| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Stannine | |||
| Other names
Stannin
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H6Sn | |||
| Molar mass | 184.813 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
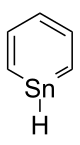
Stannabenzene (C5H6Sn) is the parent representative of a group of organotin compounds that are related to benzene with a carbon atom replaced by a tin atom. Stannabenzene itself has been studied by computational chemistry,[1] but has not been isolated.
Stable derivatives of stannabenzene
[edit]Stable derivatives of stannabenzene have been isolated. The 2-stannanaphthalene depicted below is stable in an inert atmosphere at temperatures below 140 °C.[2] The tin to carbon bond in this compound is shielded from potential reactants by two very bulky groups, one tert-butyl group and the even larger 2,4,6-tris[bis(trimethylsilyl)methyl]phenyl or Tbt group. The two Sn-C bonds have bond lengths of 202.9 and 208.1 pm which are shorter than those for Sn-C single bonds (214 pm) and comparable to that of known Sn=C double bonds (201.6 pm). The C-C bonds show little variation with bond lengths between 135.6 and 144.3 pm signaling that this compound is aromatic.
Tbt-substituted 9-stannaphenanthrene was reported in 2005.[3] At room temperature it forms the [4+2] cycloadduct.
Tbt-substituted stannabenzene was reported in 2010.[4] At room-temperature it quantitatively forms the DA dimer.

Tbt-substituted stannabenzene synthesis. Reagents lithium aluminium hydride (step 2), NBS (step 3), LDA (step 4)
See also
[edit]- 6-membered aromatic rings with one carbon replaced by another group: borabenzene, silabenzene, germabenzene, stannabenzene, pyridine, phosphorine, arsabenzene, bismabenzene, pyrylium, thiopyrylium, selenopyrylium, telluropyrylium
References
[edit]- ^ Ebrahimi, Arash Afshar; Ghiasi, Reza; Foroutan-Nejad, Cina (2010). "Topological characteristics of the ring critical points and the aromaticity of groups IIIA to VIA hetero-benzenes". Journal of Molecular Structure: THEOCHEM. 941 (1–3): 47–52. doi:10.1016/j.theochem.2009.10.038.
- ^ Mizuhata, Yoshiyuki; Sasamori, Takahiro; Takeda, Nobuhiro; Tokitoh, Norihiro (2006). "A Stable Neutral Stannaaromatic Compound: Synthesis, Structure and Complexation of a Kinetically Stabilized 2-Stannanaphthalene". Journal of the American Chemical Society. 128 (4): 1050–1. doi:10.1021/ja057531d. PMID 16433501.
- ^ Generation of 9-Stannaphenanthrene and Its Reactivities Yoshiyuki Mizuhata, Nobuhiro Takeda, Takahiro Sasamori and Norihiro Tokitoh Chemistry Letters Volume 34 Number 8 Year 2005 Page 1088 doi:10.1246/cl.2005.1088
- ^ Generation of Stannabenzenes and Their Properties Yoshiyuki Mizuhata, Naoya Noda, and Norihiro Tokitoh Organometallics, 2010, 29 (21), pp 4781–4784 doi:10.1021/om100382n


Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction