| |
| Names | |
|---|---|
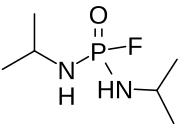
| Preferred IUPAC name
N,N′-Di(propan-2-yl)phosphorodiamidic fluoride | |
| Other names
Bis(isopropylamino)fluorophosphine oxide; Isopestox
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.006.130 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H16FN2OP | |
| Molar mass | 182.179 g·mol−1 |
| Density | 1.2 |
| Melting point | 65 °C (149 °F; 338 K) |
| Boiling point | 125 °C (257 °F; 398 K) |
| 80 g/L | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Highly toxic |
| GHS labelling: | |
 
| |
| Danger | |
| H301, H370 | |
| P260, P264, P270, P301+P310, P307+P311, P321, P330, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Mipafox is a highly toxic organophosphate insecticide that is an irreversible acetylcholinesterase inhibitor and is resistant to cholinesterase reactivators.[1] It was developed in the 1950s and is now believed to be no longer in use.[2]
Toxicity[edit]
There are case reports of delayed neurotoxicity and paralysis due to acute exposure to mipafox.[3]
Synthesis[edit]
Phosphoryl chloride is first reacted with isopropylamine. The resulting product is then reacted with potassium fluoride or ammonium fluoride to produce mipafox.[4]
See also[edit]
References[edit]
- ^ Mangas, I; Taylor, P; Vilanova, E; Estévez, J; França, TC; Komives, E; Radić, Z (March 2016). "Resolving pathways of interaction of mipafox and a sarin analog with human acetylcholinesterase by kinetics, mass spectrometry and molecular modeling approaches". Archives of Toxicology. 90 (3): 603–16. doi:10.1007/s00204-015-1481-1. PMC 4833118. PMID 25743373.
- ^ "The WHO Recommended Classification of Pesticides by Hazard and Guidelines to Classification 2019" (PDF). World Health Organization.
- ^ BIDSTRUP, PL; BONNELL, JA; BECKETT, AG (16 May 1953). "Paralysis following poisoning by a new organic phosphorus insecticide (mipafox); report on two cases". British Medical Journal. 1 (4819): 1068–72. doi:10.1136/bmj.1.4819.1068. PMC 2016450. PMID 13042137.
- ^ "Process for the preparation of bisisopropyl-amino-fluoro-phosphine oxide".
Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction