 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | ~1 hour |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.052.157 |
| Chemical and physical data | |
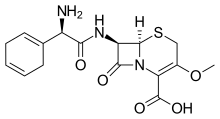
| Formula | C16H19N3O5S |
| Molar mass | 365.40 g·mol−1 |
| 3D model (JSmol) | |
| |
Cefroxadine (INN, trade names Oraspor and Cefthan-DS) is a cephalosporin antibiotic. It is structurally related to cefalexin, and both drugs share a similar spectrum of activity.[1]
It is available in Italy.[2]
Synthesis
[edit]Cefroxadine can be prepared by several routes, including one in which the enol is methylated with diazomethane as a key step. A rather more involved route starts with comparatively readily available phenoxymethylpenicillin sulfoxide benzhydryl ester (1).

This undergoes fragmentation when treated with benzothiazole-2-thiol to give 2. Ozonolysis (reductive work-up) cleaves the olefinic linkage and the unsymmetrical disulfide moiety is converted to a tosyl thioester (3). The enol moiety is methylated with diazomethane, the six-membered ring is closed by reaction with 1,5-diazabicyclo[5.4.0]undec-5-ene (DBU), and the ester protection is removed with trifluoroacetic acid to give 4. The amide side chain is removed by the usual PCl5/dimethylaniline sequence followed by reamidation with the appropriate acid chloride to give cefroxadine (5).
See also
[edit]References
[edit]- ^ Yasuda K, Kurashige S, Mitsuhashi S (July 1980). "Cefroxadine (CGP-9000), an orally active cephalosporin". Antimicrobial Agents and Chemotherapy. 18 (1): 105–10. doi:10.1128/AAC.18.1.105. PMC 283947. PMID 6998373.
- ^ [No authors listed]. "Oraspor". Prontuario.it (in Italian). Elsevier. Retrieved 2010-07-31.
- ^ DE 2331133, Bickel, Hans & Scartazzini, Riccardo, "Enolderivate [Enol derivatives]", published 1974-01-17, assigned to Ciba-Geigy AG
- ^ R. Scartazzini, H. Bickel, U.S. patent 4,073,902 (1978 to Ciba-Geigy).
- ^ R. B. Woodward and H. Bickel, U.S. patent 4,147,864 (1979); Chem. Abstr., 91, 74633J (1979).
Well, that’s interesting to know that Psilotum nudum are known as whisk ferns. Psilotum nudum is the commoner species of the two. While the P. flaccidum is a rare species and is found in the tropical islands. Both the species are usually epiphytic in habit and grow upon tree ferns. These species may also be terrestrial and grow in humus or in the crevices of the rocks.
View the detailed Guide of Psilotum nudum: Detailed Study Of Psilotum Nudum (Whisk Fern), Classification, Anatomy, Reproduction