| |||

| |||
| Identifiers | |||
|---|---|---|---|
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.155.006 | ||
| EC Number |
| ||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| C7H5FeIO2 | |||
| Molar mass | 303.864 g·mol−1 | ||
| Appearance | Black crystalline solid (density = 2.374 g/cm3) | ||
| Melting point | 119 °C (246 °F; 392 K) | ||
| Structure | |||
| Cs | |||
| Hazards | |||
| GHS labelling: | |||

| |||
| Warning | |||
| H302, H312, H332 | |||
| Safety data sheet (SDS) | Here | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
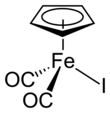
Cyclopentadienyliron dicarbonyl iodide is an organoiron compound with the formula (C5H5)Fe(CO)2I. It is a dark brown solid that is soluble in common organic solvents. (C5H5)Fe(CO)2I, or FpI as it is often known, is an intermediate for the preparation of other organoiron compounds such as in ferraboranes.
Preparation[edit]
Cyclopentadienyliron dicarbonyl iodide is synthesized by the reaction of cyclopentadienyliron dicarbonyl dimer with I2:[1]
- Cp2Fe2(CO)4 + I2 → 2 CpFe(CO)2I
It was first reported by Pauson and Hallam.[2]
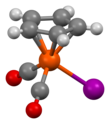
Structure[edit]
The compound has Cs symmetry, with a mirror plane intersecting one carbon of the Cp ring as well as the iron and iodide centre. The compound adopts a piano stool structure: the cyclopentadienyl ligand is the "seat" and three other ligands are "legs". Such compounds are members of the half-sandwich family of compounds, which is a subgroup of the metallocenes. X-ray crystallography shows the following features: Fe-Cp centroid = 1.72, Fe-I = 2.61, and Fe-CO = 1.78 Å.[3]
Electron counting of this iron(II) complex indicates that (C5H5)Fe(CO)2I is an 18-electron complex: using the neutral method there are 8 electrons from the iron, 5 electrons from the cyclopentadienyl anion, 2 electrons from each of the carbonyls, and 1 electron from the iodide.
References[edit]
- ^ King, R. B.; Stone, F. G. A "Cyclopentadienyl Metal Carbonyls and some Derivatives" Inorg. Synth. 1963, volume 7, pp. 99-115. doi:10.1002/9780470132388.ch31
- ^ B. F. Hallam, P. L. Pauson "Ferrocene Derivatives. Part III. Cyclopentadienyliron Carbonyls" J. Chem. Soc. 1956, pp. 3031-3037.
- ^ Zeller, M.; Lazich, E.; Hunter, A. D. (2003). "Dicarbonyl(η5-cyclopentadienyl)iodoiron(II)". Acta Crystallographica Section E. 59 (10): m914–m915. doi:10.1107/S1600536803019950.
Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction