Tova Hella (talk | contribs) →Chronic use: Non-randomized studies cannot show causation. |
Tova Hella (talk | contribs) →Harm assessment: Update on ACMD 2009 report on MDMA |
||
| Line 289: | Line 289: | ||
[[David Nutt]], chairman of the U.K. [[Advisory Council on the Misuse of Drugs]], stated in the ''Journal of Psychopharmacology'' in January 2009 that ecstasy use compared favorably with [[equestrianism|horse riding]] in terms of risk, leading to around 50 deaths a year compared to about 100 from horse riding. The ACMD subsequently made clear that Professor Nutt's comments were not made on behalf of the ACMD.<ref>[http://news.bbc.co.uk/1/hi/uk/7876425.stm Ecstasy 'not worse than riding'], BBC News, 7 February 2009</ref> |
[[David Nutt]], chairman of the U.K. [[Advisory Council on the Misuse of Drugs]], stated in the ''Journal of Psychopharmacology'' in January 2009 that ecstasy use compared favorably with [[equestrianism|horse riding]] in terms of risk, leading to around 50 deaths a year compared to about 100 from horse riding. The ACMD subsequently made clear that Professor Nutt's comments were not made on behalf of the ACMD.<ref>[http://news.bbc.co.uk/1/hi/uk/7876425.stm Ecstasy 'not worse than riding'], BBC News, 7 February 2009</ref> |
||
In February 2009, the the UK Advisory Council on the Misuse of Drugs issued "A review of MDMA ('ecstasy'), its harms and classification under the Misuse of Drugs Act 1971", which recommended that MDMA be downgraded in the UK from a class A drug to a class B drug.<ref>[http://drugs.homeoffice.gov.uk/publication-search/acmd/mdma-report A review of MDMA ('ecstasy'), its harms and classification under the Misuse of Drugs Act 1971], Advisory Council on the Misuse of Drugs (ACMD), 11 February 2009</ref> The UK Home Office rejected this advice from it's independent scientific advisory board, "saying it is not prepared to send a message to young people that it takes ecstasy less seriously".<ref>[http://www.bmj.com/cgi/content/extract/338/feb13_1/b592 UK government rejects advice from drugs adviser to downgrade ecstasy], BMJ, 13 February 2009</ref> |
|||
An investigation is also currently being run to reclassify MDMA as a class B drug in the U.K. It is currently a class A drug, the same class as [[heroin]].[http://news.sky.com/skynews/Home/Politics/Ecstasy-Risks-To-Be-Re-Examined-Government-Drug-Advisory-Group-Study-Could-Lead-To-Downgrading/Article/200809415107499?f=rss] |
|||
== See also == |
== See also == |
||
Revision as of 17:22, 31 August 2009
 | |
 | |
| Clinical data | |
|---|---|
| Other names | 1,3-benzodioxolyl-N-methyl-2-propanamine; 3,4-methylenedioxy-N-methyl-α-methyl-2-phenethylamine; 3,4-methylenedioxy-N-methylamphetamine; methylenedioxymethamphetamine |
| Pregnancy category |
|
| Routes of administration | Oral, sublingual, insufflation, inhalation (smoking), injection[2] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic, CYP450 extensively involved, especially CYP2D6 |
| Elimination half-life | 6–10 (though duration of effects is typically actually 3-5 hours) |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
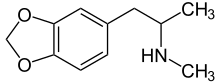
| Formula | C11H15NO2 |
| Molar mass | 193.25 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
MDMA (3,4-Methylenedioxymethamphetamine), commonly known as "Ecstasy", is a psychoactive drug and member of the amphetamine chemical class. It produces entactogenic, psychedelic, and stimulant effects.
MDMA is considered unusual for its tendency to induce a sense of intimacy with others and diminished feelings of fear and anxiety. These effects have led some to suggest it might have therapeutic benefits in certain individuals. Before it was made a controlled substance, MDMA was used as an augmentation to psychotherapy, often couples therapy, and to help treat clinical depression as well as anxiety disorders, the results of which are poorly documented. Studies have also recently been initiated to examine the therapeutic potential of MDMA for post-traumatic stress disorder (PTSD) and anxiety associated with terminal cancer.
MDMA is criminalized in most countries in the world under a United Nations (U.N.) agreement,[3] and its possession, manufacture, or sale may result in criminal prosecution, although some limited exceptions exist for scientific and medical research. MDMA is one of the most widely used recreational illicit drugs in the world[4] and is taken in a variety of contexts far removed from its roots in psychotherapeutic settings. It is commonly associated with the rave or electronica subculture and related genres of music.
There have been debates within science, health care, and drug policy circles about the risks of MDMA, specifically the possibility of neurotoxic damage to the central nervous system (CNS). Regulatory authorities in several locations around the world have approved scientific studies administering MDMA to humans to examine either its therapeutic potential, or, more commonly, simply its basic effects.[5]
Terminology
MDMA was first introduced clinically under the names "Adam" and "Empathy" when it was used in psychotherapy in the late 1970s and early to mid 1980s.[6] Later on, upon making it into the recreational drug use scene, it came to be widely known as "Ecstasy", often abbreviated as "E", "X", or "XTC".[6] MDMA in its pure, powder, crystalline, or capsule form, is commonly referred to as "Molly".[6] Various other nicknames have been used to describe MDMA as well, including "Clarity", "Essence", "Euphoria", "Magic", "Transcendence", and "Hug Drug", as well as "Love Drug", "Lover's Speed", or merely "Love", among many more.[6] A widespread term used in the United States to describe coming under the influence of MDMA is to "Roll" or to be "Rolling",[6] in reference to the involuntary eye movements experienced by those under the influence. Another term used to describe someone under its influence who is acting strange or out-of-hand under the influence of MDMA is to be an "E-Tard".[6]
History
MDMA was first synthesized in 1912 by Merck chemist Anton Köllisch. At the time, Merck was interested in developing substances that stopped abnormal bleeding. Merck wanted to evade an existing patent, held by Bayer, for one such compound - hydrastinine . At the behest of his superiors Walther Beckh and Otto Wolfes, Köllisch developed a preparation of a hydrastinine analogue, methylhydrastinine. MDMA was an intermediate compound in the synthesis of methylhydrastinine, and Merck was not interested in its properties at the time.[7] On December 24, 1912 Merck filed two patent applications that described the synthesis of MDMA[8] and its subsequent conversion to methylhydrastinine.[9]
Over the following 65 years, MDMA was largely forgotten. Merck records indicate that its researchers returned to the compound sporadically. In 1927, Max Oberlin studied the pharmacology of MDMA and observed that its effects on blood sugar and smooth muscles were similar to ephedrine's, but that, in contrast, MDMA did not appear to produce pupil dilation. Researchers at Merck conducted experiments with MDMA in 1952 and 1959.[7] In 1953 and 1954, the United States Army commissioned a study of toxicity and behavioral effects in animals of injected mescaline and several analogues, including MDMA. These originally classified investigations were declassified and published in 1973.[10] The first scientific paper on MDMA appeared in 1958 in Yakugaku Zasshi, the Journal of the Pharmaceutical Society of Japan. In this paper, Yutaka Kasuya described the synthesis of MDMA, a part of his research on antispasmodics.[11]
MDMA first appeared as a street drug in the early 1970s after its counterculture analogue, MDA, became criminalized in the United States in 1970.[12] In the mid-1970s, Alexander Shulgin, then at University of California, heard from his students about unusual effects of MDMA; among others, the drug had helped one of them to overcome his stutter. Intrigued, Shulgin synthesized MDMA and tried it himself in 1976.[13] Two years later, he and David Nichols published the first report on the drug's psychotropic effect in humans. They described "altered state of consciousness with emotional and sensual overtones" that can be compared "to marijuana, to psilocybin devoid of the hallucinatory component".[14]
Shulgin took to occasionally using MDMA for relaxation, referring to it as "my low-calorie martini", and giving the drug to his friends, researchers, and other people whom he thought could benefit from it. One such person was psychotherapist Leo Zeff, who had been known to use psychedelics in his practice. Zeff was so impressed with the action of MDMA that he came out of his semi-retirement to proselytize for it. Over the following years, Zeff traveled around the U.S. and occasionally to Europe training other psychotherapists in the use of MDMA.[13][15][16] Among underground psychotherapists, MDMA developed a reputation for enhancing communication during clinical sessions, reducing patients' psychological defenses, and increasing capacity for therapeutic introspection.[citation needed]
Due to the wording of the United Kingdom's existing Misuse of Drugs Act of 1971, MDMA was automatically classified in the U.K. as a Class A drug in 1977.
In the early 1980s in the U.S., MDMA rose to prominence as "Adam" in trendy nightclubs and gay dance clubs in the Dallas area.[17] From there, use spread to raves in major cities around the country, and then to mainstream society. The drug was first proposed for scheduling by the Drug Enforcement Administration (DEA) in July 1984[18] and was classified as a Schedule I controlled substance in the U.S. on May 31, 1985.[19]
In the late 1980s MDMA, as "ecstasy", began to be widely used in the U.K. and other parts of Europe, becoming an integral element of rave culture and other psychedelic- and dance-floor-influenced music scenes, such as Madchester and Acid House. Spreading along with rave culture, illicit MDMA use became increasingly widespread among young adults in universities and later in high schools. MDMA became one of the four most widely used illicit drugs in the U.S., along with cocaine, heroin, and marijuana.[citation needed] According to some estimates as of 2004, only marijuana attracts more first time users in the U.S..[20]
After MDMA was criminalized, most medical use stopped, although some therapists continued to prescribe the drug illegally. Later Charles Grob initiated an ascending-dose safety study in healthy volunteers. Subsequent legally-approved MDMA studies in humans have taken place in the U.S. in Detroit (Wayne State University), Chicago (University of Chicago), San Francisco (UCSF and California Pacific Medical Center), Baltimore (NIDA-NIH Intramural Program), and South Carolina, as well as in Switzerland (University Hospital of Psychiatry, Zürich), the Netherlands (Maastricht University), and Spain (Universitat Autònoma de Barcelona).[21]
Therapeutic use
There have long been suggestions that MDMA might be useful in psychotherapy, facilitating self-examination with reduced fear.[22][23][24] Indeed, some therapists, including Leo Zeff, Claudio Naranjo, George Greer, Joseph Downing, and Philip Wolfson, used MDMA in their practices until it was made illegal. George Greer synthesized MDMA in the lab of Alexander Shulgin and administered it to about 80 of his clients over the course of the remaining years preceding MDMA's Schedule I placement in 1985. In a published summary of the effects,[25] the authors reported patients felt improved in various, mild psychiatric disorders and other personal benefits, especially improved intimate communication with their significant others. In a subsequent publication on the treatment method, the authors reported that one patient with severe pain from terminal cancer experienced lasting pain relief and improved quality of life.[26] However, few of the results in this early MDMA psychotherapy were measured using methods considered reliable or convincing in scientific practice. For example, the questionnaires used might not have been sensitive to negative changes and it is not known to what extent similar patients might improve from chance or from psychotherapy.
The therapeutic potential of MDMA is currently being tested in several ongoing studies, some sponsored by the Multidisciplinary Association for Psychedelic Studies (MAPS). Studies in the U.S., Switzerland, and Israel are evaluating the efficacy of MDMA-assisted psychotherapy for treating those diagnosed with post-traumatic stress disorder (PTSD) or anxiety related to cancer. In interviews, patients and researchers from the South Carolina PTSD study report tendencies for some participants to have reduced disease severity after MDMA psychotherapy.[27][28] MAPS reported statistically significant results,[29] but the full paper is not yet published (nor confirmed in studies by other scientists).
Recreational use

The European Monitoring Centre for Drugs and Drug Addiction notes that although there are some reports of tablets being sold for as little as €1, most countries in Europe now report typical retail prices in the range of €3 to €9 per tablet.[30] The United Nations Office on Drugs and Crime claimed in its 2008 World Drug Report that typical US retail prices are higher 10 to 20 dollars per tablet, or can be from 2 to 10 dollars per tablet if bought in stacks of 10 or more.[31]
Legal alternatives
A substantial number of "ecstasy" tablets sold for recreational use (in the UK especially) are actually piperazine (mCPP or BZP) pills.[citation needed] These substances crudely mimic some of the positive effects of ecstasy, yet feature far greater negative side-effects (insomnia, migraines, nausea etc). mCPP is known to induce headaches in humans,[32][33] and has been used for testing potential anti-migraine medications.[34] These "legal highs" represent a considerable profit for the dealer; they are cheaply acquired online (with custom logos to mimic known "good" pills) and, given mCPP's legal status, there is limited risk of prosecution if caught. BZP possession does entail a greater risk.
Polysubstance use
MDMA is occasionally known for being taken in conjunction with psychedelic drugs, such as LSD or psilocybin mushrooms. As this practice has become more prevalent, most of the more common combinations have been given nicknames, such as "candy flipping", for MDMA combined with LSD,[35] and "hippie flipping" when combined with psilocybin mushrooms. Such combinations have the ability to produce an extremely powerful experience and may carry an increased risk of neurotoxicity, complications, and/or injury when compared to any individual substance. Many users use mentholated products while taking MDMA for its cooling sensation while experiencing the drug's effects. Examples include menthol cigarettes, Vicks[36] and lozenges. This sometimes has deleterious results on the upper respiratory tract.[37]
Chemistry

Safrole, a colorless or slightly yellow oil, extracted from the root-bark or the fruit of sassafras plants is the primary precursor for all manufacture of MDMA. There are numerous synthetic methods available in the literature to convert safrole into MDMA via different intermediates.[38][39][40][41] One common route is via the MDP2P (3,4-methylenedioxyphenyl-2-propanone, also known as piperonyl acetone) intermediate. This intermediate can be produced in at least two different ways. One method is to isomerize safrole to isosafrole in the presence of a strong base and then oxidize isosafrole to MDP2P. Another, reportedly better[citation needed], method is to make use of the Wacker process to oxidize safrole directly to the MDP2P (3,4-methylenedioxy phenyl-2-propanone) intermediate. This can be done with a palladium catalyst. Once the MDP2P intermediate has been produced, it is then consumed via a reductive amination to form MDMA as the product.
Relatively small quantities of essential oil are required to make large numbers of MDMA pills. The essential oil of Ocotea cymbarum typically contains between 80 and 94% safrole. This would allow 500 ml of the oil, which retails at between $20 and $100, to be used to produce an estimated 1,300 to 2,800 tablets containing approximately 120 mg of MDMA each.[42]
Pharmacology
The mechanism of action of MDMA's unusual effects has yet to be fully understood, although it is generally thought that the primary relevant pharmacological characteristic of the drug is its affinity for the plasmalemmal membrane transport protein the serotonin transporter (SERT). The SERT is a part of serotonergic neurons and removes the neurotransmitter serotonin (5-hydroxytryptamine (5-HT)) from the synapse via reuptake for termination and recycling purposes after action potentials. Not only does MDMA inhibit the reuptake of serotonin through its action at the SERT, but it also reverses its action through a process known as phosphorylation.[43] This results in the transporter conversely releasing serotonin into the synapse from inside the cell instead.
For the above reasons, MDMA is classified as a combination serotonin reuptake inhibitor (SRI) and serotonin releasing agent (SRA), though it is typically only referred to as the latter definition essentially includes SRI properties. In addition to its actions on serotonin, MDMA causes the same effects on the norepinephrine transporter (NET) and dopamine transporter (DAT), thus inducing release of norepinephrine and dopamine as well, respectively,[44] and hence, it can also be called a norepinephrine-dopamine reuptake inhibitor (NDRI) and norepinephrine-dopamine releasing agent (NDRA), or, in full, as a serotonin-norepinephrine-dopamine releasing agent (SNDRA).
MDMA's unusual entactogenic effects have been hypothesized to be, at least partly, the result of indirect oxytocin release via serotonergic neuromodulation.[45] Oxytocin is a hormone released following events like hugging, orgasm, and childbirth, and is thought to facilitate bonding and the establishment of trust. Based on studies in rats, MDMA is believed to cause the release of oxytocin, at least in part, by both directly and indirectly agonizing the serotonin 5-HT1A receptor. A placebo-controlled study in 15 human volunteers found that 100 mg MDMA increased blood levels of oxytocin and the amount of oxytocin increase was correlated with the subjective prosocial effects of MDMA.[46]
Pharmacokinetics
MDMA reaches maximal concentrations in the blood stream between 1.5 and 3 hours after ingestion. It is then slowly metabolized and excreted, with levels decreasing to half their peak concentration over approximately 8 hours. Thus, there are still high MDMA levels in the body when the experiential effects have mostly ended, indicating that acute tolerance has developed to the actions of MDMA. Taking additional supplements of MDMA at this point therefore produces higher concentrations of MDMA in the blood and brain than might be expected based on the perceived effects.
Metabolites of MDMA that have been identified in humans include 3,4-methylenedioxyamphetamine (MDA), 4-hydroxy-3-methoxy-methamphetamine (HMMA), 4-hydroxy-3-methoxyamphetamine (HMA), 3,4-dihydroxyamphetamine (DHA) (also called alpha-methyldopamine (α-Me-DA)), 3,4-methylenedioxyphenylacetone (MDP2P), and N-hydroxy-3,4-methylenedioxyamphetamine (MDOH). The contributions of these metabolites to the psychoactive and toxic effects of MDMA are an area of active research. 65% of MDMA is excreted unchanged in the urine (additionally 7% is metabolized into MDA) during the 24 hours after ingestion.[47]
MDMA is known to be metabolized by two main metabolic pathways: (1) O-demethylenation followed by catechol-O-methyltransferase (COMT)-catalyzed methylation and/or glucuronide/sulfate conjugation; and (2) N-dealkylation, deamination, and oxidation to the corresponding benzoic acid derivatives conjugated with glycine. The metabolism may be primarily by cytochrome P450 (CYP450) enzymes (CYP2D6 (in humans, but CYP2D1 in mice), and CYP3A4) and COMT. Complex, nonlinear pharmacokinetics arise via autoinhibition of CYP2D6 and CYP2D8, resulting in zeroth order kinetics at higher doses. It is thought that this can result in sustained and higher concentrations of MDMA if the user takes consecutive doses of the drug.
Because the enzyme CYP2D6 is deficient or totally absent in some people[48], it was once hypothesized that these people might have elevated risk when taking MDMA. However, there is still no evidence for this theory and available evidence argues against it.[49] It is now thought that the contribution of CYP2D6 to MDMA metabolism in humans is less than 30% of the metabolism. Indeed, an individual lacking CYP2D6 was given MDMA in a controlled clinical setting and a larger study gave MDMA to healthy volunteers after inhibiting CYP2D6 with paroxetine. Lack of the enzyme caused a modest increase in drug exposure and decreases in some metabolites, but physical effects did not appear appreciably elevated. While there is little or no evidence that low CYP2D6 activity increases risks from MDMA, it is likely that MDMA-induced CYP2D inhibition will increase risk of those prescription drugs that are metabolized by this enzyme. MDMA-induced CYP2D inhibition appears to last for up to a week after MDMA exposure.
MDMA and metabolites are primarily excreted as conjugates, such as sulfates and glucuronides.[50]
MDMA is a chiral compound and has been almost exclusively administered as a racemate. However, an early uncontrolled report suggests that the S-enantiomer is significantly more potent in humans than the R-enantiomer[51][52] indicate that the disposition of MDMA is stereoselective, with the S-enantiomer having a shorter elimination half-life and greater excretion than the R-enantiomer. For example, Fallon et al.[51] reported that the area under the blood plasma concentration versus time curve (AUC) was two to four times higher for the R-enantiomer than the S-enantiomer after a 40 mg oral dose in human volunteers. Similarly, the plasma half-life of (R)-MDMA was significantly longer than that of the S-enantiomer (5.8 ± 2.2 hours vs 3.6 ± 0.9 hours). However, because MDMA has dose dependent kinetics, it is likely that these half-lives would be higher at more typical doses (100 mg is sometimes considered a typical dose). Given as the racemate, MDMA has a half-life of around 8 hours.
Drug interactions
There are a number of reported potentially dangerous possible interactions between MDMA and other drugs. Several cases have been reported of death in individuals who ingested MDMA while taking ritonavir (Norvir), which inhibits multiple CYP450 enzymes. Toxicity or death has also been reported in people who took MDMA in combination with certain monoamine oxidase inhibitors (MAOIs) such as phenelzine (Nardil), tranylcypromine (Parnate), or moclobemide (Aurorix, Manerix).[53] On the other hand, MAOB inhibitors like selegiline (Deprenyl; Eldepryl, Zelapar, Emsam) do not seem to carry these risks when taken at selective doses, and have been used to completely block neurotoxicity in rats.[54]
People taking any type of serotonin reuptake inhibitor (SRI) on a chronic basis commonly find that the psychedelic and entactogenic effects of MDMA are near fully abolished, leaving merely stimulation instead. Many people attempt taking a dose around 3x as high and this appears to work as a remedy for the problem to some degree. Additionally, upon discontinuation of the prolonged administration of the SRI in question, such individuals may still not be able to properly experience the full desired effects of MDMA at normal doses for anywhere from a few weeks to as long as several months.
Most people who die while under the influence of MDMA have also consumed significant quantities of at least one other drug in combination. The risk of solely MDMA-induced death is overall relatively minimal.[55]
Effects
The primary effects attributable to MDMA consumption are predictable and fairly consistent amongst users. Generally, users report feeling effects within 30–60 minutes of consumption, hitting a peak at approximately 1-1.5 hours, reaching a plateau that lasts about 2–3 hours, followed by a comedown of a few hours which may be accompanied by fatigue and minor effects.[56][57][58]
Beneficial effects
The most common beneficial effects reported by users include:[59]
- A general and subjective alteration in consciousness
- A strong sense of inner peace and self-acceptance
- Diminished aggression, hostility, and jealousy
- Diminished fear, anxiety, and insecurity
- Extreme mood lift with accompanying euphoria
- Feelings of empathy, compassion, and forgiveness towards others
- Feelings of intimacy and even love for others
- Feelings of insightfulness, introspection, and mental clarity
- Improved self-confidence without the incidence of arrogance
- The ability to discuss normally anxiety-provoking topics with marked ease
- An intensification of perception, particularly tactile sensation or touch, as well as hearing and vision
- Substantial enhancement of the appreciation for or quality of music
- Mild psychedelia, consisting of mental imagery and auditory and visual distortions
As well as:
- Stimulation, arousal, and hyperactivity (e.g., many users get an "uncontrollable urge to dance" while under the influence)
- Increased energy and endurance
- Increased alertness, awareness, and wakefulness
- Increased desire, drive, and motivation
- Hypersexuality and aphrodisiac effects (along with paradoxical sexual dysfunction (see below))
- Analgesia or decreased pain sensitivity
Side effects
The most common adverse side effects reported by users include:
- Psychological
- Paradoxical anxiety and/or paranoia (though not social anxiety by any means)
- Agitation or restlessness
- Impaired attention, focus, and concentration
- Mild cognitive and memory impairment (primarily short-term memory loss)
- Physiological
- Dizziness, lightheadedness, or vertigo
- Blurry vision and/or nystagmus or involuntary and rapid eye movements
- Mydriasis or pupil dilation
- Xerostomia or dry mouth
- Nausea and emesis or vomiting
- Gastrointestinal disturbances such as diarrhea or constipation
- Headache or migraine
- Trismus or jaw clenching and/or bruxism or teeth grinding
- Trembling or muscle tremors
- Urinary retention or ischuria, caused by hyponatremia
- Sexual dysfunction, consisting of erectile dysfunction and anorgasmia or inability to ejaculate
- Anorexia or decreased appetite
- Insomnia or inability to fall to asleep
- Tachycardia or increased heart rate
- Hypertension or increased blood pressure
- Hyperthermia or increased body temperature
- Hyperhidrosis or increased perspiration or sweating
- Hyponatremia or a water-electrolyte imbalance
Rebound / withdrawal
The most common withdrawal or rebound side effects reported by users once the drug has worn off include:
- Psychological
- Anxiety and/or depression, as well as potentially even suicidal ideation
- Irritability and aggression, or even anger and rage
- Malaise or lassitude and/or fatigue or lethargy
- Agitation or restlessness
- Impaired attention, focus, and concentration, as well as drive and motivation
- Cognitive and memory impairment (especially short-term memory loss)
- Residual feelings of empathy, emotional sensitivity, and a sense of closeness to others (afterglow)
- Physiological
- Dizziness, lightheadedness, or vertigo
- Gastrointestinal disturbances such as diarrhea or constipation
- Headache, Migraine,
- Insomnia
- Panic Attacks
- Aches and pains, especially muscle and/or jaw soreness from excess physical activity (e.g., dancing) and trismus or bruxism, respectively
These effects are typically reported to last 3 to 7 days.
Overdose
Upon overdose, the potentially serious serotonin syndrome, stimulant psychosis, and/or hypertensive crisis, among other dangerous adverse reactions, may come to prominence, the symptoms of which can include the following:
- Psychological
- Disorientation and/or confusion
- Anxiety, paranoia, and/or panic attacks
- Hypervigilance or increased sensitivity to perceptual stimuli, accompanied by significantly increased threat detection
- Hypomania or full-blown mania
- Derealization and/or depersonalization
- Hallucinations and/or delusions
- Thought disorder or disorganized thinking
- Cognitive and memory impairment potentially to the point of retrograde or anterograde amnesia
- Acute delirium and/or insanity
- Physiological
- Myoclonus or involuntary and intense muscle twitching
- Hyperreflexia or overresponsive or overreactive reflexes
- Tachypnoea or rapid breathing and/or dyspnea or shortness of breath
- Palpitations or abnormal awareness of the beating of the heart
- Angina pectoris or severe chest pain, as well as pulmonary hypertension (PH)
- Cardiac arrhythmia or abnormal electical activity of the heart
- Circulatory shock or cardiogenic shock
- Vasculitis or destruction of blood vessels
- Cardiotoxicity or damage to the heart
- Cardiac arrest, myocardial infarction or heart attack, and/or heart failure
- Hemorrhage and/or stroke
- Severe hyperthermia, potentially resulting in organ failure
- Miscellaneous
- Syncope or fainting or loss of consciousness
- Seizures or convulsions
- Organ failure (as mentioned above)
- Severe neurotoxicity or brain damage
- Coma and/or death
Potential incarceration, hospitalization, institutionalization, and/or death, on account of extreme erratic behavior which may include acts of crime, accidental or intentional self-injury, and/or suicide, as well as illicit drug abuse, may ensue under such circumstances.
Chronic use
There is currently strong evidence of MDMA neurotoxicity in studies done on rodents and non-human primates. MDMA is believed to be specifically neurotoxic to the serotonergic terminals in humans. [60] Some studies indicate that repeated recreational users of MDMA have increased rates of depression and anxiety, even after quitting the drug.[61][62] In addition to this, some studies have indicated that repeated recreational users of MDMA may have impaired long-term memory[63] and cognitive function.[64] Many factors, including total lifetime MDMA consumption, the duration of abstinence between uses, the environment of use, poly-drug use/abuse, quality of mental health, various lifestyle choices, and predispositions to develop clinical depression and other disorders may contribute to various possible health consequences. MDMA use has been occasionally associated with liver damage,[65] excessive wear of teeth,[66] and (very rarely) Hallucinogen persisting perception disorder.[67]
Legal issues
MDMA is legally controlled in most of the world under the UN Convention on Psychotropic Substances and other international agreements, although exceptions exist for research. Generally, the unlicensed use, sale or manufacture of MDMA are all criminal offenses.
In the U.S., MDMA was legal and unregulated until May 31, 1985, at which time it was emergency scheduled to DEA Schedule I, for drugs deemed to have no medical uses and a high potential for abuse. During DEA hearings to schedule MDMA, most experts recommended DEA Schedule III prescription status for the drug, due to beneficial usage of MDMA in psychotherapy. The judge overseeing the hearings, Francis Young, also recommended that MDMA be placed in Schedule III. Nevertheless, the DEA classified MDMA as Schedule I.[68] In 2001, responding to a mandate from the U.S. Congress, the U.S. Sentencing Commission, resulted in an increase in the penalties for MDMA by nearly 3,000%,[69] despite scientific protest calling for a decrease in the penalties for MDMA possession and distribution.[70] The increase makes 1 gram of MDMA (four pills at 250 mg per pill's total weight regardless of purity, standard for Federal charges) equivalent to 1 gram of heroin (approximately fifty doses) or 2.2 pounds (1 kg) of marijuana for sentencing purposes at the federal level.[71]
In 1985 the World Health Organization's Expert Committee on Drug Dependence recommended that MDMA be placed in Schedule I of the Convention on Psychotropic Substances. The Convention has a provision in Article 7(a) that allows use of Schedule I drugs for "scientific and very limited medical purposes." The committee's report stated:[72]
- The Expert Committee held extensive discussions concerning therapeutic usefulness of 3,4 Methylenedioxymethamphetamine. While the Expert Committee found the reports intriguing, it felt that the studies lacked the appropriate methodological design necessary to ascertain the reliability of the observations. There was, however, sufficient interest expressed to recommend that investigations be encouraged to follow up these preliminary findings. To that end, the Expert Committee urged countries to use the provisions of article 7 of the Convention on Psychotropic Substances to facilitate research on this interesting substance.
In the U.K., MDMA is a Class A drug under the Misuse of Drugs Act 1971, making it illegal to sell, buy, or possess without a license. Penalties include a maximum of seven years and/or unlimited fine for possession; life and/or unlimited fine for production or trafficking. See list of drugs illegal in the U.K. for more information.
Health concerns
While the short-term adverse effects and contraindications of MDMA are fairly well known, there is significant debate within the scientific and medical communities regarding possible long-term physical and psychological effects of MDMA. Short-term physical health risks of MDMA consumption include hyperthermia,[73][74] and hyponatremia.[75] Continuous activity without sufficient rest or rehydration may cause body temperature to rise to dangerous levels, and loss of fluid via excessive perspiration puts the body at further risk as the stimulatory and euphoric qualities of the drug may render the user oblivious to their energy expenditure for quite some time. Diuretics such as alcohol may exacerbate these risks further.
MDMA causes a reduction in the concentration of serotonin transporters (SERTs) in the brain. The rate at which the brain recovers from serotonergic changes is unclear. A number of studies [76] have demonstrated lasting serotonergic changes occurring due to MDMA exposure. Other studies[77][78] have suggested that the brain may recover from serotonergic damage; however, damage caused by heavy, prolonged use of MDMA may be long lasting.[citation needed]
Some studies show that MDMA may be neurotoxic in humans.[79][80] Other studies, however, suggest that any potential brain damage may be at least partially reversible following prolonged abstinence from MDMA.[78][81] However, other studies suggest that SERT-depletion arises from long-term MDMA use due to receptor down-regulation, rather than true neurotoxicity.[82] Depression and deficits in memory have been shown to occur more frequently in long-term MDMA users.[83][84] However, some recent studies have suggested that MDMA use may not be associated with chronic depression.[85][86]
One study on MDMA toxicity, by George A. Ricaurte of Johns Hopkins School of Medicine, which claimed that a single recreational dose of MDMA could cause Parkinson's Disease in later life due to severe dopaminergic stress, was actually retracted by Ricaurte himself after he discovered his lab had administered not MDMA but methamphetamine, which is known to cause dopaminergic changes similar to the serotonergic changes caused by MDMA.[87] Ricaurte blamed this mistake on the chemical supply company that sold the material to his lab. Most studies have found that levels of the dopamine transporter (or other markers of dopamine function) in MDMA users deserve further study or are normal.[88][89][90][91][92][93][94]
Another concern associated with MDMA use is toxicity from chemicals other than MDMA in ecstasy tablets. Due to its near-universal illegality, the purity of a substance sold as ecstasy is unknown to the typical user. The MDMA content of tablets varies widely between regions and different brands of pills and fluctuates somewhat each year. Pills may contain other active substances meant to stimulate in a way similar to MDMA, such as amphetamine, methamphetamine, ephedrine, or caffeine, all of which may be comparatively cheap to produce and can help to boost profit overall. In some cases, tablets sold as ecstasy do not even contain any MDMA. Instead they may contain an assortment of presumably undesirable drugs such as paracetamol, ibuprofen, etc.[95]
There have been a number of deaths attributed to PMA, a potent and highly neurotoxic hallucinogenic amphetamine, being sold as Ecstasy. PMA is unique in its ability to quickly elevate body temperature and heart rate at relatively low doses, especially in comparison to MDMA.[96] Hence, a user who believes he is consuming two 120 mg pills of MDMA could actually be consuming a dose of PMA that is potentially lethal, depending on the purity of the pill. Not only does PMA cause the release of serotonin, but also acts as an MAOI. When combined with an MDMA-like substance, serotonin syndrome can result.
Harm assessment
The chief executive of the U.K. Medical Research Council stated that MDMA is "on the bottom of the scale of harm", and was rated to be of lesser concern than alcohol, tobacco, as well as several classes of prescription medications, when examining the harmfulness of twenty popular recreational drugs. The U.K. study placed great weight on the risk for acute physical harm, the propensity for physical and psychological dependency on the drug, and the negative familial and societal impacts of the drug. Based on these factors, the study placed MDMA at number 18 in the list.[97]
David Nutt, chairman of the U.K. Advisory Council on the Misuse of Drugs, stated in the Journal of Psychopharmacology in January 2009 that ecstasy use compared favorably with horse riding in terms of risk, leading to around 50 deaths a year compared to about 100 from horse riding. The ACMD subsequently made clear that Professor Nutt's comments were not made on behalf of the ACMD.[98]
In February 2009, the the UK Advisory Council on the Misuse of Drugs issued "A review of MDMA ('ecstasy'), its harms and classification under the Misuse of Drugs Act 1971", which recommended that MDMA be downgraded in the UK from a class A drug to a class B drug.[99] The UK Home Office rejected this advice from it's independent scientific advisory board, "saying it is not prepared to send a message to young people that it takes ecstasy less seriously".[100]
See also
- Effects classification
- Anorectic
- Antidepressant
- Anxiolytic
- Entactogen
- Entheogen
- Euphoriant
- Hallucinogen
- Psychedelic
- Stimulant
- Sympathomimetic
- Pharmacological classification
- Chemical classification
- Substituted phenethylamine (PEA)
- Substituted amphetamine (AMP)
- Substituted methylenedioxyphenethylamine (MDxx)
- Similar or related compounds
- Amphetamine (Adderall, Dexedrine; "Speed")
- Methamphetamine (Desoxyn; "Meth", "Crack", "Crystal", etc)
- Methylenedioxyamphetamine (MDA; "Mellow Drug of America", etc)
- Methylenedioxyethylamphetamine (MDEA; "Eve")
- Methylbenzodioxolylbutanamine (MBDB; "Eden")
- Methylenedioxymethcathinone (MDMC; Methylone; "Explosion", "Ease")
- Paramethoxyamphetamine (PMA; "Death")
- 4-Methylmethcathinone (4-MMC; Mephedrone; "Hammer", "Cameleon")
- Associated topics
- Electronica (also house, techno, and trance)
- Psychotherapy (also counseling and psychology)
- Rave (also dance party and rave music)
- Recreational drug use (or drug abuse)
References
- ^ Stimulants, Narcotics, Hallucinogens - Drugs, Pregnancy, and Lactation., Gerald G. Briggs, OB/GYN News, June 1, 2003.
- ^ "Methylenedioxymethamphetamine (MDMA, Ecstasy)". Drugs and Human Performance Fact Sheets. National Highway Traffic Safety Administration.
- ^ "Where is Ecstasy Legal?"
- ^ Ecstasy Abuse Information.
- ^ MAPS: Psychedelic Research Worldwide
- ^ a b c d e f David Pearce. "MDMA / Ecstasy: Not For Chemists - Drug Slang".
- ^ a b Bernschneider-Reif S, Oxler F, Freudenmann RW (2006). "The Origin of MDMA ("Ecstasy")--Separating the Facts From the Myths". Pharmazie. 61 (11): 966–972. doi:10.1111/j.1360-0443.2006.01511.x. PMID 17152992.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Firma E. Merck in Darmstadt (1914-05-16). "German Patent 274350: Verfahren zur Darstellung von Alkyloxyaryl-, Dialkyloxyaryl- und Alkylendioxyarylaminopropanen bzw. deren am Stickstoff monoalkylierten Derivaten". Kaiserliches Patentamt. Retrieved 2009-04-12.
- ^ Firma E. Merck in Darmstadt (1914-10-15). "German Patent 279194: Verfahren zur Darstellung von Hydrastinin Derivaten". Kaiserliches Patentamt.
- ^ Hardman HF, Haavik CO, Seevers MH (1973). "Relationship of the Structure of Mescaline and Seven Analogs to Toxicity and Behavior in Five Species of Laboratory Animals". Toxicology Applied Pharmacology. 25 (2): 299–309. doi:10.1016/S0041-008X(73)80016-X. PMID 4197635.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Kasuya Y (1958). "Antispasmodics. XI. Synthesis of Aralkylamines and Their Quarternary Ammonium Derivatives". Yakugaku Zasshi. 78: 509–511.
- ^ The first confirmed sample was seized and identified by Chicago Police in 1970, see Sreenivasan VR (1972). "Problems in Identification of Methylenedioxy and Methoxy Amphetamines". Journal of Criminal Law, Criminology & Police Science. 63 (2): 304–312. doi:10.2307/1142315.
- ^ a b Ann Shulgin; Alexander Shulgin (1991). PiHKAL: A Chemical Love Story. Part I. Chapter 12. Transform Press. ISBN 0-9630096-0-5.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Shulgin AT; Nichols DE (1978). "Characterization of Three New Psychomimetics.". In Willette, Robert E.; Stillman, Richard Joseph (ed.). The Psychopharmacology of Hallucinogens. New York: Pergamon Press. pp. 74–83. ISBN 0-08-021938-1.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Bennett, Drake (2005-01-30). "Dr. Ecstasy". New York Times Magazine. New York Times.
- ^ Myron J. Stolaroff (2005). The Secret Chief Revealed. MAPS. pp. 16–18. ISBN 0-9660019-6-6.
- ^ The Austin Chronicle - "Countdown to Ecstasy." by Marc Savlov
- ^ "Pharmaceutical Company Unravels Drug's Chequered Past". 2005.
- ^ Erowid MDMA Vault: Info #3 on Scheduling.
- ^ Primetime with Peter Jennings.
- ^ Bibliography of Psychadelic Research Studies. collected by the Multidisciplinary Association for Psychedelic Studies
- ^ Greer G. Tolbert R. "The Therapeutic Use of MDMA in Ecstasy: The Clinical, Pharmacological, and Neurotoxicological Effects of the Drug MDMA." 1990 (ed Peroutka, SJ) Boston, p. 21-36
- ^ Doblin R (2002). "A Clinical Plan for MDMA (Ecstasy) in the Treatment of Post-Traumatic Stress Disorder (PTSD): Partnering with the FDA". J Psychoactive Drugs. 34 (2): 185–194. PMID 12691208.
- ^ Sessa B, Nutt DJ (2007). "MDMA, Politics and Medical Research: Have We Thrown the Baby Out With the Bathwater?". J. Psychopharmacol. (Oxford). 21 (8): 787–791. doi:10.1177/0269881107084738. PMID 17984158.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Greer, G.; Tolbert, R. (1986). "Subjective Reports of the Effects of MDMA in a Clinical Setting". Journal of Psychoactive Drugs. 18 (4): 319–327.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Greer, G.; Tolbert, R. (1998). "A Method of Conducting Therapeutic Sessions with MDMA". Journal of Psychoactive Drugs. 30 (4): 371–379.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Shroder, T. (November 25, 2007). "The Peace Drug". Washington Post. p. W12.
{{cite news}}: Check date values in:|date=(help) - ^ Bryan Mitchell (March 4, 2009). "'Party' Drug Could be PTSD Treatment". Military.Com.
{{cite news}}: Check date values in:|date=(help) - ^ Promising Results from Pioneering MDMA (Ecstasy)-Assisted Psychotherapy Pilot Study of Subjects with Treatment-Resistant Posttraumatic Stress Disorder (PTSD)
- ^ European Monitoring Centre for Drugs and Drug Addiction (2008). Annual report: the state of the drugs problem in Europe (PDF). Luxembourg: Office for Official Publications of the European Communities. p. 49. ISBN 978-92-9168-324-6.
- ^ United Nations Office on Drugs and Crime (2008). World drug report (PDF). United Nations Publications. p. 271. ISBN 978-92-1-148229-4.
- ^ Leone, M (July 12, 2000). "The Serotonergic Agent m-Chlorophenylpiperazine Induces Migraine Attacks: A Controlled Study". Neurology. 55 (1): 136–139. PMID 10891925.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Martin RS & Martin GR. Investigations into migraine pathogenesis: time course for effects of mCPP, BW723C86 or glyceryl trinitrate on appearance of Fos-like immunoreactivity in rat trigeminal nucleus caudalis (TNC). Cephalalgia 2001; 21:46–52. London. ISSN 0333-10245
- ^ Petkov VD, Belcheva S, Konstantinova E. Anxiolytic effects of dotarizine, a possible antimigraine drug. Methods Find Exp Clin Pharmacol. 1995 December;17(10):659-68.
- ^ University of Maryland, College Park Center for Substance Abuse Research. "Ecstasy:CESAR".
- ^ "Director's Report to the National Advisory Council on Drug Abuse". National Institute on Drug Abuse. May 2000.
- ^ Erowid Experience Vaults: Vicks Used With Ecstasy - Important Warning About Vicks on Ecstasy.
- ^ Analytica Chimica Acta, 596(2), 231-241; 2007
- ^ Chemical Research in Toxicology, 19(10), 1294-1304; 2006
- ^ Reductive aminations of carbonyl compounds with borohydride and borane reducing agents. Baxter, Ellen W.; Reitz, Allen B. Organic Reactions (Hoboken, NJ, United States) (2002), 59
- ^ Forensic Science International, 155(2-3), 141-157; 2005
- ^ Nov 05 DEA Microgram newsletter
- ^ Fleckenstein AE, Volz TJ, Riddle EL, Gibb JW, Hanson GR (2007). "New Insights into the Mechanism of Action of Amphetamines". Annu. Rev. Pharmacol. Toxicol. 47: 681–698. doi:10.1146/annurev.pharmtox.47.120505.105140. PMID 17209801.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "This is Your Brain". TheDEA.
- ^ "Ecstasy Really Does Unleash the Love Hormone". New Scientist. April 4, 2007.
{{cite web}}: Check date values in:|date=(help) - ^ Dumont GJ, Sweep FC, van der Steen R, Hermsen R, Donders AR, Touw DJ, van Gerven JM, Buitelaar JK, Verkes RJ. (2009). "Increased oxytocin concentrations and prosocial feelings in humans after ecstasy (3,4-methylenedioxymethamphetamine) administration". Soc Neurosci. 4 (4): 359–366. PMID 19562632.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Verebey K, Alrazi J, Jaffe JH (1988). "The complications of 'ecstasy' (MDMA)". JAMA. 259 (11): 1649–1650. doi:10.1001/jama.259.11.1649. PMID 2893845.
{{cite journal}}: Unknown parameter|doi_brokendate=ignored (|doi-broken-date=suggested) (help)CS1 maint: multiple names: authors list (link) - ^ eMedicine - Toxicity, MDMA: Article by David Yew
- ^ de la Torre R, Farré M, Roset PN; et al. (2004). "Human pharmacology of MDMA: pharmacokinetics, metabolism, and disposition". Ther Drug Monit. 26 (2): 137–44. doi:10.1097/00007691-200404000-00009. PMID 15228154.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Shima N, Kamata H, Katagi M, Tsuchihashi H, Sakuma T, Nemoto N (2007). "Direct Determination of Glucuronide and Sulfate of 4-Hydroxy-3-methoxymethamphetamine, the Main Metabolite of MDMA, in Human Urine". J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 857 (1): 123–129. doi:10.1016/j.jchromb.2007.07.003. PMID 17643356.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b J. K. Fallon, A. T. Kicman, J. A. Henry, P. J. Milligan, D. A. Cowan, A. J. Hutt (1999). "Stereospecific Analysis and Enantiomeric Disposition of 3,4-Methylenedioxymethamphetamine (Ecstasy) in Humans". Clinical Chemistry. 45 (7): 1058–1069.
{{cite journal}}: Unknown parameter|day=ignored (help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ D. Hensley, J. T. Cody (1999). "Simultaneous Determination of Amphetamine, Methamphetamine, Methylenedioxyamphetamine (MDA), Methylenedioxymethamphetamine (MDMA), and Methylenedioxyethylamphetamine (MDEA) Enantiomers by GC–MS" (PDF). Journal of Analytical Toxicology. 23 (6): 518–523.
- ^ Vuori E, Henry JA, Ojanperä I; et al. (2003). "Death following ingestion of MDMA (ecstasy) and moclobemide". Addiction. 98 (3): 365–8. doi:10.1046/j.1360-0443.2003.00292.x. PMID 12603236.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ The Prozac Misunderstanding, at TheDEA.org
- ^ Primetime video
- ^ "Erowid MDMA Vault: Effects". Erowid.
- ^ "What Is Ecstasy?". DanceSafe.
- ^ "MDMA Technical FAQ". TheDEA.org. May 2003.
- ^ Whetstone, Kobie J. (April 16, 2009- June 11, 2009). "An Analysis of MDMA-Induced Neurotoxicity".
{{cite web}}: Check date values in:|date=(help) - ^ {{cite journal |title=Causes and consequences of methamphetamine and MDMA toxicity.|author=Quinton, M.S. and Yamamoto, B.K.|journal=The AAPS Journal|volume=8|number=2|pages=337--337|year=2006|publisher=Springer
- ^ Verheyden SL, Henry JA, Curran HV (2003). "Acute, sub-acute and long-term subjective consequences of 'ecstasy' (MDMA) consumption in 430 regular users". Hum Psychopharmacol. 18 (7): 507–17. doi:10.1002/hup.529. PMID 14533132.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Verheyden SL, Maidment R, Curran HV (2003). "Quitting ecstasy: an investigation of why people stop taking the drug and their subsequent mental health". J. Psychopharmacol. (Oxford). 17 (4): 371–8. doi:10.1177/0269881103174014. PMID 14870948.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Laws KR, Kokkalis J (2007). "Ecstasy (MDMA) and memory function: a meta-analytic update". Human Psychopharmacology: Clinical and Experimental. 22 (6): 381–88. doi:10.1002/hup.857.
- ^ Rodgers J, Buchanan T, Scholey AB, Heffernan TM, Ling J, Parrott AC (2003). "Patterns of drug use and the influence of gender on self-reports of memory ability in ecstasy users: a web-based study". J. Psychopharmacol. (Oxford). 17 (4): 389–96. doi:10.1177/0269881103174016. PMID 14870950.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Jones AL, Simpson KJ (1999). "Review article: mechanisms and management of hepatotoxicity in ecstasy (MDMA) and amphetamine intoxications" (PDF). Aliment. Pharmacol. Ther. 13 (2): 129–33. doi:10.1046/j.1365-2036.1999.00454.x. PMID 10102941.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Milosevic A, Agrawal N, Redfearn P, Mair L (1999). "The occurrence of toothwear in users of Ecstasy (3,4-methylenedioxymethamphetamine)" (PDF). Community Dent Oral Epidemiol. 27 (4): 283–7. doi:10.1111/j.1600-0528.1998.tb02022.x. PMID 10403088.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Creighton FJ, Black DL, Hyde CE (1991). "'Ecstasy' psychosis and flashbacks" (PDF). Br J Psychiatry. 159: 713–5. doi:10.1192/bjp.159.5.713. PMID 1684523.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ MAPS. "Documents from the DEA Scheduling Hearing of MDMA,".
- ^ SF Gate News March 21, 2001
- ^ Federation of American Scientists MDMA Testimony (PDF)
- ^ U.S. Federal Sentencing Guidelines (2007)
- ^ E for Ecstasy by Nicholas Saunders, Appendix 1: Reference Section
- ^ Nimmo SM, Kennedy BW, Tullett WM, Blyth AS, Dougall JR (1993). "Drug-induced hyperthermia". Anaesthesia. 48 (10): 892–5. doi:10.1111/j.1365-2044.1993.tb07423.x. PMID 7902026.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Malberg JE, Seiden LS (1998). "Small changes in ambient temperature cause large changes in 3,4-methylenedioxymethamphetamine (MDMA)-induced serotonin neurotoxicity and core body temperature in the rat". J. Neurosci. 18 (13): 5086–94. PMID 9634574.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Wolff K, Tsapakis EM, Winstock AR; et al. (2006). "Vasopressin and oxytocin secretion in response to the consumption of ecstasy in a clubbing population". J. Psychopharmacol. (Oxford). 20 (3): 400–10. doi:10.1177/0269881106061514. PMID 16574714.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Fischer C, Hatzidimitriou G, Wlos J, Katz J, Ricaurte G (1995). "Reorganization of ascending 5-HT axon projections in animals previously exposed to the recreational drug (+/-)3,4-methylenedioxymethamphetamine (MDMA, "ecstasy")". J. Neurosci. 15 (8): 5476–85. PMID 7643196.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Scheffel U, Szabo Z, Mathews WB; et al. (1998). "In vivo detection of short- and long-term MDMA neurotoxicity--a positron emission tomography study in the living baboon brain". Synapse. 29 (2): 183–92. doi:10.1002/(SICI)1098-2396(199806)29:2<183::AID-SYN9>3.0.CO;2-3. PMID 9593108.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b Reneman L, Lavalaye J, Schmand B; et al. (2001). "Cortical serotonin transporter density and verbal memory in individuals who stopped using 3,4-methylenedioxymethamphetamine (MDMA or "ecstasy"): preliminary findings". Arch. Gen. Psychiatry. 58 (10): 901–6. doi:10.1001/archpsyc.58.10.901. PMID 11576026.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Neurotoxicity at Dancesafe.org
- ^ Does MDMA Cause Brain Damage?, Matthew Baggott, and John Mendelson
- ^ Research on Ecstasy Is Clouded by Errors, Donald G. McNeil Jr., New York Times, December 2, 2003.
- ^ "p-Chlorophenylalanine Changes Serotonin Transporter mRNA Levels and Expression of the Gene Product", Journal of Neurochemistry, 1996. Online copy (pdf)
- ^ Depression at DanceSafe.org
- ^ Wareing, M. and Fisk, J.E. and Murphy, P.N. (2000). "Working memory deficits in current and previous users of MDMA ('ecstasy')" (PDF). Br J Psychol. pp. 181--8.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ^ Study claims recreational ecstasy use and depression unrelated, Wikinews, April 26, 2006]
- ^ Guillot C, Greenway D (2006). "Recreational ecstasy use and depression". J. Psychopharmacol. (Oxford). 20 (3): 411–6. doi:10.1177/0269881106063265. PMID 16574715.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Ecstasy Study Botched, Retracted, Kristen Philipkoski, Wired.com, 09.05.03
- ^ McCann UD, Ricaurte GA (2001). "Caveat emptor: editors beware" (PDF). Neuropsychopharmacology. 24 (3): 333–6. doi:10.1016/S0893-133X(00)00171-8. PMID 11256359.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Green AR, Mechan AO, Elliott JM, O'Shea E, Colado MI (2003). "The pharmacology and clinical pharmacology of 3,4-methylenedioxymethamphetamine (MDMA, "ecstasy")". Pharmacol. Rev. 55 (3): 463–508. doi:10.1124/pr.55.3.3. PMID 12869661.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Zakzanis KK, Campbell Z, Jovanovski D (2007). "The neuropsychology of ecstasy (MDMA) use: a quantitative review". Hum Psychopharmacol. 22 (7): 427–35. doi:10.1002/hup.873. PMID 17896346.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Gijsman HJ, Verkes RJ, van Gerven JM, Cohen AF (1999). "MDMA study" (PDF). Neuropsychopharmacology. 21 (4): 597. doi:10.1016/S0893-133X(99)00021-4. PMID 10481843.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Kish SJ (2002). "How strong is the evidence that brain serotonin neurons are damaged in human users of ecstasy?". Pharmacol. Biochem. Behav. 71 (4): 845–55. doi:10.1016/S0091-3057(01)00708-0. PMID 11888575.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Kish SJ (2003). "What is the evidence that Ecstasy (MDMA) can cause Parkinson's disease?" (PDF). Mov. Disord. 18 (11): 1219–23. doi:10.1002/mds.10643. PMID 14639660.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Aghajanian & Liebermann (2001) 'Caveat Emptor: Reseearchers Beware' Neuropsychopharmacology 24,3:335-6
- ^ Summary Statistics for EcstasyData.org Lab Testing Results
- ^ Club Drug Update, Jeff Morelock, PRP Online, Spring 2003
- ^ Scientists want new drug rankings, BBC News, 23 March 2007
- ^ Ecstasy 'not worse than riding', BBC News, 7 February 2009
- ^ A review of MDMA ('ecstasy'), its harms and classification under the Misuse of Drugs Act 1971, Advisory Council on the Misuse of Drugs (ACMD), 11 February 2009
- ^ UK government rejects advice from drugs adviser to downgrade ecstasy, BMJ, 13 February 2009
Further reading
- Julie Holland - "Ecstasy: The Complete Guide: A Comprehensive Look at the Risks and Benefits of MDMA" Park Street Press (2001).
- Jerome Beck, Marsha Rosenbaum - "Pursuit of Ecstasy: The MDMA Experience" SUNY Press (1994).
- Leslie Iversen - "Speed, Ecstasy, Ritalin: The Science of Amphetamines" Oxford University Press (2008).
- David Pearce - "Utopian Pharmacology: Mental Health in the Third Millenium: MDMA and Beyond" (2008).
- Kobie Whetstone - "The Brain Damage of MDMA ("Ecstasy"): Causes and Prevention for the Recreational User" (2009).
- Kobie Whetstone - "An Analysis of MDMA-induced Neurotoxicity" (2009).
- Earth Erowid - "Do Antioxidants Protect Against MDMA Hangover, Tolerance, and Neurotoxicity?" Erowid Extracts (2001).
- Marsha Rosenbaum, Rick Doblin - "Why MDMA Should Not Have Been Made Illegal" SAGE Publications (1991).
- Nature News - "Ecstasy Could Augment the Benefits of Psychotherapy" (2008).
- Reuters Health - "Ecstasy May Help PTSD Victims Get Better" (2009).
- The Economist - "Agony and Ecstasy: Ecstasy May Be Good For Those Who Can’t Get Over Something Truly Horrible" (2008).
- New Scientist - "Ecstasy's Long-Term Effects Revealed" (2009).
- Matt Palmquist Miller-McCune - "The Ecstasy and the Agony" (August 2009).
- ACMD - "MDMA (Ecstasy): A Review of its Harms and Classification Under the Misuse of Drugs Act 1971" UK Home Office (2009).
- NIDA InfoFacts - "MDMA (Ecstasy)" (August 2008).
External links
- Alexander Shulgin & Ann Shulgin - PiHKAL: "#109 MDMA: MDM; ADAM; Ecstasy; 3,4-Methylenedioxy-N-methylamphetamine" (1991).
- ABC News - "Primetime Special: Peter Jennings - Ecstasy Rising" (2004).
- The Vaults of Erowid: "MDMA ("Ecstasy")".
- Erowid Experience Vaults: "MDMA ("Ecstasy", "E", "X") Reports".
- DanceSafe: "What is Ecstasy?".
- MAPS: "R&D Medicines > MDMA".
- EcstasyData.org: Providing Access to Lab Testing Results for Street Ecstasy Tablets.
- PillReports.com: Ecstasy Test Results Database by Enlighten.

