| |
| Names | |
|---|---|
| IUPAC name
bis(tricarbonyl[η5-cyclopentadienyl]
| |
| Other names
cyclopentadienyl chromium
carbonyl dimer
Bis(tricarbonylcyclopentadienylchromium) | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C16H10Cr2O6 | |
| Molar mass | 402.242 g·mol−1 |
| Appearance | green solid |
| Density | 2.738 g/cm3 |
| insoluble | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
flammable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
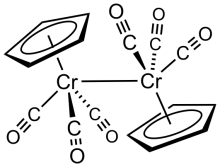
Cyclopentadienylchromium tricarbonyl dimer is the organochromium compound with the formula Cp2Cr2(CO)6, where Cp is C5H5. A dark green crystalline solid. It is the subject of research it exists in measureable equilibrium quantities with the monometallic radical CpCr(CO)3.
Structure and synthesis[edit]
The six CO ligands are terminal, and the Cr-Cr bond distance is 3.281 Å, 0.06 Å longer than the related dimolybdenum compound.[1] The compound is prepared by treatment of chromium hexacarbonyl with sodium cyclopentadienide followed by oxidation of the resulting NaCr(CO)3(C5H5).[2]
Related compounds[edit]
References[edit]
- ^ Adams, Richard D.; Collins, Douglas E.; Cotton, F. Albert (1974). "Unusual structural and magnetic resonance properties of dicyclopentadienylhexacarbonyldichromium". Journal of the American Chemical Society. 96 (5): 749–754. doi:10.1021/ja00810a019.
- ^ Manning, A. R.; Hacket, Paul; Birdwhistell, Ralph (1990). "Hexacarbonylbis(η5‐Cyclopentadienyl)Dichromium, Molybdenum, and Tungsten and their Analogs, M2(η5‐C5H4R)2(CO)6 (M = Cr, Mo, and W; R = H, Me or PhCH2)". Inorganic Syntheses. 28: 148–149. doi:10.1002/9780470132593.ch39. ISBN 9780470132593.