| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Thiirane | |||
| Systematic IUPAC name
Thiacyclopropane | |||
| Other names | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 102379 | |||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.006.359 | ||
| EC Number |
| ||
| 1278 | |||
| KEGG | |||
| MeSH | ethylene+sulfide | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1992 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H4S | |||
| Molar mass | 60.11 g·mol−1 | ||
| Appearance | Pale, yellow liquid | ||
| Density | 1.01 g cm−3 | ||
| Melting point | −109 °C (−164 °F; 164 K) | ||
| Boiling point | 56 °C; 133 °F; 329 K | ||
| Vapor pressure | 28.6 kPa (at 20 °C) | ||
| Thermochemistry | |||
Std enthalpy of
formation (ΔfH⦵298) |
51-53 kJ mol−1 | ||
Std enthalpy of
combustion (ΔcH⦵298) |
-2.0126 MJ mol−1 | ||
| Hazards | |||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H225, H301, H318, H331 | |||
| P210, P261, P280, P301+P310, P305+P351+P338, P311 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 10 °C (50 °F; 283 K) | ||
| Related compounds | |||
Related heterocycles
|
Ethylene oxide Aziridine Borirane | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
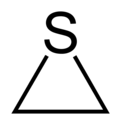
Thiirane, more commonly known as ethylene sulfide, is the cyclic chemical compound with the formula C2H4S.[2] It is the smallest sulfur-containing heterocycle and the simplest episulfide. Like many organosulfur compounds, this species has a highly unpleasant odour. Thiirane is also used to describe any derivative of the parent ethylene sulfide.
Structure[edit]
According to electron diffraction, the C-C and C-S distances in ethylene sulfide are respectively 1.473 and 1.811 Å. The C-C-S and C-S-C angles are respectively 66.0 and 48.0°.[3]
Preparation and reactions[edit]
It can be prepared by the reaction of ethylene carbonate and KSCN.[4] For this purpose the KSCN is first melted under vacuum to remove water.
- KSCN + C2H4O2CO → KOCN + C2H4S + CO2
Ethylenesulfide adds to amines to afford 2-mercaptoethylamines,[5] which are good chelating ligands.
- C2H4S + R2NH → R2NCH2CH2SH
This process is often called mercaptoethylation.[6]
Oxidation of thiirane with periodate gives ethylene episulfoxide.
References[edit]
- ^ a b "thiirane (CHEBI:30977)". Chemical Entities of Biological Interest (ChEBI). UK: European Bioinformatics Institute.
- ^ Warren Chew; David N. Harpp (1993). "Recent aspects of thiirane chemistry". Journal of Sulfur Chemistry. 15 (1): 1–39. doi:10.1080/01961779308050628.
- ^ Wataru Ando; Nami Choi; Norihiro Tokitoh (1996). "Thiiranes and Thiirenes: Monocyclic". Comprehensive Heterocyclic Chemistry II. Vol. 1A. pp. 173–240. doi:10.1016/B978-008096518-5.00005-8. ISBN 978-0-08-096518-5.
- ^ Searles, S.; Lutz, E. F.; Hays, H. R.; Mortensen, H. E. (1962). "Ethylene Sulfide". Organic Syntheses. 42: 59. doi:10.15227/orgsyn.042.0059.
- ^ R. J. Cremlyn "An Introduction to Organosulfur Chemistry" John Wiley and Sons: Chichester (1996). ISBN 0-471-95512-4.
- ^ Gunars Zelans; Jacquelyn Gervay-Hague; Ivy Maulie (2010). "Ethylene Sulfide". Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons. doi:10.1002/047084289X.re079.pub2. ISBN 978-0-471-93623-7.


Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction