 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
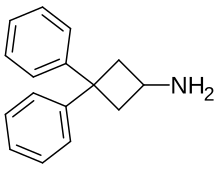
| Formula | C16H17N |
| Molar mass | 223.319 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
3,3,-Diphenylcyclobutanamine is a psychostimulant drug which was originally prepared as an antidepressant in the late 1970s.[1] It appears to inhibit the reuptake of serotonin, norepinephrine, and dopamine, and may also induce their release as well.[1] The N-methyl and N,N-dimethyl analogues of the compound are also known and are more potent.[1] All three agents produce locomotor stimulation in animal studies, with the tertiary amine being the strongest.[1]
Synthesis[edit]
A number of methods were tried in order to construct the strained four-carbon ring. A synthesis of 3,3-diphenylcyclobutanone appeared in the literature.[2] The ketone was prepared in low yield by the reaction of diphenylketene with 2 equiv of diazomethane.[3] The latter synthesis, although low yielding, was used and the desired amines were prepared from 3,3-diphenylcyclobutanone.
Diphenylketene[edit]
Diphenylketene is produced by the elimination of hydrogen chloride from diphenylacetyl chloride in the presence of triethylamine.[4]
See also[edit]
- β-Phenylmethamphetamine
- Fezolamine
- Azetidine ring variation:[5]
References[edit]
- ^ a b c d Carnmalm B, Rämsby S, Renyi AL, Ross SB, Ogren SO (January 1978). "Antidepressant agents. 9. 3,3-Diphenylcyclobutylamines, a new class of central stimulants". Journal of Medicinal Chemistry. 21 (1): 78–82. doi:10.1021/jm00199a014. PMID 22757.
- ^ Wilt JW, Dabek RA, Welzel KC (1972). "Transannular neophyl rearrangement". The Journal of Organic Chemistry. 37 (3): 425–430. doi:10.1021/jo00968a022.
- ^ Michejda CJ, Comnick RW (1975). "Acetolysis of 3,3-disubstituted cyclobutyl tosylates". The Journal of Organic Chemistry. 40 (8): 1046–1050. doi:10.1021/jo00896a010.
- ^ Taylor EC, McKillop A, Hawks GH (1972). Michejda CJ, von Riesen DD, Comnick RW, Baumgarten HE (eds.). "DIPHENYLKETENE [Ethenone, diphenyl-]". Organic Syntheses. 52: 36. doi:10.15227/orgsyn.052.0036.
- ^ Das M, Weissenfluh A, Ly N, Trudell ML (June 2020). "Synthesis of Simple 3,3-Diarylazetidines from N-Boc-3-arylazetidinols Using Friedel-Crafts Arylation Conditions". The Journal of Organic Chemistry. 85 (12): 8209–8213. doi:10.1021/acs.joc.0c00454. PMID 32449343. S2CID 218873364.

Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction