, Robert H (2002). "Reaction of Lactobacillus histidine decarboxylase with L-histidine methyl ester". Biochemistry. 26 (13): 4082–5. doi:10.1021/bi00387a051. PMID 3651438.
- ^ Komori, H; Nitta, Y; Ueno, H; Higuchi, Y (2012). "Structural Study Reveals That Ser-354 Determines Substrate Specificity on Human Histidine Decarboxylase". The Journal of Biological Chemistry. 287 (34): 29175–83. doi:10.1074/jbc.M112.381897. PMC 3436558. PMID 22767596.