Content deleted Content added
Importing Wikidata short description: "Chemical compound" (Shortdesc helper) |
|||
| (5 intermediate revisions by 5 users not shown) | |||
| Line 1: | Line 1: | ||
{{Short description|Chemical compound}} |
|||
{{Drugbox |
{{Drugbox |
||
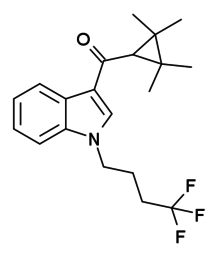
| IUPAC_name = (2,2,3,3-Tetramethylcyclopropyl)[1-(4,4,4-trifluorobutyl)-1H-indol-3-yl]methanone |
| IUPAC_name = (2,2,3,3-Tetramethylcyclopropyl)[1-(4,4,4-trifluorobutyl)-1H-indol-3-yl]methanone |
||
| Line 9: | Line 10: | ||
| pregnancy_category = |
| pregnancy_category = |
||
| legal_AU = <!-- Unscheduled / S2 / S3 / S4 / S5 / S6 / S7 / S8 / S9 --> |
| legal_AU = <!-- Unscheduled / S2 / S3 / S4 / S5 / S6 / S7 / S8 / S9 --> |
||
| legal_CA = |
| legal_CA = Schedule II |
||
| legal_UK = <!-- GSL / P / POM / CD / Class A, B, C --> |
| legal_UK = <!-- GSL / P / POM / CD / Class A, B, C --> |
||
| legal_US = |
| legal_US = |
||
| Line 23: | Line 24: | ||
<!--Identifiers--> |
<!--Identifiers--> |
||
| CAS_number_Ref = |
| CAS_number_Ref = {{cascite|correct|CAS}} |
||
| CAS_number = 895155-78-9 |
| CAS_number = 895155-78-9 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| UNII = EG79K9XQF5 |
|||
| ATC_prefix = |
| ATC_prefix = |
||
| ATC_suffix = |
| ATC_suffix = |
||
| Line 35: | Line 38: | ||
<!--Chemical data--> |
<!--Chemical data--> |
||
| C=20 | H=24 | F=3 | N=1 | O=1 |
| C=20 | H=24 | F=3 | N=1 | O=1 |
||
| molecular_weight = 351.405 g/mol |
|||
}} |
}} |
||
'''XLR-12''' is an [[indole]]-based [[synthetic cannabinoid]] drug that was invented by [[Abbott Laboratories]] in 2006.<ref name = "WO2006/069196">{{cite patent |country= WO |number= 2006069196 |status= application |title= 3-Cycloalkylcarbonyl indoles as cannabinoid receptor ligands |pubdate= 2006-06-29 |inventor= Pace JM, Tietje K, Dart MJ, Meyer MD |assign1= Abbott Laboratories }}</ref> It is an analogue of [[XLR-11 (drug)|XLR-11]] where the 5-fluoropentyl chain has been replaced with a 4,4,4-trifluorobutyl chain. XLR-12 is relatively highly selective for the CB<sub>2</sub> receptor, with a K<sub>i</sub> of 0.09 nM and 167x selectivity over the related CB<sub>1</sub> receptor, however it still retains appreciable affinity for CB<sub>1</sub> with a K<sub>i</sub> of 15 nM.<ref>{{ |
'''XLR-12''' is an [[indole]]-based [[synthetic cannabinoid]] drug that was invented by [[Abbott Laboratories]] in 2006.<ref name = "WO2006/069196">{{cite patent |country= WO |number= 2006069196 |status= application |title= 3-Cycloalkylcarbonyl indoles as cannabinoid receptor ligands |pubdate= 2006-06-29 |inventor= Pace JM, Tietje K, Dart MJ, Meyer MD |assign1= Abbott Laboratories }}</ref> It is an analogue of [[XLR-11 (drug)|XLR-11]] where the 5-fluoropentyl chain has been replaced with a 4,4,4-trifluorobutyl chain. XLR-12 is relatively highly selective for the CB<sub>2</sub> receptor, with a K<sub>i</sub> of 0.09 nM and 167x selectivity over the related CB<sub>1</sub> receptor, however it still retains appreciable affinity for CB<sub>1</sub> with a K<sub>i</sub> of 15 nM.<ref>{{cite journal | vauthors = Frost JM, Dart MJ, Tietje KR, Garrison TR, Grayson GK, Daza AV, El-Kouhen OF, Yao BB, Hsieh GC, Pai M, Zhu CZ, Chandran P, Meyer MD | display-authors = 6 | title = Indol-3-ylcycloalkyl ketones: effects of N1 substituted indole side chain variations on CB(2) cannabinoid receptor activity | journal = Journal of Medicinal Chemistry | volume = 53 | issue = 1 | pages = 295–315 | date = January 2010 | pmid = 19921781 | doi = 10.1021/jm901214q }}</ref> |
||
== Legal status == |
== Legal status == |
||
XLR-12 is illegal in Hungary<ref>[http://www.daath.hu/incoming/designer_jogi_lista_20150903_BSZKI_Daath_kieg.pdf A Magyarországon megjelent, a Kábítószer és Kábítószer-függőség Európai Megfigyelő Központjának Korai Jelzőrendszerébe (EMCDDA EWS) 2005 óta bejelentett ellenőrzött anyagok büntetőjogi vonatkozású besorolása]</ref> and Japan.<ref>{{cite web | url=http://www.mhlw.go.jp/seisakunitsuite/bunya/kenkou_iryou/iyakuhin/yakubuturanyou/dl/meisho.pdf | title=指定薬物名称・構造式一覧(平成27年9月16日現在) | publisher=厚生労働省 | date=16 September 2015 | language=Japanese | |
XLR-12 is illegal in Hungary<ref>[http://www.daath.hu/incoming/designer_jogi_lista_20150903_BSZKI_Daath_kieg.pdf A Magyarországon megjelent, a Kábítószer és Kábítószer-függőség Európai Megfigyelő Központjának Korai Jelzőrendszerébe (EMCDDA EWS) 2005 óta bejelentett ellenőrzött anyagok büntetőjogi vonatkozású besorolása]</ref> and Japan.<ref>{{cite web | url=http://www.mhlw.go.jp/seisakunitsuite/bunya/kenkou_iryou/iyakuhin/yakubuturanyou/dl/meisho.pdf | title=指定薬物名称・構造式一覧(平成27年9月16日現在) | publisher=厚生労働省 | date=16 September 2015 | language=Japanese | access-date=8 October 2015}}</ref> |
||
==See also== |
== See also == |
||
* [[UR-144]] |
* [[UR-144]] |
||
* [[FUB-144]] |
* [[FUB-144]] |
||
* [[JTE 7-31]] |
* [[JTE 7-31]] |
||
==References== |
== References == |
||
{{reflist}} |
{{reflist}} |
||
| Line 55: | Line 57: | ||
[[Category:Cannabinoids]] |
[[Category:Cannabinoids]] |
||
[[Category:Designer drugs]] |
[[Category:Designer drugs]] |
||
[[Category:Indoles]] |
|||
[[Category:Tetramethylcyclopropanoylindoles]] |
[[Category:Tetramethylcyclopropanoylindoles]] |
||
{{cannabinoid-stub}} |
{{cannabinoid-stub}} |
||
Latest revision as of 03:56, 13 April 2022
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C20H24F3NO |
| Molar mass | 351.413 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
XLR-12 is an indole-based synthetic cannabinoid drug that was invented by Abbott Laboratories in 2006.[1] It is an analogue of XLR-11 where the 5-fluoropentyl chain has been replaced with a 4,4,4-trifluorobutyl chain. XLR-12 is relatively highly selective for the CB2 receptor, with a Ki of 0.09 nM and 167x selectivity over the related CB1 receptor, however it still retains appreciable affinity for CB1 with a Ki of 15 nM.[2]
Legal status[edit]
XLR-12 is illegal in Hungary[3] and Japan.[4]
See also[edit]
References[edit]
- ^ WO application 2006069196, Pace JM, Tietje K, Dart MJ, Meyer MD, "3-Cycloalkylcarbonyl indoles as cannabinoid receptor ligands", published 2006-06-29, assigned to Abbott Laboratories
- ^ Frost JM, Dart MJ, Tietje KR, Garrison TR, Grayson GK, Daza AV, et al. (January 2010). "Indol-3-ylcycloalkyl ketones: effects of N1 substituted indole side chain variations on CB(2) cannabinoid receptor activity". Journal of Medicinal Chemistry. 53 (1): 295–315. doi:10.1021/jm901214q. PMID 19921781.
- ^ A Magyarországon megjelent, a Kábítószer és Kábítószer-függőség Európai Megfigyelő Központjának Korai Jelzőrendszerébe (EMCDDA EWS) 2005 óta bejelentett ellenőrzött anyagok büntetőjogi vonatkozású besorolása
- ^ "指定薬物名称・構造式一覧(平成27年9月16日現在)" (PDF) (in Japanese). 厚生労働省. 16 September 2015. Retrieved 8 October 2015.