| |
| Names | |
|---|---|
| Other names
Lithium 2-hydroxypropanoate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.011.602 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C 3H 5LiO 3[1][2] | |
| Molar mass | 96.01 |
| Appearance | Amorphous solid |
| Density | g/cm3 |
| Melting point | 300 °C (572 °F; 573 K) |
| Very soluble | |
| Hazards | |
| GHS labelling:[3] | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| Related compounds | |
Other cations
|
Sodium lactate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
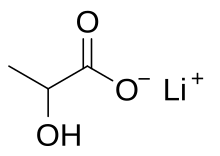
Lithium lactate is a chemical compound, a salt of lithium and lactic acid with the formula CH3CH(OH)COOLi,[4] an amorphous solid, very soluble in water.[5]
Synthesis[edit]
Synthesis is by neutralization of lactic acid with lithium hydroxide:
- LiOH + CH3CH(OH)COOH → CH3CH(OH)COOLi + H2O
Physical properties[edit]
Lithium lactate forms an amorphous solid.
It dissolves very well in water[6] and organic solvents.
The compound demonstrates optical isomerism.
Lithium lactate emits acrid smoke when heated to decomposition.[7]
Chemical properties[edit]
Lithium lactate reacts with triphosgene to obtain lactic acid-O-internal anhydride.[8] It can be used as a precursor to prepare Li4SiO4,[9] Li4Ti5O12/C[10] and other materials.
Use[edit]
Lithium lactate is part of drugs that promote the excretion of uric acid from the body.[11]
It is also used as an Antipsychotic.[12]
References[edit]
- ^ "867-55-0 CAS | LITHIUM LACTATE | Laboratory Chemicals | Article No. 04444". Loba Chemie. Retrieved 17 January 2022.
- ^ "27848-80-2 - L-(+)-Lactic acid lithium salt, Thermo Scientific - Lithium Lactate - J18160 - Alfa Aesar". Alfa Aesar. Retrieved 17 January 2022.
- ^ "Lithium lactate". pubchem.ncbi.nlm.nih.gov. Retrieved 22 January 2022.
- ^ "Lithium Lactate". American Elements. Retrieved 17 January 2022.
- ^ "Lithium lactate". Sigma Aldrich. Retrieved 17 January 2022.
- ^ Lewis, Robert A. (31 May 2016). Hawley's Condensed Chemical Dictionary. John Wiley & Sons. p. 840. ISBN 978-1-118-13515-0. Retrieved 17 January 2022.
- ^ Lewis, Richard J. (13 June 2008). Hazardous Chemicals Desk Reference. John Wiley & Sons. p. 844. ISBN 978-0-470-18024-2. Retrieved 17 January 2022.
- ^ Nölle, Roman; Schmiegel, Jan-Patrick; Winter, Martin; Placke, Tobias (14 January 2020). "Tailoring Electrolyte Additives with Synergistic Functional Moieties for Silicon Negative Electrode-Based Lithium Ion Batteries: A Case Study on Lactic Acid O-Carboxyanhydride". Chemistry of Materials. 32 (1): 173–185. doi:10.1021/acs.chemmater.9b03173. ISSN 0897-4756. S2CID 213665815. Retrieved 23 January 2022.
- ^ Yang, Xinwei; Liu, Wenqiang; Sun, Jian; Hu, Yingchao; Wang, Wenyu; Chen, Hongqiang; Zhang, Yang; Li, Xian; Xu, Minghou (2016). "Preparation of Novel Li4SiO4 Sorbents with Superior Performance at Low CO2 Concentration". ChemSusChem. 9 (13): 1607–1613. doi:10.1002/cssc.201501699. ISSN 1864-564X. PMID 27312486. Retrieved 23 January 2022.
- ^ Stenina, I. A.; Sobolev, A. N.; Kuz’mina, A. A.; Kulova, T. L.; Yaroslavtsev, A. B. (1 August 2019). "Effect of the Carbon Source on the Electrochemical Properties of Li4Ti5O12/C Composites Prepared by Solid-State Synthesis". Inorganic Materials. 55 (8): 803–809. doi:10.1134/S0020168519080156. ISSN 1608-3172. S2CID 201715779. Retrieved 23 January 2022.
- ^ Handler, J. S. (October 1, 1960). "The role of lactic acid in the reduced excretion of uric acid in toxemia of pregnancy". The Journal of Clinical Investigation. 39 (10): 1526–1532. doi:10.1172/JCI104172. ISSN 0021-9738. PMC 441887. PMID 13711188.
- ^ Wang, Quan; Xu, Xinxiu; Li, Jun; Liu, Jing; Gu, Haifeng; Zhang, Ru; Chen, Jiekai; Kuang, Yin; Fei, Jian; Jiang, Cong; Wang, Ping; Pei, Duanqing; Ding, Sheng; Xie, Xin (July 5, 2011). "Lithium, an anti-psychotic drug, greatly enhances the generation of induced pluripotent stem cells". Cell Research. 21 (10): 1424–1435. doi:10.1038/cr.2011.108. ISSN 1748-7838. PMC 3193456. PMID 21727907.
Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction