| |
| Names | |
|---|---|
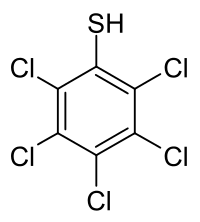
| Preferred IUPAC name
Pentachlorobenzenethiol | |
| Other names
Pentachlorobenzenethiol, pentachlorothiophenol, PCBT
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.004.644 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6HCl5S | |
| Molar mass | 282.38 g·mol−1 |
| Appearance | Gray solid |
| Density | 1.7±0.1 g/cm3 |
| Melting point | 231.5 °C (448.7 °F; 504.6 K) |
| Insoluble | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| Flash point | 144.6 °C (292.3 °F; 417.8 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Pentachlorobenzenethiol is a chemical compound from the group of thiols and organochlorine compounds.[1] The chemical formula is C
6HCl
5S.[2]
Synthesis[edit]
Pentachlorobenzenethiol can be obtained from hexachlorobenzene.[3]
Properties[edit]
Pentachlorobenzenethiol is a combustible gray solid with an unpleasant odor, practically insoluble in water. It has a monoclinic crystal structure.[4] The compound is not well-biodegradable and presumably bioaccumulable and toxic for aquatic organisms. Pentachlorobenzenethiol is itself a metabolite of hexachlorobenzene and is found in the urine and the excretions of animals receiving hexachlorobenzene.[5] Pentachlorobenzenethiol has a high potential for long-range transport via air as it is very slowly degraded in atmosphere.[6]
Applications[edit]
Pentachlorobenzenethiol is used in the rubber industry. The compound is added to rubber (both natural and synthetic) to facilitate processing (mastication).
See also[edit]
References[edit]
- ^ "Pentachlorobenzenethiol". chemindustry.com. Retrieved 9 June 2017.
- ^ "2,3,4,5,6-pentachlorobenzenethiol". Retrieved 9 June 2017.
- ^ Kulka, Marshall (1959). "Reaction of Hexachlorobenzene with Mercaptides". J. Org. Chem. 24 (4): 235–237. doi:10.1021/jo01084a021.
- ^ Wojcik, G.; Charbonneau, G. P.; Delugeard, Y.; Toupet, L. (1980). "The disordered crystal structure of pentachlorothiophenol". Acta Crystallogr. B36 (2): 506–507. Bibcode:1980AcCrB..36..506W. doi:10.1107/S0567740880003706.
- ^ Koss, G.; Koransky, W.; Steinbach, K. (June 1976). "Studies on the toxicology of hexachlorobenzene: II. Identification and determination of metabolites". Archives of Toxicology. 35 (2): 107–114. doi:10.1007/BF00372764. PMID 947309. S2CID 25403466.
- ^ "TC NES SUBGROUP ON IDENTIFICATION OF PBT AND VPVB SUBSTANCES". EChA. echa.europa.eu. Retrieved 9 June 2017.
Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction