| |

| |
| Names | |
|---|---|
| Other names
Boron Trifluoride Ethyl Ether
Boron Trifluoride Diethyl Etherate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.355 |
PubChem CID
|
|
| UNII | |
| UN number | 2604 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10BF3O | |
| Molar mass | 141.93 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.15 g cm3 |
| Melting point | −58 °C (−72 °F; 215 K) |
| Boiling point | 126 °C (259 °F; 399 K) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Flammable, Reacts with water, Corrosive |
| GHS labelling: | |
    
| |
| Danger | |
| NFPA 704 (fire diamond) | |
| Flash point | 58.5 °C (137.3 °F; 331.6 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
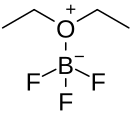
Boron trifluoride etherate, strictly boron trifluoride diethyl etherate, or boron trifluoride–ether complex, is the chemical compound with the formula BF3O(C2H5)2, often abbreviated BF3OEt2. It is a colorless liquid, although older samples can appear brown. The compound is used as a source of boron trifluoride in many chemical reactions that require a Lewis acid.[1] The compound features tetrahedral boron coordinated to a diethylether ligand.[2] Many analogues are known, including the methanol complex.
Reactions[edit]
Boron trifluoride etherate serves as a source of boron trifluoride according to the equilibrium:
- BF3OEt2 BF3 + OEt2
The BF3 binds to even weak Lewis bases, inducing reactions of the resulting adducts with nucleophiles.[1]
References[edit]
- ^ a b Veronica Cornel; Carl J. Lovely (2007). "Boron Trifluoride Etherate". Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons. pp. rb249.pub2. doi:10.1002/047084289X.rb249.pub2. ISBN 978-0-471-93623-7.
- ^ V. V. Saraev; P. B. Kraikivskii; I. Svoboda; A. S. Kuzakov; R. F. Jordan (2008). "Synthesis, Molecular Structure, and EPR Analysis of the Three-Coordinate Ni(I) Complex [Ni(PPh3)3][BF4]". J. Phys. Chem. A. 112 (48): 12449–12455. Bibcode:2008JPCA..11212449S. doi:10.1021/jp802462x. PMID 18991433.


Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction