m clean up, References after punctuation per WP:REFPUNC and WP:PAIC, removed stub tag using AWB (8435) |
m <ref> clean up |
||
| Line 31: | Line 31: | ||
}} |
}} |
||
'''WIN 55,212-2''' is a chemical described as an [[aminoalkylindole]] derivative, which produces effects similar to those of [[cannabinoid]]s such as [[tetrahydrocannabinol]] (THC) but has an entirely different [[chemical structure]].<ref>Compton DR, |
'''WIN 55,212-2''' is a chemical described as an [[aminoalkylindole]] derivative, which produces effects similar to those of [[cannabinoid]]s such as [[tetrahydrocannabinol]] (THC) but has an entirely different [[chemical structure]].<ref>Compton DR, ''et al.'' Aminoalkylindole Analogs: Cannabimimetic Activity of a Class of Compounds Structurally Distinct from Δ<sup>9</sup>-Tetrahydrocannabinol. ''Journal of Pharmacology and Experimental Therapeutics''. 1992; 263(3):1118-1126.</ref><ref>Ferraro L, ''et al.'' The Cannabinoid Receptor Agonist WIN 55,212-2 Regulates Glutamate Transmission in Rat Cerebral Cortex: an In Vitro and In Vivo Study. ''Cerebral Cortex''. 2001; (11):728-733.</ref><ref>{{Cite pmid|12228183}}</ref> |
||
WIN 55,212-2 is a potent cannabinoid [[receptor agonist]]<ref>C.C. Felder ''et al. Mol. Pharmacol.'' 1995 '''48''' 443</ref> that has been found to be a potent analgesic<ref>I.D. Meng ''et al. Nature'' 1998 '''395''' 381</ref> in a rat model of neuropathic pain.<ref>U. Herzberg ''et al. Neurosci. Lett.'' 1997 '''221''' 157</ref> It activates [[p42 MAP kinase|p42]] and [[p44 MAP kinase|p44]] [[MAP kinase]] via receptor-mediated signaling.<ref>M. Bouaboula ''et al. Biochem. J.'' 1995 '''312''' 637</ref> |
WIN 55,212-2 is a potent cannabinoid [[receptor agonist]]<ref>C.C. Felder ''et al. Mol. Pharmacol.'' 1995 '''48''' 443</ref> that has been found to be a potent analgesic<ref>I.D. Meng ''et al. Nature'' 1998 '''395''' 381</ref> in a rat model of neuropathic pain.<ref>U. Herzberg ''et al. Neurosci. Lett.'' 1997 '''221''' 157</ref> It activates [[p42 MAP kinase|p42]] and [[p44 MAP kinase|p44]] [[MAP kinase]] via receptor-mediated signaling.<ref>M. Bouaboula ''et al. Biochem. J.'' 1995 '''312''' 637</ref> |
||
At 5 µM WIN 55,212-2 inhibit [[ATP]] production in [[sperm]] in a [[CB1 receptor|CB<sub>1</sub> receptor]]-dependent fashion.<ref>Morgan DJ, |
At 5 µM WIN 55,212-2 inhibit [[ATP]] production in [[sperm]] in a [[CB1 receptor|CB<sub>1</sub> receptor]]-dependent fashion.<ref>Morgan DJ,''et al.'' Δ9-Tetrahydrocannabinol (Δ9-THC) attenuates mouse sperm motility and male fecundity. Br J Pharmacol. 2012. 165(8):2575-83. doi: 10.1111/j.1476-5381.2011.01506.x. {{Cite pmid|21615727}}</ref> |
||
WIN 55,212-2, along with [[HU-210]] and [[JWH-133]], may prevent the inflammation caused by [[amyloid beta]] proteins involved in [[Alzheimer's disease]], in addition to preventing cognitive impairment and loss of neuronal [[genetic markers|markers]]. This anti-inflammatory action is induced through agonist action at [[cannabinoid receptor]]s, which prevents [[microglia]]l activation that elicits the inflammation. Additionally, cannabinoids completely abolish neurotoxicity related to [[Microglia#Activated|microglial activation]] in rat models. |
WIN 55,212-2, along with [[HU-210]] and [[JWH-133]], may prevent the inflammation caused by [[amyloid beta]] proteins involved in [[Alzheimer's disease]], in addition to preventing cognitive impairment and loss of neuronal [[genetic markers|markers]]. This anti-inflammatory action is induced through agonist action at [[cannabinoid receptor]]s, which prevents [[microglia]]l activation that elicits the inflammation. Additionally, cannabinoids completely abolish neurotoxicity related to [[Microglia#Activated|microglial activation]] in rat models.{{Citation needed|date=December 2010}} |
||
WIN 55,212-2 is a full agonist at the [[CB1 receptor|CB<sub>1</sub> cannabinoid receptor]] ([[Dissociation constant|''K''<sub>i</sub>]] = 1.9 nM) and has much higher affinity than [[tetrahydrocannabinol|THC]] (''K''<sub>i</sub> = 41 nM) for this receptor.<ref>{{Cite pmid|8450470}}</ref> |
WIN 55,212-2 is a full agonist at the [[CB1 receptor|CB<sub>1</sub> cannabinoid receptor]] ([[Dissociation constant|''K''<sub>i</sub>]] = 1.9 nM) and has much higher affinity than [[tetrahydrocannabinol|THC]] (''K''<sub>i</sub> = 41 nM) for this receptor.<ref>{{Cite pmid|8450470}}</ref> |
||
WIN 55,212-2 reduces voluntary wheel running in laboratory mice, but with effects that depend on both |
WIN 55,212-2 reduces voluntary wheel running in laboratory mice, but with effects that depend on both genetic background and sex.<ref>{{cite journal |author= Keeney BK, ''et al.'' |title=Sex differences in cannabinoid receptor-1 (CB1) pharmacology in mice selectively bred for high voluntary wheel-running behavior |journal=Pharmacology, Biochemistry and Behavior |volume=101 |pages=528-537 |year=2012 |doi=10.1016/j.pbb.2012.02.017}}</ref> |
||
|author= Keeney BK, Meek TH, Middleton KM, Holness LF, [[Theodore Garland, Jr.|Garland T, Jr.]] |
|||
|title=Sex differences in cannabinoid receptor-1 (CB1) [[pharmacology]] in mice [[selective breeding|selectively bred]] for high voluntary wheel-running [[behavior]] |
|||
|journal=Pharmacology, Biochemistry and Behavior |
|||
|volume=101 |
|||
|issue= |
|||
|pages=528-537 |
|||
|year=2012 |
|||
|pmid= |
|||
|doi=10.1016/j.pbb.2012.02.017} |
|||
}</ref> |
|||
[[File:Pancreatic stellate cell cropped.png|thumb|left|[[Pancreatic stellate cell]]s. The cells in the lower frame are under the action of WIN 55,212-2. They are thought to assume a more "[[G0 phase|quiescent]]" phenotype. From Michalski et al., 2008.<ref name="PSCWIN">{{Cite pmid|18301776}}</ref>]] |
[[File:Pancreatic stellate cell cropped.png|thumb|left|[[Pancreatic stellate cell]]s. The cells in the lower frame are under the action of WIN 55,212-2. They are thought to assume a more "[[G0 phase|quiescent]]" phenotype. From Michalski et al., 2008.<ref name="PSCWIN">{{Cite pmid|18301776}}</ref>]] |
||
Revision as of 23:17, 13 October 2012
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
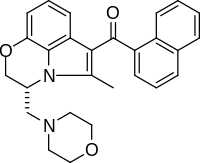
| Formula | C27H26N2O3 |
| Molar mass | 426.52 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| | |
WIN 55,212-2 is a chemical described as an aminoalkylindole derivative, which produces effects similar to those of cannabinoids such as tetrahydrocannabinol (THC) but has an entirely different chemical structure.[1][2][3]
WIN 55,212-2 is a potent cannabinoid receptor agonist[4] that has been found to be a potent analgesic[5] in a rat model of neuropathic pain.[6] It activates p42 and p44 MAP kinase via receptor-mediated signaling.[7]
At 5 µM WIN 55,212-2 inhibit ATP production in sperm in a CB1 receptor-dependent fashion.[8]
WIN 55,212-2, along with HU-210 and JWH-133, may prevent the inflammation caused by amyloid beta proteins involved in Alzheimer's disease, in addition to preventing cognitive impairment and loss of neuronal markers. This anti-inflammatory action is induced through agonist action at cannabinoid receptors, which prevents microglial activation that elicits the inflammation. Additionally, cannabinoids completely abolish neurotoxicity related to microglial activation in rat models.[citation needed]
WIN 55,212-2 is a full agonist at the CB1 cannabinoid receptor (Ki = 1.9 nM) and has much higher affinity than THC (Ki = 41 nM) for this receptor.[9]
WIN 55,212-2 reduces voluntary wheel running in laboratory mice, but with effects that depend on both genetic background and sex.[10]

External links
- Biomol Win 55,212-2 Data Sheet
- The cannabinoid WIN 55,212-2 inhibits transient receptor potential vanilloid 1 (TRPV1) and evokes peripheral antihyperalgesia via calcineurin. 2006 Jul 18; PMID 16849427
- JNeurosci.org Prevention of Alzheimer's Disease Pathology by Cannabinoids: Neuroprotection Mediated by Blockade of Microglial Activation
- New Scientist: Hope for cannabis-based drug for Alzheimer's
See also
References
- ^ Compton DR, et al. Aminoalkylindole Analogs: Cannabimimetic Activity of a Class of Compounds Structurally Distinct from Δ9-Tetrahydrocannabinol. Journal of Pharmacology and Experimental Therapeutics. 1992; 263(3):1118-1126.
- ^ Ferraro L, et al. The Cannabinoid Receptor Agonist WIN 55,212-2 Regulates Glutamate Transmission in Rat Cerebral Cortex: an In Vitro and In Vivo Study. Cerebral Cortex. 2001; (11):728-733.
- ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 12228183, please use {{cite journal}} with
|pmid=12228183instead. - ^ C.C. Felder et al. Mol. Pharmacol. 1995 48 443
- ^ I.D. Meng et al. Nature 1998 395 381
- ^ U. Herzberg et al. Neurosci. Lett. 1997 221 157
- ^ M. Bouaboula et al. Biochem. J. 1995 312 637
- ^ Morgan DJ,et al. Δ9-Tetrahydrocannabinol (Δ9-THC) attenuates mouse sperm motility and male fecundity. Br J Pharmacol. 2012. 165(8):2575-83. doi: 10.1111/j.1476-5381.2011.01506.x. Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 21615727, please use {{cite journal}} with
|pmid=21615727instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 8450470, please use {{cite journal}} with
|pmid=8450470instead. - ^ Keeney BK; et al. (2012). "Sex differences in cannabinoid receptor-1 (CB1) pharmacology in mice selectively bred for high voluntary wheel-running behavior". Pharmacology, Biochemistry and Behavior. 101: 528–537. doi:10.1016/j.pbb.2012.02.017.
{{cite journal}}: Explicit use of et al. in:|author=(help) - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 18301776, please use {{cite journal}} with
|pmid=18301776instead.