| Post-traumatic stress disorder | |
|---|---|
| Specialty | Psychiatry, clinical psychology |
| Symptoms | Disturbing thoughts, feelings, or dreams related to the event; mental or physical distress to trauma-related cues; efforts to avoid trauma-related situations; increased fight-or-flight response[1] |
| Complications | Suicide; cardiac, respiratory, musculoskeletal, gastrointestinal, and immunological disorders [2] |
| Duration | > 1 month[a] |
| Causes | Exposure to a traumatic event[1] |
| Diagnostic method | Based on symptoms[2] |
| Treatment | Counseling, medication,[4] MDMA-assisted psychotherapy,[5] selective serotonin reuptake inhibitors[6] |
| Frequency | 8.7% (lifetime risk); 3.5% (12-month risk) (US)[7] |
Post-traumatic stress disorder (PTSD)[b] is a mental and behavioral disorder[8] that develops from experiencing a traumatic event, such as sexual assault, warfare, traffic collisions, child abuse, domestic violence, or other threats on a person's life or well-being.[1][9] Symptoms may include disturbing thoughts, feelings, or dreams related to the events, mental or physical distress to trauma-related cues, attempts to avoid trauma-related cues, alterations in the way a person thinks and feels, and an increase in the fight-or-flight response.[1][4][10] These symptoms last for more than a month after the event.[1] Young children are less likely to show distress, but instead may express their memories through play.[1] A person with PTSD is at a higher risk of suicide and intentional self-harm.[2][11]
Most people who experience traumatic events do not develop PTSD.[2] People who experience interpersonal violence such as rape, other sexual assaults, being kidnapped, stalking, physical abuse by an intimate partner, and childhood abuse are more likely to develop PTSD than those who experience non-assault based trauma, such as accidents and natural disasters.[12][13][14] Those who experience prolonged trauma, such as slavery, concentration camps, or chronic domestic abuse, may develop complex post-traumatic stress disorder (C-PTSD). C-PTSD is similar to PTSD, but has a distinct effect on a person's emotional regulation and core identity.[15]
Prevention may be possible when counselling is targeted at those with early symptoms, but is not effective when provided to all trauma-exposed individuals regardless of whether symptoms are present.[2] The main treatments for people with PTSD are counselling (psychotherapy) and medication.[4][16] Antidepressants of the SSRI or SNRI type are the first-line medications used for PTSD and are moderately beneficial for about half of people.[6] Benefits from medication are less than those seen with counselling.[2] It is not known whether using medications and counselling together has greater benefit than either method separately.[2][17] Medications, other than some SSRIs or SNRIs, do not have enough evidence to support their use and, in the case of benzodiazepines, may worsen outcomes.[18][19]
In the United States, about 3.5% of adults have PTSD in a given year, and 9% of people develop it at some point in their life.[1] In much of the rest of the world, rates during a given year are between 0.5% and 1%.[1] Higher rates may occur in regions of armed conflict.[2] It is more common in women than men.[4]
Symptoms of trauma-related mental disorders have been documented since at least the time of the ancient Greeks.[20] A few instances of evidence of post-traumatic illness have been argued to exist from the seventeenth and eighteenth centuries, such as the diary of Samuel Pepys, who described intrusive and distressing symptoms following the 1666 Fire of London.[21] During the world wars, the condition was known under various terms, including 'shell shock', 'war nerves', neurasthenia and 'combat neurosis'.[22][23] The term "post-traumatic stress disorder" came into use in the 1970s, in large part due to the diagnoses of U.S. military veterans of the Vietnam War.[24] It was officially recognized by the American Psychiatric Association in 1980 in the third edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-III).[25]
Symptoms

Symptoms of PTSD generally begin within the first three months after the inciting traumatic event, but may not begin until years later.[1][4] In the typical case, the individual with PTSD persistently avoids either trauma-related thoughts and emotions or discussion of the traumatic event and may even have amnesia of the event (Dissociative amnesia).[1] However, the event is commonly relived by the individual through intrusive, recurrent recollections, dissociative episodes of reliving the trauma ("flashbacks"), and nightmares (50 to 70%).[26][27][28] While it is common to have symptoms after any traumatic event, these must persist to a sufficient degree (i.e., causing dysfunction in life or clinical levels of distress) for longer than one month after the trauma to be classified as PTSD (clinically significant dysfunction or distress for less than one month after the trauma may be acute stress disorder).[1][29][30][31] Some following a traumatic event experience post-traumatic growth.[32]
Associated medical conditions
Trauma survivors often develop depression, anxiety disorders, and mood disorders in addition to PTSD.[33] More than 50% of those with PTSD have co-morbid anxiety, mood or substance use disorders.[34]
Substance use disorder, such as alcohol use disorder, commonly co-occur with PTSD.[35] Recovery from post-traumatic stress disorder or other anxiety disorders may be hindered, or the condition worsened, when substance use disorders are comorbid with PTSD. Resolving these problems can bring about improvement in an individual's mental health status and anxiety levels.[36][37]
PTSD has a strong association with tinnitus,[38] and can even possibly be the tinnitus' cause.[39]
In children and adolescents, there is a strong association between emotional regulation difficulties (e.g. mood swings, anger outbursts, temper tantrums) and post-traumatic stress symptoms, independent of age, gender, or type of trauma.[40]
Moral injury the feeling of moral distress such as a shame or guilt following a moral transgression is associated with PTSD but is distinguished from it. Moral injury is associated with shame and guilt, while PTSD is associated with anxiety and fear.[41]: 2,8,11
In a population based study examining veterans of the Vietnam War, the presence of PTSD and exposure to high level stressors on the battlefield were associated with a 2-fold increased risk of death, with the leading causes of death being ischemic heart disease or cancers of the respiratory tract including lung cancer.[34][42]
Risk factors

Persons considered at risk for developing PTSD include combat military personnel, survivors of natural disasters, concentration camp survivors, and survivors of violent crime. Persons employed in occupations that expose them to violence (such as soldiers) or disasters (such as emergency service workers) are also at risk.[44] Other occupations at an increased risk include police officers, firefighters, ambulance personnel, health care professionals, train drivers, divers, journalists, and sailors, as well as people who work at banks, post offices or in stores.[45] The intensity of the traumatic event is also associated with a subsequent risk of developing PTSD, with experiences related to witnessed death, or witnessed or experienced torture, injury, bodily disfigurement, traumatic brain injury being highly associated with the development of PTSD. Similarly, experiences that are unexpected or in which the victim cannot escape are also associated with a high risk of developing PTSD.[34]
Trauma
PTSD has been associated with a wide range of traumatic events. The risk of developing PTSD after a traumatic event varies by trauma type[46][47] and is the highest following exposure to sexual violence (11.4%), particularly rape (19.0%).[48] Men are more likely to experience a traumatic event (of any type), but women are more likely to experience the kind of high-impact traumatic event that can lead to PTSD, such as interpersonal violence and sexual assault.[49]
Motor vehicle collision survivors, both children and adults, are at an increased risk of PTSD.[50][51] Globally, about 2.6% of adults are diagnosed with PTSD following a non-life-threatening traffic accident, and a similar proportion of children develop PTSD.[48] Risk of PTSD almost doubles to 4.6% for life-threatening auto accidents.[48] Females were more likely to be diagnosed with PTSD following a road traffic accident, whether the accident occurred during childhood or adulthood.[50][51]
Post-traumatic stress reactions have been studied in children and adolescents.[52] The rate of PTSD might be lower in children than adults, but in the absence of therapy, symptoms may continue for decades.[53] One estimate suggests that the proportion of children and adolescents having PTSD in a non-wartorn population in a developed country may be 1% compared to 1.5% to 3% of adults.[53] On average, 16% of children exposed to a traumatic event develop PTSD, with the incidence varying according to type of exposure and gender.[54] Similar to the adult population, risk factors for PTSD in children include: female gender, exposure to disasters (natural or man-made), negative coping behaviors, and/or lacking proper social support systems.[55]
Predictor models have consistently found that childhood trauma, chronic adversity, neurobiological differences, and familial stressors are associated with risk for PTSD after a traumatic event in adulthood.[56][57][58] It has been difficult to find consistently aspects of the events that predict, but peritraumatic dissociation has been a fairly consistent predictive indicator of the development of PTSD.[59] Proximity to, duration of, and severity of the trauma make an impact. It has been speculated that interpersonal traumas cause more problems than impersonal ones,[60] but this is controversial.[61] The risk of developing PTSD is increased in individuals who are exposed to physical abuse, physical assault, or kidnapping.[62][63] Women who experience physical violence are more likely to develop PTSD than men.[62]
Intimate partner violence
An individual that has been exposed to domestic violence is predisposed to the development of PTSD. There is a strong association between the development of PTSD in mothers that experienced domestic violence during the perinatal period of their pregnancy.[64]
Those who have experienced sexual assault or rape may develop symptoms of PTSD.[65][66] The likelihood of sustained symptoms of PTSD is higher if the rapist confined or restrained the person, if the person being raped believed the rapist would kill them, the person who was raped was very young or very old, and if the rapist was someone they knew. The likelihood of sustained severe symptoms is also higher if people around the survivor ignore (or are ignorant of) the rape or blame the rape survivor.[67]
Military service in combat is a risk factor for developing PTSD.[34] Around 78% of people exposed to combat do not develop PTSD; in about 25% of military personnel who develop PTSD, its appearance is delayed.[34]
Refugees are also at an increased risk for PTSD due to their exposure to war, hardships, and traumatic events. The rates for PTSD within refugee populations range from 4% to 86%.[68] While the stresses of war affect everyone involved, displaced persons have been shown to be more so than others.[69]
Challenges related to the overall psychosocial well-being of refugees are complex and individually nuanced. Refugees have reduced levels of well-being and a high rate of mental distress due to past and ongoing trauma. Groups that are particularly affected and whose needs often remain unmet are women, older people and unaccompanied minors.[70] Post-traumatic stress and depression in refugee populations also tend to affect their educational success.[70]
Unexpected death of a loved one
Sudden, unexpected death of a loved one is the most common traumatic event type reported in cross-national studies.[48][71] However, the majority of people who experience this type of event will not develop PTSD. An analysis from the WHO World Mental Health Surveys found a 5.2% risk of developing PTSD after learning of the unexpected death of a loved one.[71] Because of the high prevalence of this type of traumatic event, unexpected death of a loved one accounts for approximately 20% of PTSD cases worldwide.[48]
Life-threatening illness
Medical conditions associated with an increased risk of PTSD include cancer,[72][73][74] heart attack,[75] and stroke.[76] 22% of cancer survivors present with lifelong PTSD like symptoms.[77] Intensive-care unit (ICU) hospitalization is also a risk factor for PTSD.[78] Some women experience PTSD from their experiences related to breast cancer and mastectomy.[79][80][72] Loved ones of those who experience life-threatening illnesses are also at risk for developing PTSD, such as parents of a child with chronic illnesses.[81]
Research exists which demonstrates that survivors of psychotic episodes, which exist in diseases such as schizophrenia, schizoaffective disorder, bipolar I disorder, and others, are at greater risk for PTSD due to the experiences one may have during and after psychosis. Such traumatic experiences include, but are not limited to, the treatment patients experience in psychiatric hospitals, police interactions due to psychotic behavior, suicidal behavior and attempts, social stigma and embarrassment due to behavior while in psychosis, frequent terrifying experiences due to psychosis, and the fear of losing control or actual loss of control. The incidence of PTSD in survivors of psychosis may be as low as 11% and as high at 67%.[82][83][84]
Women who experience miscarriage are at risk of PTSD.[85][86][87] Those who experience subsequent miscarriages have an increased risk of PTSD compared to those experiencing only one.[85] PTSD can also occur after childbirth and the risk increases if a woman has experienced trauma prior to the pregnancy.[88][89] Prevalence of PTSD following normal childbirth (that is, excluding stillbirth or major complications) is estimated to be between 2.8 and 5.6% at six weeks postpartum,[90] with rates dropping to 1.5% at six months postpartum.[90][91] Symptoms of PTSD are common following childbirth, with prevalence of 24–30.1%[90] at six weeks, dropping to 13.6% at six months.[92] Emergency childbirth is also associated with PTSD.[93]
Natural disasters
Genetics
There is evidence that susceptibility to PTSD is hereditary. Approximately 30% of the variance in PTSD is caused from genetics alone.[59] For twin pairs exposed to combat in Vietnam, having a monozygotic (identical) twin with PTSD was associated with an increased risk of the co-twin's having PTSD compared to twins that were dizygotic (non-identical twins).[94] Women with a smaller hippocampus might be more likely to develop PTSD following a traumatic event based on preliminary findings.[95] Research has also found that PTSD shares many genetic influences common to other psychiatric disorders. Panic and generalized anxiety disorders and PTSD share 60% of the same genetic variance. Alcohol, nicotine, and drug dependence share greater than 40% genetic similarities.[59]
Several biological indicators have been identified that are related to later PTSD development. Heightened startle responses and, with only preliminary results, a smaller hippocampal volume have been identified as possible biomarkers for heightened risk of developing PTSD.[96]
Pathophysiology
Neuroendocrinology
PTSD symptoms may result when a traumatic event causes an over-reactive adrenaline response, which creates deep neurological patterns in the brain. These patterns can persist long after the event that triggered the fear, making an individual hyper-responsive to future fearful situations.[29][97] During traumatic experiences, the high levels of stress hormones secreted suppress hypothalamic activity that may be a major factor toward the development of PTSD.[98]
PTSD causes biochemical changes in the brain and body, that differ from other psychiatric disorders such as major depression. Individuals diagnosed with PTSD respond more strongly to a dexamethasone suppression test than individuals diagnosed with clinical depression.[99][100]
Most people with PTSD show a low secretion of cortisol and high secretion of catecholamines in urine,[101] with a norepinephrine/cortisol ratio consequently higher than comparable non-diagnosed individuals.[102] This is in contrast to the normative fight-or-flight response, in which both catecholamine and cortisol levels are elevated after exposure to a stressor.[103]
Brain catecholamine levels are high,[104] and corticotropin-releasing factor (CRF) concentrations are high.[105][106] Together, these findings suggest abnormality in the hypothalamic-pituitary-adrenal (HPA) axis.
The maintenance of fear has been shown to include the HPA axis, the locus coeruleus-noradrenergic systems, and the connections between the limbic system and frontal cortex. The HPA axis that coordinates the hormonal response to stress,[107] which activates the LC-noradrenergic system, is implicated in the over-consolidation of memories that occurs in the aftermath of trauma.[108] This over-consolidation increases the likelihood of one's developing PTSD. The amygdala is responsible for threat detection and the conditioned and unconditioned fear responses that are carried out as a response to a threat.[59]
The HPA axis is responsible for coordinating the hormonal response to stress.[59] Given the strong cortisol suppression to dexamethasone in PTSD, HPA axis abnormalities are likely predicated on strong negative feedback inhibition of cortisol, itself likely due to an increased sensitivity of glucocorticoid receptors.[109]
PTSD has been hypothesized to be a maladaptive learning pathway to fear response through a hypersensitive, hyperreactive, and hyperresponsive HPA axis.[110]
Low cortisol levels may predispose individuals to PTSD: Following war trauma, Swedish soldiers serving in Bosnia and Herzegovina with low pre-service salivary cortisol levels had a higher risk of reacting with PTSD symptoms, following war trauma, than soldiers with normal pre-service levels.[111] Because cortisol is normally important in restoring homeostasis after the stress response, it is thought that trauma survivors with low cortisol experience a poorly contained—that is, longer and more distressing—response, setting the stage for PTSD.
It is thought that the locus coeruleus-noradrenergic system mediates the over-consolidation of fear memory. High levels of cortisol reduce noradrenergic activity, and because people with PTSD tend to have reduced levels of cortisol, it has been proposed that individuals with PTSD cannot regulate the increased noradrenergic response to traumatic stress.[98] Intrusive memories and conditioned fear responses are thought to be a result of the response to associated triggers. Neuropeptide Y (NPY) has been reported to reduce the release of norepinephrine and has been demonstrated to have anxiolytic properties in animal models. Studies have shown people with PTSD demonstrate reduced levels of NPY, possibly indicating their increased anxiety levels.[59]
Other studies indicate that people with PTSD have chronically low levels of serotonin, which contributes to the commonly associated behavioral symptoms such as anxiety, ruminations, irritability, aggression, suicidality, and impulsivity.[112] Serotonin also contributes to the stabilization of glucocorticoid production.
Dopamine levels in a person with PTSD can contribute to symptoms: low levels can contribute to anhedonia, apathy, impaired attention, and motor deficits; high levels can contribute to psychosis, agitation, and restlessness.[112]
Several studies described elevated concentrations of the thyroid hormone triiodothyronine in PTSD.[113] This kind of type 2 allostatic adaptation may contribute to increased sensitivity to catecholamines and other stress mediators.
Hyperresponsiveness in the norepinephrine system can also be caused by continued exposure to high stress. Overactivation of norepinephrine receptors in the prefrontal cortex can be connected to the flashbacks and nightmares frequently experienced by those with PTSD. A decrease in other norepinephrine functions (awareness of the current environment) prevents the memory mechanisms in the brain from processing the experience, and emotions the person is experiencing during a flashback are not associated with the current environment.[112]
There is considerable controversy within the medical community regarding the neurobiology of PTSD. A 2012 review showed no clear relationship between cortisol levels and PTSD. The majority of reports indicate people with PTSD have elevated levels of corticotropin-releasing hormone, lower basal cortisol levels, and enhanced negative feedback suppression of the HPA axis by dexamethasone.[59][114]
Neuroanatomy
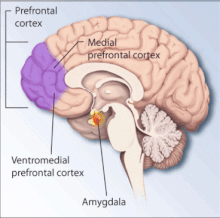
A meta-analysis of structural MRI studies found an association with reduced total brain volume, intracranial volume, and volumes of the hippocampus, insula cortex, and anterior cingulate.[116] Much of this research stems from PTSD in those exposed to the Vietnam War.[117][118]
People with PTSD have decreased brain activity in the dorsal and rostral anterior cingulate cortices and the ventromedial prefrontal cortex, areas linked to the experience and regulation of emotion.[119]
The amygdala is strongly involved in forming emotional memories, especially fear-related memories. During high stress, the hippocampus, which is associated with placing memories in the correct context of space and time and memory recall, is suppressed. According to one theory, this suppression may be the cause of the flashbacks that can affect people with PTSD. When someone with PTSD undergoes stimuli similar to the traumatic event, the body perceives the event as occurring again because the memory was never properly recorded in the person's memory.[59][120]
The amygdalocentric model of PTSD proposes that the amygdala is very much aroused and insufficiently controlled by the medial prefrontal cortex and the hippocampus, in particular during extinction.[121] This is consistent with an interpretation of PTSD as a syndrome of deficient extinction ability.[121][122]
The basolateral nucleus (BLA) of the amygdala is responsible for the comparison and development of associations between unconditioned and conditioned responses to stimuli, which results in the fear conditioning present in PTSD. The BLA activates the central nucleus (CeA) of the amygdala, which elaborates the fear response, (including behavioral response to threat and elevated startle response). Descending inhibitory inputs from the medial prefrontal cortex (mPFC) regulate the transmission from the BLA to the CeA, which is hypothesized to play a role in the extinction of conditioned fear responses.[59]
While as a whole, amygdala hyperactivity is reported by meta analysis of functional neuroimaging in PTSD, there is a large degree of heterogeniety, more so than in social anxiety disorder or phobic disorder. Comparing dorsal (roughly the CeA) and ventral (roughly the BLA) clusters, hyperactivity is more robust in the ventral cluster, while hypoactivity is evident in the dorsal cluster. The distinction may explain the blunted emotions in PTSD (via desensitization in the CeA) as well as the fear related component.[123]
In a 2007 study, Vietnam War combat veterans with PTSD showed a 20% reduction in the volume of their hippocampus compared with veterans who did not have such symptoms.[124] This finding was not replicated in chronic PTSD patients traumatized at an air show plane crash in 1988 (Ramstein, Germany).[125]
Evidence suggests that endogenous cannabinoid levels are reduced in PTSD, particularly anandamide, and that cannabinoid receptors (CB1) are increased in order to compensate.[126] There appears to be a link between increased CB1 receptor availability in the amygdala and abnormal threat processing and hyperarousal, but not dysphoria, in trauma survivors.
A 2020 study found no evidence for conclusions from prior research that suggested low IQ is a risk factor for developing PTSD.[127]
Diagnosis
PTSD can be difficult to diagnose, because of:
- the subjective nature of most of the diagnostic criteria (although this is true for many mental disorders);
- the potential for over-reporting, e.g., while seeking disability benefits, or when PTSD could be a mitigating factor at criminal sentencing[128]
- the potential for under-reporting, e.g., stigma, pride, fear that a PTSD diagnosis might preclude certain employment opportunities;
- symptom overlap with other mental disorders such as obsessive compulsive disorder and generalized anxiety disorder;[129]
- association with other mental disorders such as major depressive disorder and generalized anxiety disorder;
- substance use disorders, which often produce some of the same signs and symptoms as PTSD; and
- substance use disorders can increase vulnerability to PTSD or exacerbate PTSD symptoms or both; and
- PTSD increases the risk for developing substance use disorders.[130]
- the differential expression of symptoms culturally (specifically with respect to avoidance and numbing symptoms, distressing dreams, and somatic symptoms)[131]
Screening
There are a number of PTSD screening instruments for adults, such as the PTSD Checklist for DSM-5 (PCL-5)[132][133] and the Primary Care PTSD Screen for DSM-5 (PC-PTSD-5).[134] The 17 item PTSD checklist is also capable of monitoring the severity of symptoms and the response to treatment.[34]
There are also several screening and assessment instruments for use with children and adolescents. These include the Child PTSD Symptom Scale (CPSS),[135][136] Child Trauma Screening Questionnaire,[137][138] and UCLA Post-traumatic Stress Disorder Reaction Index for DSM-IV.[139][140]
In addition, there are also screening and assessment instruments for caregivers of very young children (six years of age and younger). These include the Young Child PTSD Screen,[141] the Young Child PTSD Checklist,[141] and the Diagnostic Infant and Preschool Assessment.[142]
Assessment
Evidence-based assessment principles, including a multimethod assessment approach, form the foundation of PTSD assessment.[143][144][145]: 25 Those who conduct assessments for PTSD may use various clinician-administered interviews and instruments to provide an official PTSD diagnosis.[146] Some commonly used, reliable, and valid assessment instruments for PTSD diagnosis, in accordance with the DSM-5, include the Clinician-Administered PTSD Scale for the DSM-5 (CAPS-5), PTSD Symptom Scale Interview (PSS-I-5), and Structured Clinical Interview for DSM-5 – PTSD Module (SCID-5 PTSD Module).[147][148][149][150]
Diagnostic and statistical manual
PTSD was classified as an anxiety disorder in the DSM-IV, but has since been reclassified as a "trauma- and stressor-related disorder" in the DSM-5.[1] The DSM-5 diagnostic criteria for PTSD include four symptom clusters: re-experiencing, avoidance, negative alterations in cognition/mood, and alterations in arousal and reactivity.[1][4]
International classification of diseases
The International Classification of Diseases and Related Health Problems 10 (ICD-10) classifies PTSD under "Reaction to severe stress, and adjustment disorders."[151] The ICD-10 criteria for PTSD include re-experiencing, avoidance, and either increased reactivity or inability to recall certain details related to the event.[151]
The ICD-11 diagnostic description for PTSD contains three components or symptom groups (1) re-experiencing, (2) avoidance, and (3) heightened sense of threat.[152][153] ICD-11 no longer includes verbal thoughts about the traumatic event as a symptom.[153] There is a predicted lower rate of diagnosed PTSD using ICD-11 compared to ICD10 or DSM-5.[153] ICD-11 also proposes identifying a distinct group with complex post-traumatic stress disorder (CPTSD), who have more often experienced several or sustained traumas and have greater functional impairment than those with PTSD.[153]
Differential diagnosis
A diagnosis of PTSD requires that the person has been exposed to an extreme stressor. Any stressor can result in a diagnosis of adjustment disorder and it is an appropriate diagnosis for a stressor and a symptom pattern that does not meet the criteria for PTSD.
The symptom pattern for acute stress disorder must occur and be resolved within four weeks of the trauma. If it lasts longer, and the symptom pattern fits that characteristic of PTSD, the diagnosis may be changed.[27]
Obsessive compulsive disorder may be diagnosed for intrusive thoughts that are recurring but not related to a specific traumatic event.[27]
In extreme cases of prolonged, repeated traumatization where there is no viable chance of escape, survivors may develop complex post-traumatic stress disorder.[154] This occurs as a result of layers of trauma rather than a single traumatic event, and includes additional symptomatology, such as the loss of a coherent sense of self.[155]
Prevention
Modest benefits have been seen from early access to cognitive behavioral therapy. Critical incident stress management has been suggested as a means of preventing PTSD, but subsequent studies suggest the likelihood of its producing negative outcomes.[156][157] A 2019 Cochrane review did not find any evidence to support the use of an intervention offered to everyone, and that "multiple session interventions may result in worse outcome than no intervention for some individuals."[158] The World Health Organization recommends against the use of benzodiazepines and antidepressants in for acute stress (symptoms lasting less than one month).[159] Some evidence supports the use of hydrocortisone for prevention in adults, although there is limited or no evidence supporting propranolol, escitalopram, temazepam, or gabapentin.[160][161]
Psychological debriefing
Trauma-exposed individuals often receive treatment called psychological debriefing in an effort to prevent PTSD, which consists of interviews that are meant to allow individuals to directly confront the event and share their feelings with the counselor and to help structure their memories of the event.[162] However, several meta-analyses find that psychological debriefing is unhelpful, is potentially harmful and does not reduce the future risk of developing PTSD.[34][162][163][164] This is true for both single-session debriefing and multiple session interventions.[158] As of 2017 the American Psychological Association assessed psychological debriefing as No Research Support/Treatment is Potentially Harmful.[165]
Early intervention
Trauma focused intervention delivered within days or weeks of the potentially traumatic event has been found to decrease PTSD symptoms.[166] Similar to psychological debriefing, the goal of early intervention is to lessen the intensity and frequency of stress symptoms, with the aim of preventing new-onset or relapsed mental disorders and further distress later in the healing process.[167]
Risk-targeted interventions
Risk-targeted interventions are those that attempt to mitigate specific formative information or events. It can target modeling normal behaviors, instruction on a task, or giving information on the event.[168][169]
Management
Reviews of studies have found that combination therapy (psychological and pharmacotherapy) is no more effective than psychological therapy alone.[17]
Counselling
The approaches with the strongest evidence include behavioral and cognitive-behavioral therapies such as prolonged exposure therapy,[170] cognitive processing therapy, and eye movement desensitization and reprocessing (EMDR).[171][172][173] There is some evidence for brief eclectic psychotherapy (BEP), narrative exposure therapy (NET), and written exposure therapy.[174][175]
A 2019 Cochrane review evaluated couples and family therapies compared to no care and individual and group therapies for the treatment of PTSD.[176] There were too few studies on couples therapies to determine if substantive benefits were derived, but preliminary RCTs suggested that couples therapies may be beneficial for reducing PTSD symptoms.[176]
A meta-analytic comparison of EMDR and cognitive behavioral therapy (CBT) found both protocols indistinguishable in terms of effectiveness in treating PTSD; however, "the contribution of the eye movement component in EMDR to treatment outcome" is unclear.[177] A meta-analysis in children and adolescents also found that EMDR was as efficacious as CBT.[178]
Children with PTSD are far more likely to pursue treatment at school (because of its proximity and ease) than at a free clinic.[179]
Cognitive behavioral therapy

CBT seeks to change the way a person feels and acts by changing the patterns of thinking or behavior, or both, responsible for negative emotions. Results from a 2018 systematic review found high strength of evidence that supports CBT-exposure therapy efficacious for a reduction in PTSD and depression symptoms, as well as the loss of PTSD diagnosis.[180] CBT has been proven to be an effective treatment for PTSD and is currently considered the standard of care for PTSD by the United States Department of Defense.[181][182]
In CBT, individuals learn to identify thoughts that make them feel afraid or upset and replace them with less distressing thoughts. The goal is to understand how certain thoughts about events cause PTSD-related stress.[183][184] A study assessing an online version of CBT for people with mild-to-moderate PTSD found that the online approach was as effective as, and cheaper than, the same therapy given face-to-face.[185][186] A 2021 Cochrane review assessed the provision of CBT in an Internet-based format found similar beneficial effects for Internet-based therapy as in face-to-face. However, the quality of the evidence was low due to the small number of trials reviewed.[187]
Exposure therapy is a type of cognitive behavioral therapy[188] that involves assisting trauma survivors to re-experience distressing trauma-related memories and reminders in order to facilitate habituation and successful emotional processing of the trauma memory. Most exposure therapy programs include both imaginal confrontation with the traumatic memories and real-life exposure to trauma reminders; this type of CBT has shown benefit in the treatment of PTSD.[189][34]
Some organizations[which?] have endorsed the need for exposure.[190][191] The U.S. Department of Veterans Affairs has been actively training mental health treatment staff in prolonged exposure therapy[192] and Cognitive Processing Therapy[193] in an effort to better treat U.S. veterans with PTSD.
Recent research on contextually based third-generation behavior therapies suggests that they may produce results comparable to some of the better validated therapies.[194] Many of these therapy methods have a significant element of exposure[195] and have demonstrated success in treating the primary problems of PTSD and co-occurring depressive symptoms.[196]
Eye movement desensitization and reprocessing
Eye movement desensitization and reprocessing (EMDR) is a form of psychotherapy developed and studied by Francine Shapiro.[197] She had noticed that, when she was thinking about disturbing memories herself, her eyes were moving rapidly. When she brought her eye movements under control while thinking, the thoughts were less distressing.[197]
In 2002, Shapiro and Maxfield published a theory of why this might work, called adaptive information processing.[198] This theory proposes that eye movement can be used to facilitate emotional processing of memories, changing the person's memory to attend to more adaptive information.[199] The therapist initiates voluntary rapid eye movements while the person focuses on memories, feelings or thoughts about a particular trauma.[53][200] The therapist uses hand movements to get the person to move their eyes backward and forward, but hand-tapping or tones can also be used.[53] EMDR closely resembles cognitive behavior therapy as it combines exposure (re-visiting the traumatic event), working on cognitive processes and relaxation/self-monitoring.[53] However, exposure by way of being asked to think about the experience rather than talk about it has been highlighted as one of the more important distinguishing elements of EMDR.[201]
There have been several small, controlled trials of four to eight weeks of EMDR in adults[202] as well as children and adolescents.[200] There is moderate strength of evidence to support the efficacy of EMDR "for reduction in PTSD symptoms, loss of diagnosis, and reduction in depressive symptoms" according to a 2018 systematic review update.[180] EMDR reduced PTSD symptoms enough in the short term that one in two adults no longer met the criteria for PTSD, but the number of people involved in these trials was small and thus results should be interpreted with caution pending further research.[202] There was not enough evidence to know whether EMDR could eliminate PTSD in adults.[202]
In children and adolescents, a recent meta-analysis of randomized controlled trials using MetaNSUE to avoid biases related to missing information found that EMDR was at least as efficacious as CBT, and superior to waitlist or placebo.[178] There was some evidence that EMDR might prevent depression.[202] There were no studies comparing EMDR to other psychological treatments or to medication.[202] Adverse effects were largely unstudied.[202] The benefits were greater for women with a history of sexual assault compared with people who had experienced other types of traumatizing events (such as accidents, physical assaults and war). There is a small amount of evidence that EMDR may improve re-experiencing symptoms in children and adolescents, but EMDR has not been shown to improve other PTSD symptoms, anxiety, or depression.[200]
The eye movement component of the therapy may not be critical for benefit.[53][199] As there has been no major, high quality randomized trial of EMDR with eye movements versus EMDR without eye movements, the controversy over effectiveness is likely to continue.[201] Authors of a meta-analysis published in 2013[172] stated, "We found that people treated with eye movement therapy had greater improvement in their symptoms of post-traumatic stress disorder than people given therapy without eye movements.... Secondly, we found that in laboratory studies the evidence concludes that thinking of upsetting memories and simultaneously doing a task that facilitates eye movements reduces the vividness and distress associated with the upsetting memories."[203]
Interpersonal psychotherapy
Other approaches, in particular involving social supports,[204][205] may also be important. An open trial of interpersonal psychotherapy[206] reported high rates of remission from PTSD symptoms without using exposure.[207]
Medication
While many medications do not have enough evidence to support their use, four (sertraline, fluoxetine, paroxetine, and venlafaxine) have been shown to have a small to modest benefit over placebo.[19] With many medications, residual PTSD symptoms following treatment is the rule rather than the exception.[208]
Antidepressants
Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) may have some benefit for PTSD symptoms.[19][209][210] Tricyclic antidepressants are equally effective, but are less well tolerated.[211] Evidence provides support for a small or modest improvement with sertraline, fluoxetine, paroxetine, and venlafaxine.[19][212] Thus, these four medications are considered to be first-line medications for PTSD.[209][6] The SSRIs paroxetine and sertraline are FDA approved for the treatment of PTSD.[34]
Benzodiazepines
Benzodiazepines are not recommended for the treatment of PTSD due to a lack of evidence of benefit and risk of worsening PTSD symptoms.[213] Some authors believe that the use of benzodiazepines is contraindicated for acute stress, as this group of drugs can cause dissociation.[214] Nevertheless, some use benzodiazepines with caution for short-term anxiety and insomnia.[215][216][217] While benzodiazepines can alleviate acute anxiety, there is no consistent evidence that they can stop the development of PTSD and may actually increase the risk of developing PTSD 2–5 times.[18] Benzodiazepines should not be used in the immediate aftermath of a traumatic event as they may increase symptoms related to PTSD.[34]
Benzodiazepines may reduce the effectiveness of psychotherapeutic interventions, and there is some evidence that benzodiazepines may actually contribute to the development and chronification of PTSD. For those who already have PTSD, benzodiazepines may worsen and prolong the course of illness, by worsening psychotherapy outcomes, and causing or exacerbating aggression, depression (including suicidality), and substance use.[18] Drawbacks include the risk of developing a benzodiazepine dependence, tolerance (i.e., short-term benefits wearing off with time), and withdrawal syndrome; additionally, individuals with PTSD (even those without a history of alcohol or drug misuse) are at an increased risk of abusing benzodiazepines.[6][218]
Due to a number of other treatments with greater efficacy for PTSD and fewer risks, benzodiazepines should be considered relatively contraindicated until all other treatment options are exhausted.[16][219]
Benzodiazepines also carry a risk of disinhibition (associated with suicidality, aggression and crimes) and their use may delay or inhibit more definitive treatments for PTSD.[6][219][220]
Prazosin
Prazosin, an alpha-1 adrenergic antagonist, has been used in veterans with PTSD to reduce nightmares. Studies show variability in the symptom improvement, appropriate dosages, and efficacy in this population.[221][222][26]
Glucocorticoids
Glucocorticoids may be useful for short-term therapy to protect against neurodegeneration caused by the extended stress response that characterizes PTSD, but long-term use may actually promote neurodegeneration.[223]
Cannabinoids
Cannabis is not recommended as a treatment for PTSD because scientific evidence does not currently exist demonstrating treatment efficacy for cannabinoids.[224][225][c] However, use of cannabis or derived products is widespread among U.S. veterans with PTSD.[226]
The cannabinoid nabilone is sometimes used for nightmares in PTSD. Although some short-term benefit was shown, adverse effects are common and it has not been adequately studied to determine efficacy.[227] An increasing number of states permit and have legalized the use of medical cannabis for the treatment of PTSD.[228]
Other
Exercise, sport and physical activity
Physical activity can influence people's psychological[229] and physical health.[230] The U.S. National Center for PTSD recommends moderate exercise as a way to distract from disturbing emotions, build self-esteem and increase feelings of being in control again. They recommend a discussion with a doctor before starting an exercise program.[231]
Play therapy for children
Play is thought to help children link their inner thoughts with their outer world, connecting real experiences with abstract thought.[232] Repetitive play can also be one way a child relives traumatic events, and that can be a symptom of trauma in a child or young person.[233] Although it is commonly used, there have not been enough studies comparing outcomes in groups of children receiving and not receiving play therapy, so the effects of play therapy are not yet understood.[53][232]
Military programs
Many veterans of the wars in Iraq and Afghanistan have faced significant physical, emotional, and relational disruptions. In response, the United States Marine Corps has instituted programs to assist them in re-adjusting to civilian life, especially in their relationships with spouses and loved ones, to help them communicate better and understand what the other has gone through.[234] Walter Reed Army Institute of Research (WRAIR) developed the Battlemind program to assist service members avoid or ameliorate PTSD and related problems. Wounded Warrior Project partnered with the US Department of Veterans Affairs to create Warrior Care Network, a national health system of PTSD treatment centers.[235][236]
Nightmares
In 2020, the United States Food and Drug Administration granted marketing approval for an Apple Watch app call NightWare. The app aims to improve sleep for people suffering from PTSD-related nightmares, by vibrating when it detects a nightmare in progress based on monitoring heart rate and body movement.[237]
The "colour cure"
Towards the end of the First World War art connoisseur Howard Kemp Prossor came up with what he called the "colour cure" – the use of specific colours to ease the suffering of people with shell shock.
Epidemiology

There is debate over the rates of PTSD found in populations, but, despite changes in diagnosis and the criteria used to define PTSD between 1997 and 2013, epidemiological rates have not changed significantly.[239][240] Most of the current reliable data regarding the epidemiology of PTSD is based on DSM-IV criteria, as the DSM-5 was not introduced until 2013.
The United Nations' World Health Organization publishes estimates of PTSD impact for each of its member states; the latest data available are for 2004. Considering only the 25 most populated countries ranked by overall age-standardized Disability-Adjusted Life Year (DALY) rate, the top half of the ranked list is dominated by Asian/Pacific countries, the US, and Egypt.[238] Ranking the countries by the male-only or female-only rates produces much the same result, but with less meaningfulness, as the score range in the single-sex rankings is much-reduced (4 for women, 3 for men, as compared with 14 for the overall score range), suggesting that the differences between female and male rates, within each country, is what drives the distinctions between the countries.[241][242]
As of 2017, the cross-national lifetime prevalence of PTSD was 3.9%, based on a survey where 5.6% had been exposed to trauma.[243] The primary factor impacting treatment-seeking behavior, which can help to mitigate PTSD development after trauma was income, while being younger, female, and having less social status (less education, lower individual income, and being unemployed) were all factors associated with less treatment-seeking behavior.[243]
| Region | Country | PTSD DALY rate, overall[238] |
PTSD DALY rate, females[241] |
PTSD DALY rate, males[242] |
|---|---|---|---|---|
| Asia / Pacific | Thailand | 59 | 86 | 30 |
| Asia / Pacific | Indonesia | 58 | 86 | 30 |
| Asia / Pacific | Philippines | 58 | 86 | 30 |
| Americas | USA | 58 | 86 | 30 |
| Asia / Pacific | Bangladesh | 57 | 85 | 29 |
| Africa | Egypt | 56 | 83 | 30 |
| Asia / Pacific | India | 56 | 85 | 29 |
| Asia / Pacific | Iran | 56 | 83 | 30 |
| Asia / Pacific | Pakistan | 56 | 85 | 29 |
| Asia / Pacific | Japan | 55 | 80 | 31 |
| Asia / Pacific | Myanmar | 55 | 81 | 30 |
| Europe | Turkey | 55 | 81 | 30 |
| Asia / Pacific | Vietnam | 55 | 80 | 30 |
| Europe | France | 54 | 80 | 28 |
| Europe | Germany | 54 | 80 | 28 |
| Europe | Italy | 54 | 80 | 28 |
| Asia / Pacific | Russian Federation | 54 | 78 | 30 |
| Europe | United Kingdom | 54 | 80 | 28 |
| Africa | Nigeria | 53 | 76 | 29 |
| Africa | Dem. Republ. of Congo | 52 | 76 | 28 |
| Africa | Ethiopia | 52 | 76 | 28 |
| Africa | South Africa | 52 | 76 | 28 |
| Asia / Pacific | China | 51 | 76 | 28 |
| Americas | Mexico | 46 | 60 | 30 |
| Americas | Brazil | 45 | 60 | 30 |
United States
PTSD affects about 5% of the US adult population each year.[244] The National Comorbidity Survey Replication has estimated that the lifetime prevalence of PTSD among adult Americans is 6.8%, with women (9.7%) more than twice as likely as men[112] (3.6%) to have PTSD at some point in their lives.[62] More than 60% of men and more than 60% of women experience at least one traumatic event in their life. The most frequently reported traumatic events by men are rape, combat, and childhood neglect or physical abuse. Women most frequently report instances of rape, sexual molestation, physical attack, being threatened with a weapon and childhood physical abuse.[112] 88% of men and 79% of women with lifetime PTSD have at least one comorbid psychiatric disorder. Major depressive disorder, 48% of men and 49% of women, and lifetime alcohol use disorder or dependence, 51.9% of men and 27.9% of women, are the most common comorbid disorders.[245]
Military combat
The United States Department of Veterans Affairs estimates that 830,000 Vietnam War veterans had symptoms of PTSD.[246] The National Vietnam Veterans' Readjustment Study (NVVRS) found 15% of male and 9% of female Vietnam veterans had PTSD at the time of the study. Life-time prevalence of PTSD was 31% for males and 27% for females. In a reanalysis of the NVVRS data, along with analysis of the data from the Matsunaga Vietnam Veterans Project, Schnurr, Lunney, Sengupta, and Waelde found that, contrary to the initial analysis of the NVVRS data, a large majority of Vietnam veterans had PTSD symptoms (but not the disorder itself). Four out of five reported recent symptoms when interviewed 20–25 years after Vietnam.[247]
A 2011 study from Georgia State University and San Diego State University found that rates of PTSD diagnosis increased significantly when troops were stationed in combat zones, had tours of longer than a year, experienced combat, or were injured. Military personnel serving in combat zones were 12.1 percentage points more likely to receive a PTSD diagnosis than their active-duty counterparts in non-combat zones. Those serving more than 12 months in a combat zone were 14.3 percentage points more likely to be diagnosed with PTSD than those having served less than one year.[248]
Experiencing an enemy firefight was associated with an 18.3 percentage point increase in the probability of PTSD, while being wounded or injured in combat was associated with a 23.9 percentage point increase in the likelihood of a PTSD diagnosis. For the 2.16 million U.S. troops deployed in combat zones between 2001 and 2010, the total estimated two-year costs of treatment for combat-related PTSD are between $1.54 billion and $2.69 billion.[248]
As of 2013, rates of PTSD have been estimated at up to 20% for veterans returning from Iraq and Afghanistan.[249] As of 2013 13% of veterans returning from Iraq were unemployed.[250]
Human-made disasters
The September 11 attacks took the lives of nearly 3,000 people, leaving 6,000 injured.[251] First responders (police, firefighters, and emergency medical technicians), sanitation workers, and volunteers were all involved in the recovery efforts. The prevalence of probable PTSD in these highly exposed populations was estimated across several studies using in-person, telephone, and online interviews and questionnaires.[251][252][253] Overall prevalence of PTSD was highest immediately following the attacks and decreased over time. However, disparities were found among the different types of recovery workers.[251][252] The rate of probable PTSD for first responders was lowest directly after the attacks and increased from ranges of 4.8–7.8% to 7.4–16.5% between the 5–6 year follow-up and a later assessment.[251]
When comparing traditional responders to non-traditional responders (volunteers), the probable PTSD prevalence 2.5 years after the initial visit was greater in volunteers with estimates of 11.7% and 17.2% respectively.[251] Volunteer participation in tasks atypical to the defined occupational role was a significant risk factor for PTSD.[252] Other risk factors included exposure intensity, earlier start date, duration of time spent on site, and constant, negative reminders of the trauma.[251][252]
Additional research has been performed to understand the social consequences of the September 11 attacks. Alcohol consumption was assessed in a cohort of World Trade Center workers using the cut-annoyed-guilty-eye (CAGE) questionnaire for alcohol use disorder. Almost 50% of World Trade Center workers who self-identified as alcohol users reported drinking more during the rescue efforts.[253] Nearly a quarter of these individuals reported drinking more following the recovery.[253] If determined to have probable PTSD status, the risk of developing an alcohol problem was double compared to those without psychological morbidity.[253] Social disability was also studied in this cohort as a social consequence of the September 11 attacks. Defined by the disruption of family, work, and social life, the risk of developing social disability increased 17-fold when categorized as having probable PTSD.[253]
Anthropology
Cultural and medical anthropologists have questioned the validity of applying the diagnostic criteria of PTSD cross-culturally.[254]
Trauma (and resulting PTSD) is often experienced through the outermost limits of suffering, pain and fear. The images and experiences relived through PTSD often defy easy description through language. Therefore, the translation of these experiences from one language to another is problematic, and the primarily Euro-American research on trauma is necessarily limited.[255] The Sapir-Whorf hypothesis suggests that people perceive the world differently according to the language they speak: language and the world it exists within reflect back on the perceptions of the speaker.[256]
For example, ethnopsychology studies in Nepal have found that cultural idioms and concepts related to trauma often do not translate to western terminologies: piDaa is a term that may align to trauma/suffering, but also people who suffer from piDaa are considered paagal (mad) and are subject to negative social stigma, indicating the need for culturally appropriate and carefully tailored support interventions.[257] More generally, different cultures remember traumatic experiences within different linguistic and cultural paradigms. As such, cultural and medical anthropologists have questioned the validity of applying the diagnostic criteria of PTSD cross-culturally, as defined in the Diagnostic and Statistical Manual of Mental Disorders (DSM-III),[needs update] and constructed through the Euro-American paradigm of psychology.[254]
There remains a dearth of studies into the conceptual frameworks that surround trauma in non-Western cultures.[254] There is little evidence to suggest therapeutic benefit in synthesizing local idioms of distress into a culturally constructed disorder of the post-Vietnam era, a practice anthropologist believe contributes to category fallacy.[clarification needed][254] For many cultures there is no single linguistic corollary to PTSD, psychological trauma being a multi-faceted concept with corresponding variances of expression.[257]
Designating the effects of trauma as an affliction of the spirit is common in many non-Western cultures where idioms such as "soul loss" and "weak heart" indicate a preference to confer suffering to a spirit-body or heart-body diametric. These idioms reflect the emphasis that collectivist cultures place on healing trauma through familial, cultural and religious activities while avoiding the stigma that accompanies a mind-body approach.[254] Prescribing PTSD diagnostics within these communities is ineffective and often detrimental.[citation needed] For trauma that extends beyond the individual, such as the effects of war, anthropologists believe applying the term "social suffering" or "cultural bereavement" to be more beneficial.[258]
Every facet of society is affected by conflict; the prolonged exposure to mass violence can lead to a 'continuous suffering' among civilians, soldiers, and bordering countries.[259] Entered into the DSM in 1980, clinicians and psychiatrists based the diagnostic criteria for PTSD around American veterans of the Vietnam War.[260] Though the DSM gets reviewed and updated regularly, it is unable to fully encompass the disorder due to its Americanization (or Westernization).[261] That is, what may be considered characteristics of PTSD in western society, may not directly translate across to other cultures around the world. Displaced people of the African country Burundi experienced symptoms of depression and anxiety, though few symptoms specific to PTSD were noted.[262]
In a similar review, Sudanese refugees relocated in Uganda were 'concerned with material [effects]' (lack of food, shelter, and healthcare), rather than psychological distress.[262] In this case, many refugees did not present symptoms at all, with a minor few developing anxiety and depression.[262] War-related stresses and traumas will be ingrained in the individual,[259] however they will be affected differently from culture to culture, and the "clear-cut" rubric for diagnosing PTSD does not allow for culturally contextual reactions to take place.[citation needed]
Veterans

United States
The United States provides a range of benefits for veterans that the VA has determined have PTSD, which developed during, or as a result of, their military service. These benefits may include tax-free cash payments,[263] free or low-cost mental health treatment and other healthcare,[264] vocational rehabilitation services,[265] employment assistance,[266] and independent living support.[267][268]
United Kingdom
In the UK, there are various charities and service organisations dedicated to aiding veterans in readjusting to civilian life. The Royal British Legion and the more recently established Help for Heroes are two of Britain's more high-profile veterans' organisations which have actively advocated for veterans over the years. There has been some controversy that the NHS has not done enough in tackling mental health issues and is instead "dumping" veterans on charities such as Combat Stress.[269][270]
Canada
Veterans Affairs Canada offers a new program that includes rehabilitation, financial benefits, job placement, health benefits program, disability awards, peer support[271][272][273] and family support.[274]
History
Aspects of PTSD in soldiers of ancient Assyria have been identified using written sources from 1300 to 600 BCE. These Assyrian soldiers would undergo a three-year rotation of combat before being allowed to return home, and were reported to have faced immense challenges in reconciling their past actions in war with their civilian lives.[275]
Connections between the actions of Viking berserkers and the hyperarousal of post-traumatic stress disorder have also been drawn.[276]
Psychiatrist Jonathan Shay has proposed that Lady Percy's soliloquy in the William Shakespeare play Henry IV, Part 1 (act 2, scene 3, lines 40–62[277]), written around 1597, represents an unusually accurate description of the symptom constellation of PTSD.[278]
A study based on personal letters from soldiers of the 18th-century Prussian Army concludes that combatants may have had PTSD.[279]
Many historical wartime diagnoses such as railway spine, stress syndrome, nostalgia, soldier's heart, shell shock, battle fatigue, combat stress reaction, and traumatic war neurosis are now associated with PTSD.[280][281]
The correlations between combat and PTSD are undeniable; according to Stéphane Audoin-Rouzeau and Annette Becker, "One-tenth of mobilized American men were hospitalized for mental disturbances between 1942 and 1945, and, after thirty-five days of uninterrupted combat, 98% of them manifested psychiatric disturbances in varying degrees."[282]
The DSM-I (1952) includes a diagnosis of "gross stress reaction", which has similarities to the modern definition and understanding of PTSD.[283] Gross stress reaction is defined as a normal personality using established patterns of reaction to deal with overwhelming fear as a response to conditions of great stress.[284] The diagnosis includes language which relates the condition to combat as well as to "civilian catastrophe".[284]

The addition of the term to the DSM-III was greatly influenced by the experiences and conditions of U.S. military veterans of the Vietnam War.[285] In fact, much of the available published research regarding PTSD is based on studies done on veterans of the war in Vietnam.
Because of the initial overt focus on PTSD as a combat related disorder when it was first fleshed out in the years following the war in Vietnam, in 1975 Ann Wolbert Burgess and Lynda Lytle Holmstrom defined rape trauma syndrome (RTS) in order to draw attention to the striking similarities between the experiences of soldiers returning from war and of rape victims.[286] This paved the way for a more comprehensive understanding of causes of PTSD.
Early in 1978, the diagnosis term "post-traumatic stress disorder" was first recommended in a working group finding presented to the Committee of Reactive Disorders.[287]
A USAF study carried out in 1979 focused on individuals (civilian and military) who had worked to recover or identify the remains of those who died in Jonestown. The bodies had been dead for several days, and a third of them had been children. The study used the term "dysphoria" to describe PTSD-like symptoms.[288]
After PTSD became an official American psychiatric diagnosis with the publication of DSM-III (1980), the number of personal injury lawsuits (tort claims) asserting the plaintiff had PTSD increased rapidly. However, triers of fact (judges and juries) often regarded the PTSD diagnostic criteria as imprecise, a view shared by legal scholars, trauma specialists, forensic psychologists, and forensic psychiatrists. The condition was termed "posttraumatic stress disorder" in the DSM-III (1980).[283][287]
Professional discussions and debates in academic journals, at conferences, and between thought leaders, led to a more clearly-defined set of diagnostic criteria in DSM-IV (1994), particularly the definition of a "traumatic event".[289] The DSM-IV classified PTSD under anxiety disorders. In the ICD-10 (first used in 1994), the spelling of the condition was "post-traumatic stress disorder".[290]
In 2012, the researchers from the Grady Trauma Project highlighted the tendency people have to focus on the combat side of PTSD: "less public awareness has focused on civilian PTSD, which results from trauma exposure that is not combat related..." and "much of the research on civilian PTSD has focused on the sequelae of a single, disastrous event, such as the Oklahoma City bombing, September 11th attacks, and Hurricane Katrina".[291] Disparity in the focus of PTSD research affected the already popular perception of the exclusive interconnectedness of combat and PTSD. This is misleading when it comes to understanding the implications and extent of PTSD as a neurological disorder.
The DSM-5 (2013) created a new category called "trauma and stressor-related disorders", in which PTSD is now classified.[1]
America's 2014 National Comorbidity Survey reports that "the traumas most commonly associated with PTSD are combat exposure and witnessing among men and rape and sexual molestation among women."[62]
Terminology
The Diagnostic and Statistical Manual of Mental Disorders does not hyphenate "post" and "traumatic", thus, the DSM-5 lists the disorder as posttraumatic stress disorder. However, many scientific journal articles and other scholarly publications do hyphenate the name of the disorder, viz., "post-traumatic stress disorder".[292] Dictionaries also differ with regard to the preferred spelling of the disorder with the Collins English Dictionary – Complete and Unabridged using the hyphenated spelling, and the American Heritage Dictionary of the English Language, Fifth Edition and the Random House Kernerman Webster's College Dictionary giving the non-hyphenated spelling.[293]
Some authors have used the terms "post-traumatic stress syndrome" or "post-traumatic stress symptoms" ("PTSS"),[294] or simply "post-traumatic stress" ("PTS") in the case of the U.S. Department of Defense,[295] to avoid stigma associated with the word "disorder".
The comedian George Carlin criticized the euphemism treadmill which led to progressive change of the way PTSD was referred to over the course of the 20th century, from "shell shock" in the First World War to the "battle fatigue" in the Second World War, to "operational exhaustion" in the Korean War, to the current "post-traumatic stress disorder", coined during the Vietnam War, which "added a hyphen" and which, he commented, "completely burie[s] [the pain] under jargon". He also stated that the name given to the condition has had a direct effect on the way veteran soldiers with PTSD were treated and perceived by civilian populations over time.[296]
Research
Most knowledge regarding PTSD comes from studies in high-income countries.[297]
To recapitulate some of the neurological and neurobehavioral symptoms experienced by the veteran population of recent conflicts in Iraq and Afghanistan, researchers at the Roskamp Institute and the James A Haley Veteran's Hospital (Tampa) have developed an animal model to study the consequences of mild traumatic brain injury (mTBI) and PTSD.[298] In the laboratory, the researchers exposed mice to a repeated session of unpredictable stressor (i.e. predator odor while restrained), and physical trauma in the form of inescapable foot-shock, and this was also combined with a mTBI. In this study, PTSD animals demonstrated recall of traumatic memories, anxiety, and an impaired social behavior, while animals subject to both mTBI and PTSD had a pattern of disinhibitory-like behavior. mTBI abrogated both contextual fear and impairments in social behavior seen in PTSD animals. In comparison with other animal studies,[298][299] examination of neuroendocrine and neuroimmune responses in plasma revealed a trend toward increase in corticosterone in PTSD and combination groups.
Stellate ganglion block is an experimental procedure for the treatment of PTSD.[300]
Researchers are investigating a number of experimental FAAH and MAGL-inhibiting drugs in hopes of finding a better treatment for anxiety and stress-related illnesses.[301] In 2016, the FAAH-inhibitor drug BIA 10-2474 was withdrawn from human trials in France due to adverse effects.[302]
Preliminary evidence suggests that MDMA-assisted psychotherapy might be an effective treatment for PTSD.[303][304] However, it is important to note that the results in clinical trials of MDMA-assisted psychotherapy might be substantially influenced by expectancy effects given the unblinding of participants.[305][306] Furthermore, there is a lack of trials comparing MDMA-assisted psychotherapy to existent first-line treatments for PTSD, such as trauma-focused psychological treatments, which seems to achieve similar or even better outcomes than MDMA-assisted psychotherapy.[307]
Psychotherapy
Trauma-focused psychotherapies for PTSD (also known as "exposure-based" or "exposure" psychotherapies), such as prolonged exposure therapy (PE), eye movement desensitization and reprocessing (EMDR), and cognitive-reprocessing therapy (CPT) have the most evidence for efficacy and are recommended as first-line treatment for PTSD by almost all clinical practice guidelines.[308][309][310] Exposure-based psychotherapies demonstrate efficacy for PTSD caused by different trauma "types", such as combat, sexual-assault, or natural disasters.[308] At the same time, many trauma-focused psychotherapies evince high drop-out rates.[311]
Most systematic reviews and clinical guidelines indicate that psychotherapies for PTSD, most of which are trauma-focused therapies, are more effective than pharmacotherapy (medication),[312] although there are reviews that suggest exposure-based psychotherapies for PTSD and pharmacotherapy are equally effective.[313] Interpersonal psychotherapy shows preliminary evidence of probable efficacy, but more research is needed to reach definitive conclusions.[314]
See also
- Childbirth-related posttraumatic stress disorder
- Internet-based treatments for trauma survivors
- Internet interventions for post-traumatic stress
- Post-traumatic stress disorder and substance use disorders
- Post-traumatic embitterment disorder
- Post-traumatic growth
- Post-traumatic stress disorder after World War II
- Symptoms of victimization
- Traumatic stress
Notes
- ^ At least 1 month of symptoms for clinical diagnosis is required, while symptoms may persist from 6 months to multiple years.[1][3]
- ^ Acceptable variants of this term exist; see the Terminology section in this article.
- ^ As an example of such research, see: Bonn-Miller MO, Sisley S, Riggs P, Yazar-Klosinski B, Wang JB, Loflin MJE, et al. (2021) The short-term impact of 3 smoked cannabis preparations versus placebo on PTSD symptoms: A randomized cross-over clinical trial. PLOS ONE 16(3): e0246990.
References
- ^ a b c d e f g h i j k l m n o American Psychiatric Association (2013). Diagnostic and Statistical Manual of Mental Disorders (5th ed.). Arlington, VA: American Psychiatric Publishing. pp. 271–80. ISBN 978-0-89042-555-8.
- ^ a b c d e f g h Bisson JI, Cosgrove S, Lewis C, Robert NP (November 2015). "Post-traumatic stress disorder". BMJ. 351: h6161. doi:10.1136/bmj.h6161. PMC 4663500. PMID 26611143.
- ^ "Post-Traumatic Stress Disorder". www.nimh.nih.gov. Retrieved 13 January 2024.
- ^ a b c d e f "Post-Traumatic Stress Disorder". National Institute of Mental Health. February 2016. Archived from the original on 9 March 2016. Retrieved 10 March 2016.
- ^ "Notice of final decision to amend (or not amend) the current Poisons Standard – June 2022 ACMS #38 – Psilocybine and MDMA". Therapeutic Goods Administration, Department of Health and Aged Care, Australian Government. Retrieved 19 April 2023.
- ^ a b c d e Berger W, Mendlowicz MV, Marques-Portella C, Kinrys G, Fontenelle LF, Marmar CR, et al. (March 2009). "Pharmacologic alternatives to antidepressants in posttraumatic stress disorder: a systematic review". Progress in Neuro-Psychopharmacology & Biological Psychiatry. 33 (2): 169–80. doi:10.1016/j.pnpbp.2008.12.004. PMC 2720612. PMID 19141307.
- ^ Diagnostic and statistical manual of mental disorders: DSM-5 (5th ed.). Arlington, VA: American Psychiatric Association. 2013. p. 276. ISBN 978-0-89042-555-8. OCLC 830807378.
- ^ Sartorius N, Henderson A, Strotzka H, Lipowski Z, Yu-cun S, You-xin X, et al. "The ICD-10 Classification of Mental and Behavioural Disorders Clinical descriptions and diagnostic guidelines" (PDF). www.who.int World Health Organization. Microsoft Word. bluebook.doc. p. 110. Retrieved 3 July 2021 – via Microsoft Bing.
- ^ "Post-traumatic stress disorder (PTSD) – Symptoms and causes". Mayo Clinic. Retrieved 8 October 2019.
- ^ Forman-Hoffman V, Cook Middleton J, Feltner C, Gaynes BN, Palmieri Weber R, Bann C, et al. (17 May 2018). Psychological and Pharmacological Treatments for Adults With Posttraumatic Stress Disorder: A Systematic Review Update (Report). Agency for Healthcare Research and Quality (AHRQ). doi:10.23970/ahrqepccer207 (inactive 23 March 2024).
{{cite report}}: CS1 maint: DOI inactive as of March 2024 (link) - ^ Panagioti M, Gooding PA, Triantafyllou K, Tarrier N (April 2015). "Suicidality and posttraumatic stress disorder (PTSD) in adolescents: a systematic review and meta-analysis". Social Psychiatry and Psychiatric Epidemiology. 50 (4): 525–537. doi:10.1007/s00127-014-0978-x. PMID 25398198. S2CID 23314414.
- ^ Zoladz PR, Diamond DM (June 2013). "Current status on behavioral and biological markers of PTSD: a search for clarity in a conflicting literature". Neuroscience and Biobehavioral Reviews. 37 (5): 860–895. doi:10.1016/j.neubiorev.2013.03.024. PMID 23567521. S2CID 14440116.
- ^ Kessler RC, Aguilar-Gaxiola S, Alonso J, Benjet C, Bromet EJ, Cardoso G, et al. (27 October 2017). "Trauma and PTSD in the WHO World Mental Health Surveys". European Journal of Psychotraumatology. 8 (sup5): 1353383. doi:10.1080/20008198.2017.1353383. PMC 5632781. PMID 29075426.
As detailed in another recent WMH report, conditional risk of PTSD after trauma exposure is 4.0%, but varies significantly by trauma type. The highest conditional risk is associated with being raped (19.0%), physical abuse by a romantic partner (11.7%), being kidnapped (11.0%), and being sexually assaulted other than rape (10.5%). In terms of broader categories, the traumas associated with the highest PTSD risk are those involving intimate partner or sexual violence (11.4%), and other traumas (9.2%), with aggregate conditional risk much lower in the other broad trauma categories (2.0–5.4%) [citations omitted; emphasis added].
- ^ Darves-Bornoz JM, Alonso J, de Girolamo G, de Graaf R, Haro JM, Kovess-Masfety V, et al. (October 2008). "Main traumatic events in Europe: PTSD in the European study of the epidemiology of mental disorders survey". Journal of Traumatic Stress. 21 (5): 455–462. doi:10.1002/jts.20357. PMID 18956444.
In univariate analyses adjusted on gender, six events were found to be the most significantly associated with PTSD ( p < .001) among individuals exposed to at least one event. They were being raped (OR = 8.9), being beaten up by spouse or romantic partner (OR = 7.3), experiencing an undisclosed private event (OR = 5.5), having a child with serious illness (OR = 5.1), being beaten up by a caregiver (OR = 4.5), or being stalked (OR = 4.2)" [OR = odds ratio].
- ^ Brewin CR, Cloitre M, Hyland P, Shevlin M, Maercker A, Bryant RA, et al. (December 2017). "A review of current evidence regarding the ICD-11 proposals for diagnosing PTSD and complex PTSD" (PDF). Clinical Psychology Review. 58: 1–15. doi:10.1016/j.cpr.2017.09.001. PMID 29029837. S2CID 4874961.
- ^ a b Haagen JF, Smid GE, Knipscheer JW, Kleber RJ (August 2015). "The efficacy of recommended treatments for veterans with PTSD: A metaregression analysis". Clinical Psychology Review. 40: 184–94. doi:10.1016/j.cpr.2015.06.008. PMID 26164548.
- ^ a b Hetrick SE, Purcell R, Garner B, Parslow R (July 2010). "Combined pharmacotherapy and psychological therapies for post traumatic stress disorder (PTSD)" (PDF). The Cochrane Database of Systematic Reviews (7): CD007316. doi:10.1002/14651858.CD007316.pub2. PMID 20614457.
- ^ a b c Guina J, Rossetter SR, DeRHODES BJ, Nahhas RW, Welton RS (July 2015). "Benzodiazepines for PTSD: A Systematic Review and Meta-Analysis". Journal of Psychiatric Practice. 21 (4): 281–303. doi:10.1097/pra.0000000000000091. PMID 26164054. S2CID 24968844.
- ^ a b c d Hoskins M, Pearce J, Bethell A, Dankova L, Barbui C, Tol WA, et al. (February 2015). "Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis". The British Journal of Psychiatry. 206 (2): 93–100. doi:10.1192/bjp.bp.114.148551. PMID 25644881.
Some drugs have a small positive impact on PTSD symptoms
- ^ Carlstedt R (2009). Handbook of Integrative Clinical Psychology, Psychiatry, and Behavioral Medicine Perspectives, Practices, and Research. New York: Springer Pub. Co. p. 353. ISBN 978-0-8261-1095-4 – via Google Books.
- ^ O'Brien S (1998). Traumatic Events and Mental Health. Cambridge University Press. p. 7.
- ^ Herman J (2015). Trauma and Recovery: The Aftermath of Violence – From Domestic Abuse to Political Terror. Basic Books. p. 9. ISBN 978-0-465-09873-6 – via Google Books.
- ^ After War: A Conversation with Author Nancy Sherman, by John Waters, Real Clear Defense, 4 June 2015
- ^ Klykylo WM (2012). "15". Clinical child psychiatry (3 ed.). Chichester, West Sussex, UK: John Wiley & Sons. ISBN 978-1-119-96770-5 – via Google Books.
- ^ Friedman MJ (October 2013). "Finalizing PTSD in DSM-5: getting here from there and where to go next". Journal of Traumatic Stress. 26 (5): 548–56. doi:10.1002/jts.21840. PMID 24151001.
- ^ a b Waltman SM, Shearer D, Moore BA (11 October 2018). "Management of Post-Traumatic Nightmares: a Review of Pharmacologic and Nonpharmacologic Treatments Since 2013". Current Psychiatry Reports. 20 (12). Springer Science and Business Media LLC: 108. doi:10.1007/s11920-018-0971-2. ISSN 1523-3812. PMID 30306339. S2CID 52958432.
- ^ a b c American Psychiatric Association (1994). Diagnostic and statistical manual of mental disorders: DSM-IV. Washington, DC: American Psychiatric Association. ISBN 978-0-89042-061-4.[page needed];
- ^ Rebecca C. "The primary care PTSD screen (PC-PTSD): development and operating characteristics" (PDF). Washington University. Primary Care Psychiatry. Retrieved 19 November 2022.
- ^ a b Rothschild B (2000). The Body Remembers: The Psychophysiology of Trauma and Trauma Treatment. New York: W.W. Norton & Company. ISBN 978-0-393-70327-6.[page needed]
- ^ Kaplan HI, Sadock BJ (1994). Grebb JA (ed.). Kaplan and Sadock's synopsis of psychiatry: Behavioral sciences, clinical psychiatry (7th ed.). Baltimore: Williams & Williams. pp. 606–609.[page needed]
- ^ Satcher D (1999). "Chapter 4". Mental Health: A Report of the Surgeon General. Surgeon General of the United States. Archived from the original on 2 July 2010.
- ^ Bernstein M, Pfefferbaum B (May 2018). "Posttraumatic Growth as a Response to Natural Disasters in Children and Adolescents". Current Psychiatry Reports. 20 (5): 37. doi:10.1007/s11920-018-0900-4. PMID 29766312. S2CID 21721645.
- ^ O'Donnell ML, Creamer M, Bryant RA, Schnyder U, Shalev A (July 2003). "Posttraumatic disorders following injury: an empirical and methodological review". Clinical Psychology Review. 23 (4): 587–603. doi:10.1016/S0272-7358(03)00036-9. PMID 12788111.
- ^ a b c d e f g h i j Shalev A, Liberzon I, Marmar C (June 2017). "Post-Traumatic Stress Disorder". The New England Journal of Medicine. 376 (25): 2459–2469. doi:10.1056/NEJMra1612499. PMID 28636846.
- ^ Maxmen JS, Ward NG (2002). Psychotropic drugs: fast facts (3rd ed.). New York: W.W. Norton & Company. p. 348. ISBN 978-0-393-70301-6.
- ^ Cohen SI (February 1995). "Alcohol and benzodiazepines generate anxiety, panic and phobias". Journal of the Royal Society of Medicine. 88 (2): 73–7. PMC 1295099. PMID 7769598.
- ^ Spates R, Souza T (2007). "Treatment of PTSD and Substance Abuse Comorbidity" (PDF). The Behavior Analyst Today. 9 (1): 11–26. doi:10.1037/h0100643. Archived from the original (PDF) on 6 November 2014.
- ^ Cima R, Mazurek B, Haider H, Kikidis D, Lapira A, Noreña A, et al. (2019). "A multidisciplinary European guideline for tinnitus: diagnostics, assessment, and treatment". HNO. 67 (Suppl 1): 10–42. doi:10.1007/s00106-019-0633-7. PMID 30847513. S2CID 71145857.
- ^ Mazurek B, Haupt H, Olze H, Szczepeck A (2022). "Stress and tinnitus—from bedside to bench and back". Frontiers in Systems Neuroscience. 6 (47): 47. doi:10.3389/fnsys.2012.00047. PMC 3371598. PMID 22701404.
- ^ Villalta L, Smith P, Hickin N, Stringaris A (April 2018). "Emotion regulation difficulties in traumatized youth: a meta-analysis and conceptual review" (PDF). European Child & Adolescent Psychiatry. 27 (4): 527–544. doi:10.1007/s00787-018-1105-4. PMID 29380069. S2CID 4731753.
- ^ Hall NA, Everson AT, Billingsley MR, Miller MB (January 2022). "Moral injury, mental health and behavioural health outcomes: A systematic review of the literature". Clinical Psychology & Psychotherapy. 29 (1): 92–110. doi:10.1002/cpp.2607. PMID 33931926. S2CID 233471425.
- ^ Schlenger WE, Corry NH, Williams CS, Kulka RA, Mulvaney-Day N, DeBakey S, et al. (2 December 2015). "A Prospective Study of Mortality and Trauma-Related Risk Factors Among a Nationally Representative Sample of Vietnam Veterans". American Journal of Epidemiology. 182 (12): 980–990. doi:10.1093/aje/kwv217. PMID 26634285.
- ^ Robinson M (27 May 2006). "Review of Francisco Goya's Disasters of War". Associated Press. Archived from the original on 28 July 2014.
- ^ Fullerton CS, Ursano RJ, Wang L (August 2004). "Acute stress disorder, posttraumatic stress disorder, and depression in disaster or rescue workers". The American Journal of Psychiatry. 161 (8): 1370–6. CiteSeerX 10.1.1.600.4486. doi:10.1176/appi.ajp.161.8.1370. PMID 15285961.
- ^ Skogstad M, Skorstad M, Lie A, Conradi HS, Heir T, Weisæth L (April 2013). "Work-related post-traumatic stress disorder". Occupational Medicine. 63 (3): 175–82. doi:10.1093/occmed/kqt003. PMID 23564090.
- ^ Vieweg WV, Julius DA, Fernandez A, Beatty-Brooks M, Hettema JM, Pandurangi AK (May 2006). "Posttraumatic stress disorder: clinical features, pathophysiology, and treatment". The American Journal of Medicine. 119 (5): 383–90. doi:10.1016/j.amjmed.2005.09.027. PMID 16651048.
- ^ Dekel S, Gilbertson MW, Orr SP, Rauch SL, Wood NE, Pitman RK (2016). "Trauma and Posttraumatic Stress Disorder". In Stern TA, Fava M, Wilens TE, Rosenbaum JF (eds.). Massachusetts General Hospital comprehensive clinical psychiatry (Second ed.). London: Elsevier. pp. 380–392. ISBN 978-0-323-29507-9. OCLC 905232521.
- ^ a b c d e Kessler RC, Aguilar-Gaxiola S, Alonso J, Benjet C, Bromet EJ, Cardoso G, et al. (27 October 2017). "Trauma and PTSD in the WHO World Mental Health Surveys". European Journal of Psychotraumatology. 8 (sup5): 1353383. doi:10.1080/20008198.2017.1353383. PMC 5632781. PMID 29075426.
- ^ National Collaborating Centre for Mental Health (UK) (2005). Post-Traumatic Stress Disorder: The Management of PTSD in Adults and Children in Primary and Secondary Care. National Institute for Health and Clinical Excellence: Guidance. Gaskell (Royal College of Psychiatrists). ISBN 978-1-904671-25-1. NICE Clinical Guidelines, No. 26.
- ^ a b Lin W, Gong L, Xia M, Dai W (January 2018). "Prevalence of posttraumatic stress disorder among road traffic accident survivors: A PRISMA-compliant meta-analysis". Medicine. 97 (3): e9693. doi:10.1097/md.0000000000009693. PMC 5779792. PMID 29505023.
- ^ a b Dai W, Liu A, Kaminga AC, Deng J, Lai Z, Wen SW (August 2018). "Prevalence of Posttraumatic Stress Disorder among Children and Adolescents following Road Traffic Accidents: A Meta-Analysis". Canadian Journal of Psychiatry. 63 (12): 798–808. doi:10.1177/0706743718792194. PMC 6309043. PMID 30081648.
- ^ Bisson JI, Berliner L, Cloitre M, Forbes D, Jensen TK, Lewis C, et al. (August 2019). "The International Society for Traumatic Stress Studies New Guidelines for the Prevention and Treatment of Posttraumatic Stress Disorder: Methodology and Development Process" (PDF). Journal of Traumatic Stress. 32 (4): 475–483. doi:10.1002/jts.22421. hdl:10852/77258. PMID 31283056. S2CID 195830995.
- ^ a b c d e f g National Collaborating Centre for Mental Health (UK) (2005). Post-Traumatic Stress Disorder: The Management of PTSD in Adults and Children in Primary and Secondary Care. National Institute for Health and Clinical Excellence: Guidance No. 26. Gaskell (Royal College of Psychiatrists). ISBN 978-1-904671-25-1. Archived from the original on 8 September 2017.
- ^ Alisic E, Zalta AK, van Wesel F, Larsen SE, Hafstad GS, Hassanpour K, et al. (2014). "Rates of post-traumatic stress disorder in trauma-exposed children and adolescents: meta-analysis" (PDF). The British Journal of Psychiatry. 204 (5): 335–40. doi:10.1192/bjp.bp.113.131227. PMID 24785767.
- ^ Lai BS, Lewis R, Livings MS, La Greca AM, Esnard AM (December 2017). "Posttraumatic Stress Symptom Trajectories Among Children After Disaster Exposure: A Review". Journal of Traumatic Stress. 30 (6): 571–582. doi:10.1002/jts.22242. PMC 5953201. PMID 29193316.
- ^ Koenen KC, Moffitt TE, Poulton R, Martin J, Caspi A (February 2007). "Early childhood factors associated with the development of post-traumatic stress disorder: results from a longitudinal birth cohort". Psychological Medicine. 37 (2): 181–92. doi:10.1017/S0033291706009019. PMC 2254221. PMID 17052377.
- ^ Lapp KG, Bosworth HB, Strauss JL, Stechuchak KM, Horner RD, Calhoun PS, et al. (September 2005). "Lifetime sexual and physical victimization among male veterans with combat-related post-traumatic stress disorder". Military Medicine. 170 (9): 787–90. doi:10.7205/MILMED.170.9.787. PMID 16261985.
- ^ Otte C, Neylan TC, Pole N, Metzler T, Best S, Henn-Haase C, et al. (January 2005). "Association between childhood trauma and catecholamine response to psychological stress in police academy recruits". Biological Psychiatry. 57 (1): 27–32. doi:10.1016/j.biopsych.2004.10.009. PMID 15607297. S2CID 35801179.
- ^ a b c d e f g h i Skelton K, Ressler KJ, Norrholm SD, Jovanovic T, Bradley-Davino B (February 2012). "PTSD and gene variants: new pathways and new thinking". Neuropharmacology. 62 (2): 628–37. doi:10.1016/j.neuropharm.2011.02.013. PMC 3136568. PMID 21356219.
- ^ Janoff-Bulman R (1992). Shattered Assumptions: Toward a New Psychology of Trauma. New York: Free Press.[page needed]
- ^ Scheeringa MS (2015). "Untangling Psychiatric Comorbidity in Young Children Who Experienced Single, Repeated, or Hurricane Katrina Traumatic Events". Child and Youth Care Forum. 44 (4): 475–492. doi:10.1007/s10566-014-9293-7. PMC 4511493. PMID 26213455.
- ^ a b c d Kessler RC, Sonnega A, Bromet E, Hughes M, Nelson CB (December 1995). "Posttraumatic stress disorder in the National Comorbidity Survey". Archives of General Psychiatry. 52 (12): 1048–60. doi:10.1001/archpsyc.1995.03950240066012. PMID 7492257. S2CID 14189766.
- ^ Liu H, Petukhova MV, Sampson NA, Aguilar-Gaxiola S, Alonso J, Andrade LH, et al. (March 2017). "Association of DSM-IV Posttraumatic Stress Disorder With Traumatic Experience Type and History in the World Health Organization World Mental Health Surveys". JAMA Psychiatry. 74 (3): 270–281. doi:10.1001/jamapsychiatry.2016.3783. PMC 5441566. PMID 28055082.
- ^ Howard LM, Oram S, Galley H, Trevillion K, Feder G (2013). "Domestic violence and perinatal mental disorders: a systematic review and meta-analysis". PLOS Medicine. 10 (5): e1001452. doi:10.1371/journal.pmed.1001452. PMC 3665851. PMID 23723741.
- ^ Hoffman BL, Schorge JO, Bradshaw KD, Halvorson LM, Schaffer JI, Corton MM, eds. (2016). Williams Gynecology (3rd ed.). McGraw Hill Professional. ISBN 978-0-07-184909-8.
- ^ Surís A, Lind L, Kashner TM, Borman PD, Petty F (2004). "Sexual assault in women veterans: an examination of PTSD risk, health care utilization, and cost of care". Psychosomatic Medicine. 66 (5): 749–56. CiteSeerX 10.1.1.508.9827. doi:10.1097/01.psy.0000138117.58559.7b. PMID 15385701. S2CID 14118203.
- ^ Mason F, Lodrick Z (February 2013). "Psychological consequences of sexual assault". Best Practice & Research. Clinical Obstetrics & Gynaecology. 27 (1): 27–37. doi:10.1016/j.bpobgyn.2012.08.015. PMID 23182852.
- ^ Hollifield M, Warner TD, Lian N, Krakow B, Jenkins JH, Kesler J, et al. (August 2002). "Measuring trauma and health status in refugees: a critical review". JAMA. 288 (5): 611–21. doi:10.1001/jama.288.5.611. PMID 12150673.
- ^ Porter M, Haslam N (October 2001). "Forced displacement in Yugoslavia: a meta-analysis of psychological consequences and their moderators". Journal of Traumatic Stress. 14 (4): 817–34. doi:10.1023/A:1013054524810. PMID 11776427. S2CID 41804120.
- ^ a b UNESCO (2018). A Lifeline to learning: leveraging mobile technology to support education for refugees. UNESCO. ISBN 978-92-3-100262-5.
- ^ a b Atwoli L, Stein DJ, King A, Petukhova M, Aguilar-Gaxiola S, Alonso J, et al. (April 2017). "Posttraumatic stress disorder associated with unexpected death of a loved one: Cross-national findings from the world mental health surveys". Depression and Anxiety. 34 (4): 315–326. doi:10.1002/da.22579. PMC 5661943. PMID 27921352.
- ^ a b "Cancer-Related Post-traumatic Stress". National Cancer Institute. January 1980. Retrieved 16 September 2017.
- ^ Swartzman S, Booth JN, Munro A, Sani F (April 2017). "Posttraumatic stress disorder after cancer diagnosis in adults: A meta-analysis". Depression and Anxiety (Submitted manuscript). 34 (4): 327–339. doi:10.1002/da.22542. hdl:20.500.11820/b8651f89-9611-4f50-8766-3d5b64b8be23. PMID 27466972. S2CID 1828418.
- ^ Cordova MJ, Riba MB, Spiegel D (April 2017). "Post-traumatic stress disorder and cancer". The Lancet. Psychiatry. 4 (4): 330–338. doi:10.1016/S2215-0366(17)30014-7. PMC 5676567. PMID 28109647.
- ^ Edmondson D, Richardson S, Falzon L, Davidson KW, Mills MA, Neria Y (2012). "Posttraumatic stress disorder prevalence and risk of recurrence in acute coronary syndrome patients: a meta-analytic review". PLOS ONE. 7 (6): e38915. Bibcode:2012PLoSO...738915E. doi:10.1371/journal.pone.0038915. PMC 3380054. PMID 22745687.
- ^ Edmondson D, Richardson S, Fausett JK, Falzon L, Howard VJ, Kronish IM (19 June 2013). "Prevalence of PTSD in Survivors of Stroke and Transient Ischemic Attack: A Meta-Analytic Review". PLOS ONE. 8 (6): e66435. Bibcode:2013PLoSO...866435E. doi:10.1371/journal.pone.0066435. PMC 3686746. PMID 23840467.
- ^ Abbey G, Thompson SB, Hickish T, Heathcote D (April 2015). "A meta-analysis of prevalence rates and moderating factors for cancer-related post-traumatic stress disorder". Psycho-Oncology. 24 (4): 371–81. doi:10.1002/pon.3654. PMC 4409098. PMID 25146298.
- ^ Davydow DS, Gifford JM, Desai SV, Needham DM, Bienvenu OJ (September 2008). "Posttraumatic stress disorder in general intensive care unit survivors: a systematic review". General Hospital Psychiatry. 30 (5): 421–34. doi:10.1016/j.genhosppsych.2008.05.006. PMC 2572638. PMID 18774425.
- ^ Arnaboldi P, Riva S, Crico C, Pravettoni G (2017). "A systematic literature review exploring the prevalence of post-traumatic stress disorder and the role played by stress and traumatic stress in breast cancer diagnosis and trajectory". Breast Cancer: Targets and Therapy. 9: 473–485. doi:10.2147/BCTT.S111101. PMC 5505536. PMID 28740430.
- ^ Liu C, Zhang Y, Jiang H, Wu H (5 May 2017). "Association between social support and post-traumatic stress disorder symptoms among Chinese patients with ovarian cancer: A multiple mediation model". PLOS ONE. 12 (5): e0177055. Bibcode:2017PLoSO..1277055L. doi:10.1371/journal.pone.0177055. PMC 5419605. PMID 28475593.
- ^ "PsycNET". psycnet.apa.org. Retrieved 30 September 2018.
- ^ Buswell G, Haime Z, Lloyd-Evans B, Billings J (January 2021). "A systematic review of PTSD to the experience of psychosis: prevalence and associated factors". BMC Psychiatry. 21 (1): 9. doi:10.1186/s12888-020-02999-x. PMC 7789184. PMID 33413179.
- ^ Shaw K, McFarlane A, Bookless C (July 1997). "The phenomenology of traumatic reactions to psychotic illness". The Journal of Nervous and Mental Disease. 185 (7): 434–441. doi:10.1097/00005053-199707000-00003. PMID 9240361.
- ^ Lu W, Mueser KT, Rosenberg SD, Yanos PT, Mahmoud N (19 July 2017). "Posttraumatic Reactions to Psychosis: A Qualitative Analysis". Frontiers in Psychiatry. 8: 129. doi:10.3389/fpsyt.2017.00129. PMC 5515869. PMID 28769826.
- ^ a b Christiansen DM (February 2017). "Posttraumatic stress disorder in parents following infant death: A systematic review". Clinical Psychology Review. 51: 60–74. doi:10.1016/j.cpr.2016.10.007. PMID 27838460.
- ^ Kersting A, Wagner B (June 2012). "Complicated grief after perinatal loss". Dialogues in Clinical Neuroscience. 14 (2): 187–94. doi:10.31887/DCNS.2012.14.2/akersting. PMC 3384447. PMID 22754291.
- ^ Daugirdaitė V, van den Akker O, Purewal S (2015). "Posttraumatic stress and posttraumatic stress disorder after termination of pregnancy and reproductive loss: a systematic review". Journal of Pregnancy. 2015: 646345. doi:10.1155/2015/646345. PMC 4334933. PMID 25734016.
- ^ Ayers S, Bond R, Bertullies S, Wijma K (April 2016). "The aetiology of post-traumatic stress following childbirth: a meta-analysis and theoretical framework". Psychological Medicine. 46 (6): 1121–34. doi:10.1017/s0033291715002706. PMID 26878223.
- ^ James S (December 2015). "Women's experiences of symptoms of posttraumatic stress disorder (PTSD) after traumatic childbirth: a review and critical appraisal". Archives of Women's Mental Health. 18 (6): 761–71. doi:10.1007/s00737-015-0560-x. PMC 4624822. PMID 26264506.
- ^ a b c Olde E, van der Hart O, Kleber R, van Son M (January 2006). "Posttraumatic stress following childbirth: a review". Clinical Psychology Review. 26 (1): 1–16. doi:10.1016/j.cpr.2005.07.002. hdl:1874/16760. PMID 16176853. S2CID 22137961.
- ^ Alder J, Stadlmayr W, Tschudin S, Bitzer J (June 2006). "Post-traumatic symptoms after childbirth: what should we offer?". Journal of Psychosomatic Obstetrics and Gynaecology. 27 (2): 107–12. doi:10.1080/01674820600714632. PMID 16808085. S2CID 21859634.
- ^ Montmasson H, Bertrand P, Perrotin F, El-Hage W (October 2012). "[Predictors of postpartum post-traumatic stress disorder in primiparous mothers]". Journal de Gynécologie, Obstétrique et Biologie de la Reproduction. 41 (6): 553–60. doi:10.1016/j.jgyn.2012.04.010. PMID 22622194. S2CID 196363612.
- ^ Martin C (2012). Perinatal Mental Health: a Clinical Guide. Cumbria, England: M & K Pub. p. 26. ISBN 978-1-907830-49-5.
- ^ True WR, Rice J, Eisen SA, Heath AC, Goldberg J, Lyons MJ, et al. (April 1993). "A twin study of genetic and environmental contributions to liability for posttraumatic stress symptoms". Archives of General Psychiatry. 50 (4): 257–264. doi:10.1001/archpsyc.1993.01820160019002. PMID 8466386.
- ^ Quidé Y, Andersson F, Dufour-Rainfray D, Descriaud C, Brizard B, Gissot V, et al. (October 2018). "Smaller hippocampal volume following sexual assault in women is associated with post-traumatic stress disorder". Acta Psychiatrica Scandinavica. 138 (4): 312–324. doi:10.1111/acps.12920. PMID 29952088. S2CID 49484570.
- ^ Yamasue H, Kasai K, Iwanami A, Ohtani T, Yamada H, Abe O, et al. (July 2003). "Voxel-based analysis of MRI reveals anterior cingulate gray-matter volume reduction in posttraumatic stress disorder due to terrorism". Proceedings of the National Academy of Sciences of the United States of America. 100 (15): 9039–43. Bibcode:2003PNAS..100.9039Y. doi:10.1073/pnas.1530467100. PMC 166434. PMID 12853571.
- ^ The Secret Life of the Brain (Series), episode 4. PBS. 2001. Archived from the original on 2 February 2014. Retrieved 29 January 2014.
- ^ a b Zohar J, Juven-Wetzler A, Myers V, Fostick L (January 2008). "Post-traumatic stress disorder: facts and fiction". Current Opinion in Psychiatry. 21 (1): 74–7. doi:10.1097/YCO.0b013e3282f269ee. PMID 18281844. S2CID 206142172.
- ^ Yehuda R, Halligan SL, Golier JA, Grossman R, Bierer LM (April 2004). "Effects of trauma exposure on the cortisol response to dexamethasone administration in PTSD and major depressive disorder". Psychoneuroendocrinology. 29 (3): 389–404. doi:10.1016/S0306-4530(03)00052-0. PMID 14644068. S2CID 21615196.
- ^ Yehuda R, Halligan SL, Grossman R, Golier JA, Wong C (September 2002). "The cortisol and glucocorticoid receptor response to low dose dexamethasone administration in aging combat veterans and holocaust survivors with and without posttraumatic stress disorder". Biological Psychiatry. 52 (5): 393–403. doi:10.1016/S0006-3223(02)01357-4. PMID 12242055. S2CID 21403230.
- ^ Heim C, Ehlert U, Hellhammer DH (January 2000). "The potential role of hypocortisolism in the pathophysiology of stress-related bodily disorders". Psychoneuroendocrinology. 25 (1): 1–35. doi:10.1016/S0306-4530(99)00035-9. PMID 10633533. S2CID 25151441.
- ^ Mason JW, Giller EL, Kosten TR, Harkness L (August 1988). "Elevation of urinary norepinephrine/cortisol ratio in posttraumatic stress disorder". The Journal of Nervous and Mental Disease. 176 (8): 498–502. doi:10.1097/00005053-198808000-00008. PMID 3404142. S2CID 24585702.
- ^ Bohnen N, Nicolson N, Sulon J, Jolles J (1991). "Coping style, trait anxiety and cortisol reactivity during mental stress". Journal of Psychosomatic Research. 35 (2–3): 141–7. CiteSeerX 10.1.1.467.4323. doi:10.1016/0022-3999(91)90068-Y. PMID 2046048.
- ^ Geracioti TD, Baker DG, Ekhator NN, West SA, Hill KK, Bruce AB, et al. (August 2001). "CSF norepinephrine concentrations in posttraumatic stress disorder". The American Journal of Psychiatry. 158 (8): 1227–30. doi:10.1176/appi.ajp.158.8.1227. PMID 11481155.
- ^ Sautter FJ, Bissette G, Wiley J, Manguno-Mire G, Schoenbachler B, Myers L, et al. (December 2003). "Corticotropin-releasing factor in posttraumatic stress disorder (PTSD) with secondary psychotic symptoms, nonpsychotic PTSD, and healthy control subjects". Biological Psychiatry. 54 (12): 1382–8. doi:10.1016/S0006-3223(03)00571-7. PMID 14675802. S2CID 35766262.
- ^ de Kloet CS, Vermetten E, Geuze E, Lentjes EG, Heijnen CJ, Stalla GK, et al. (2008). "Elevated plasma corticotrophin-releasing hormone levels in veterans with posttraumatic stress disorder". Stress Hormones and Post Traumatic Stress Disorder Basic Studies and Clinical Perspectives. Progress in Brain Research. Vol. 167. pp. 287–91. doi:10.1016/S0079-6123(07)67025-3. ISBN 978-0-444-53140-7. PMID 18037027.
- ^ Radley JJ, Kabbaj M, Jacobson L, Heydendael W, Yehuda R, Herman JP (September 2011). "Stress risk factors and stress-related pathology: neuroplasticity, epigenetics and endophenotypes". Stress. 14 (5): 481–97. doi:10.3109/10253890.2011.604751. PMC 3641164. PMID 21848436.
- ^ Pitman RK (July 1989). "Post-traumatic stress disorder, hormones, and memory". Biological Psychiatry. 26 (3): 221–3. doi:10.1016/0006-3223(89)90033-4. PMID 2545287. S2CID 39057765.
- ^ Yehuda R (2001). "Biology of posttraumatic stress disorder". The Journal of Clinical Psychiatry. 62. 62 (Suppl 17): 41–46. PMID 11495096.
- ^ Yehuda R (2002). "Clinical relevance of biologic findings in PTSD". The Psychiatric Quarterly. 73 (2): 123–33. doi:10.1023/A:1015055711424. PMID 12025720. S2CID 19767960.
- ^ Aardal-Eriksson E, Eriksson TE, Thorell LH (December 2001). "Salivary cortisol, posttraumatic stress symptoms, and general health in the acute phase and during 9-month follow-up". Biological Psychiatry. 50 (12): 986–93. doi:10.1016/S0006-3223(01)01253-7. PMID 11750895. S2CID 9149956.
- ^ a b c d e Olszewski TM, Varrasse JF (June 2005). "The neurobiology of PTSD: implications for nurses". Journal of Psychosocial Nursing and Mental Health Services. 43 (6): 40–7. doi:10.3928/02793695-20050601-09. PMID 16018133.
- ^ Chatzitomaris A, Hoermann R, Midgley JE, Hering S, Urban A, Dietrich B, et al. (20 July 2017). "Thyroid Allostasis-Adaptive Responses of Thyrotropic Feedback Control to Conditions of Strain, Stress, and Developmental Programming". Frontiers in Endocrinology. 8: 163. doi:10.3389/fendo.2017.00163. PMC 5517413. PMID 28775711.
- ^ Lindley SE, Carlson EB, Benoit M (May 2004). "Basal and dexamethasone suppressed salivary cortisol concentrations in a community sample of patients with posttraumatic stress disorder". Biological Psychiatry. 55 (9): 940–5. doi:10.1016/j.biopsych.2003.12.021. PMID 15110738. S2CID 31580825.
- ^ "NIMH · Post Traumatic Stress Disorder Research Fact Sheet". National Institutes of Health. Archived from the original on 23 January 2014. Retrieved 29 January 2014.
- ^ Bromis K, Calem M, Reinders AA, Williams SC, Kempton MJ (July 2018). "Meta-Analysis of 89 Structural MRI Studies in Posttraumatic Stress Disorder and Comparison With Major Depressive Disorder". The American Journal of Psychiatry. 175 (10): 989–998. doi:10.1176/appi.ajp.2018.17111199. PMC 6169727. PMID 30021460.
- ^ Liberzon I, Sripada CS (2008). "The functional neuroanatomy of PTSD: A critical review". Stress Hormones and Post Traumatic Stress Disorder Basic Studies and Clinical Perspectives. Progress in Brain Research. Vol. 167. pp. 151–69. doi:10.1016/S0079-6123(07)67011-3. ISBN 978-0-444-53140-7. PMID 18037013.
- ^ Hughes KC, Shin LM (February 2011). "Functional neuroimaging studies of post-traumatic stress disorder". Expert Review of Neurotherapeutics. 11 (2): 275–85. doi:10.1586/ern.10.198. PMC 3142267. PMID 21306214.
- ^ Etkin A, Wager TD (October 2007). "Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia". The American Journal of Psychiatry. 164 (10): 1476–88. doi:10.1176/appi.ajp.2007.07030504. PMC 3318959. PMID 17898336.
- ^ van der Kolk B (March 2000). "Posttraumatic stress disorder and the nature of trauma". Dialogues in Clinical Neuroscience. 2 (1): 7–22. doi:10.31887/DCNS.2000.2.1/bvdkolk. PMC 3181584. PMID 22034447.
- ^ a b Milad MR, Pitman RK, Ellis CB, Gold AL, Shin LM, Lasko NB, et al. (December 2009). "Neurobiological basis of failure to recall extinction memory in posttraumatic stress disorder". Biological Psychiatry. 66 (12): 1075–82. doi:10.1016/j.biopsych.2009.06.026. PMC 2787650. PMID 19748076.
- ^ Stein MB, Paulus MP (December 2009). "Imbalance of approach and avoidance: the yin and yang of anxiety disorders". Biological Psychiatry. 66 (12): 1072–4. doi:10.1016/j.biopsych.2009.09.023. PMC 2825567. PMID 19944792.
- ^ Goodkind M, Etkin A. "Functional Neurocircuitry and Neuroimaging Studies of Anxiety Disorders". In Sklar P, Buxbaum J, Nestler E, Charney D (eds.). Neurobiology of Mental Illness (5th ed.). Oxford University Press.
- ^ Carlson NR (2007). Physiology of Behavior (9th ed.). Pearson Education.
- ^ Jatzko A, Rothenhöfer S, Schmitt A, Gaser C, Demirakca T, Weber-Fahr W, et al. (August 2006). "Hippocampal volume in chronic posttraumatic stress disorder (PTSD): MRI study using two different evaluation methods" (PDF). Journal of Affective Disorders. 94 (1–3): 121–6. doi:10.1016/j.jad.2006.03.010. PMID 16701903. Archived (PDF) from the original on 19 October 2013.
- ^ Neumeister A, Seidel J, Ragen BJ, Pietrzak RH (January 2015). "Translational evidence for a role of endocannabinoids in the etiology and treatment of posttraumatic stress disorder". Psychoneuroendocrinology. 51: 577–84. doi:10.1016/j.psyneuen.2014.10.012. PMC 4268027. PMID 25456347.
- ^ Shura RD, Epstein EL, Ord AS, Martindale SL, Rowland JA, Brearly TW, et al. (September 2020). "Relationship between intelligence and posttraumatic stress disorder in veterans". Intelligence. 82: 101472. doi:10.1016/j.intell.2020.101472. ISSN 0160-2896.
- ^ Boskovic I, Merckelbach H (2018). "Fake Posttraumatic Stress Disorder (PTSD) Costs Real Money". The Inquisitive Mind. 4 (36). Retrieved 30 September 2021.
- ^ First MB (2013). DSM-5® Handbook of Differential Diagnosis. American Psychiatric Pub. p. 225. ISBN 978-1-58562-998-5 – via Google Books.
- ^ Brady K (October 2004). "Substance Abuse and Posttraumatic Stress Disorder". Current Directions in Psychological Science. 13 (5): 206–209. doi:10.1111/j.0963-7214.2004.00309.x. S2CID 145248041.
- ^ "Trauma- and Stressor-Related Disorders", Diagnostic and Statistical Manual of Mental Disorders, American Psychiatric Association, 22 May 2013, doi:10.1176/appi.books.9780890425596.dsm07, ISBN 978-0-89042-555-8, S2CID 148757484
- ^ "PTSD Checklist for DSM-5 (PCL-5)". National Center for PTSD. 11 May 2017.
- ^ Blevins CA, Weathers FW, Davis MT, Witte TK, Domino JL (December 2015). "The Posttraumatic Stress Disorder Checklist for DSM-5 (PCL-5): Development and Initial Psychometric Evaluation". Journal of Traumatic Stress. 28 (6): 489–98. doi:10.1002/jts.22059. PMID 26606250.
- ^ "Primary Care PTSD Screen for DSM-5 (PC-PTSD-5)". National Center for PTSD. 7 April 2017.
- ^ "Child PTSD Symptom Scale". International Society for Traumatic Stress Studies. Archived from the original on 6 October 2019. Retrieved 14 December 2017.
- ^ Foa EB, Johnson KM, Feeny NC, Treadwell KR (September 2001). "The child PTSD Symptom Scale: a preliminary examination of its psychometric properties". Journal of Clinical Child Psychology. 30 (3): 376–84. doi:10.1207/S15374424JCCP3003_9. PMID 11501254. S2CID 9334984.
- ^ "Child Trauma Screening Questionnaire". The National Child Traumatic Stress Network. 5 September 2013. Archived from the original on 25 December 2017. Retrieved 14 December 2017.
- ^ Kenardy JA, Spence SH, Macleod AC (September 2006). "Screening for posttraumatic stress disorder in children after accidental injury". Pediatrics. 118 (3): 1002–9. doi:10.1542/peds.2006-0406. PMID 16950991. S2CID 1320859.
- ^ "UCLA Posttraumatic Stress Disorder Reaction Index". International Society for Traumatic Stress Studies. Archived from the original on 6 October 2019. Retrieved 14 December 2017.
- ^ Elhai JD, Layne CM, Steinberg AM, Brymer MJ, Briggs EC, Ostrowski SA, et al. (February 2013). "Psychometric properties of the UCLA PTSD reaction index. part II: investigating factor structure findings in a national clinic-referred youth sample". Journal of Traumatic Stress. 26 (1): 10–8. doi:10.1002/jts.21755. PMID 23417874.
- ^ a b Scheeringa M. "Young Child PTSD Screen". Tulane University. Archived from the original on 29 August 2021. Retrieved 8 April 2018.
- ^ Scheeringa MS, Haslett N (June 2010). "The reliability and criterion validity of the Diagnostic Infant and Preschool Assessment: a new diagnostic instrument for young children". Child Psychiatry and Human Development. 41 (3): 299–312. doi:10.1007/s10578-009-0169-2. PMC 2862973. PMID 20052532.
- ^ Bovin MJ, Marx BP, Schnurr PP (2015). "Evolving DSM Diagnostic Criteria for PTSD: Relevance for Assessment and Treatment". Current Treatment Options in Psychiatry. 2 (1): 86–98. doi:10.1007/s40501-015-0032-y.
... the use of a multi-measure approach eliminates the bias associated with any given instrument ....
- ^ Ben Barnes J, Presseau C, Jordan AH, Kline NK, Young-McCaughan S, Keane TM, et al. (May 2019). "Common Data Elements in the Assessment of Military-Related PTSD Research Applied in the Consortium to Alleviate PTSD". Military Medicine. 184 (5–6): e218–e226. doi:10.1093/milmed/usy226. PMID 30252077.
- ^ Weathers FW, Keane TM, Foa EB (2009). "Assessment and Diagnosis of Adults". In Foa EB, Keane TM, Friedman MJ (eds.). Effective Treatments for PTSD: Practice Guidelines from the International Society for Traumatic Stress Studies (2nd ed.). New York: Guilford. pp. 23–61.
"Thus, ample resources are now available to conduct psychometrically sound assessments of trauma survivors in any context, and it is no longer defensible for clinicians to do otherwise.
- ^ Weathers FW, Marx BP, Friedman MJ, Schnurr PP (2014). "Posttraumatic Stress Disorder in DSM-5: New Criteria, New Measures, and Implications for Assessment". Psychological Injury and Law. 7 (2): 93–107. doi:10.1007/s12207-014-9191-1. S2CID 16911948.
- ^ Weathers FW, Bovin MJ, Lee DJ, Sloan DM, Schnurr PP, Kaloupek DG, et al. (March 2018). "The Clinician-Administered PTSD Scale for DSM-5 (CAPS-5): Development and initial psychometric evaluation in military veterans". Psychological Assessment. 30 (3): 383–395. doi:10.1037/pas0000486. PMC 5805662. PMID 28493729.
- ^ Karimi M, Rahnejat AM, Dabaghi P, Taghva A, Majdian M, Donyavi V, et al. (2020). "The Psychometric Properties of the Post-Traumatic Stress Disorder Symptom Scale–Interview Based on DSM-5, in Military Personnel Participated in Warfare". Iranian Journal of War & Public Health. 12 (2): 115–124. doi:10.29252/ijwph.12.2.115. S2CID 235026213.
- ^ Foa EB, Tolin DF (April 2000). "Comparison of the PTSD Symptom Scale-Interview Version and the Clinician-Administered PTSD scale". Journal of Traumatic Stress. 13 (2): 181–191. doi:10.1023/A:1007781909213. PMID 10838669. S2CID 7913088.
- ^ First MB, Williams JB, Karg RS, Spitzer RL (2016). Structured Clinical Interview for DSM-5 Disorders, Clinician Version (SCID-5-CV). Arlington, VA: American Psychiatric Association. ISBN 978-88-6030-885-6.
- ^ a b "The ICD-10 Classification of Mental and Behavioural Disorders" (PDF). World Health Organization. pp. 120–121. Archived (PDF) from the original on 23 March 2014. Retrieved 29 January 2014.
- ^ "WHO releases new International Classification of Diseases (ICD 11)". World Health Organization. Retrieved 15 November 2018.
- ^ a b c d Brewin CR, Cloitre M, Hyland P, Shevlin M, Maercker A, Bryant RA, et al. (December 2017). "A review of current evidence regarding the ICD-11 proposals for diagnosing PTSD and complex PTSD" (PDF). Clinical Psychology Review. 58: 1–15. doi:10.1016/j.cpr.2017.09.001. PMID 29029837. S2CID 4874961.
- ^ Herman JL (July 1992). "Complex PTSD: A syndrome in survivors of prolonged and repeated trauma". Journal of Traumatic Stress. 5 (3): 377–391. doi:10.1007/BF00977235. S2CID 189943097.
- ^ Herman JL (1997). Trauma and Recovery (2nd ed.). New York: Basic Books. pp. 119–122. ISBN 978-0-465-08730-3.
- ^ Carlier IV, Lamberts RD, van Uchelen AJ, Gersons BP (1998). "Disaster-related post-traumatic stress in police officers: A field study of the impact of debriefing". Stress Medicine. 14 (3): 143–8. doi:10.1002/(SICI)1099-1700(199807)14:3<143::AID-SMI770>3.0.CO;2-S.
- ^ Mayou RA, Ehlers A, Hobbs M (June 2000). "Psychological debriefing for road traffic accident victims. Three-year follow-up of a randomised controlled trial". The British Journal of Psychiatry. 176 (6): 589–93. doi:10.1192/bjp.176.6.589. PMID 10974967.
- ^ a b Roberts NP, Kitchiner NJ, Kenardy J, Robertson L, Lewis C, Bisson JI (August 2019). "Multiple session early psychological interventions for the prevention of post-traumatic stress disorder". The Cochrane Database of Systematic Reviews. 8 (8): CD006869. doi:10.1002/14651858.CD006869.pub3. PMC 6699654. PMID 31425615.
- ^ Assessment and Management of Conditions Specifically Related to Stress (PDF). Geneva: World Health Organization. 2013. ISBN 978-92-4-150593-2. Archived (PDF) from the original on 1 February 2014. Retrieved 29 January 2014.
- ^ Amos T, Stein DJ, Ipser JC (July 2014). "Pharmacological interventions for preventing post-traumatic stress disorder (PTSD)". The Cochrane Database of Systematic Reviews. 7 (7): CD006239. doi:10.1002/14651858.CD006239.pub2. PMID 25001071.
- ^ Bertolini F, Robertson L, Bisson JI, Meader N, Churchill R, Ostuzzi G, et al. (10 February 2022). Cochrane Common Mental Disorders Group (ed.). "Early pharmacological interventions for universal prevention of post-traumatic stress disorder (PTSD)". Cochrane Database of Systematic Reviews. 2022 (2): CD013443. doi:10.1002/14651858.CD013443.pub2. PMC 8829470. PMID 35141873.
- ^ a b Gartlehner G, Forneris CA, Brownley KA, Gaynes BN, Sonis J, Coker-Schwimmer E, et al. (2013). "Discussion". Interventions for the Prevention of Posttraumatic Stress Disorder (PTSD) in Adults After Exposure to Psychological Trauma. Agency for Healthcare Research and Quality (US). PMID 23658936.
- ^ Feldner MT, Monson CM, Friedman MJ (January 2007). "A critical analysis of approaches to targeted PTSD prevention: current status and theoretically derived future directions". Behavior Modification. 31 (1): 80–116. CiteSeerX 10.1.1.595.9186. doi:10.1177/0145445506295057. PMID 17179532. S2CID 44619491.
- ^ Rose S, Bisson J, Churchill R, Wessely S (2002). "Psychological debriefing for preventing post traumatic stress disorder (PTSD)". The Cochrane Database of Systematic Reviews (2): CD000560. doi:10.1002/14651858.CD000560. PMC 7032695. PMID 12076399.
- ^ "Psychological Debriefing for Post-Traumatic Stress Disorder". www.div12.org. Society of Clinical Psychology: Division 12 of The American Psychological Association. 19 August 2014. Retrieved 9 September 2017.
- ^ Birur B, Moore NC, Davis LL (February 2017). "An Evidence-Based Review of Early Intervention and Prevention of Posttraumatic Stress Disorder". Community Mental Health Journal. 53 (2): 183–201. doi:10.1007/s10597-016-0047-x. PMID 27470261. S2CID 28150745.
- ^ Litz B (1 December 2015). "Early intervention for trauma and loss: overview and working care model". European Journal of Psychotraumatology. 6 (1): 28543. doi:10.3402/ejpt.v6.28543. PMC 4466306. PMID 26073207.
- ^ Wiseman T, Foster K, Curtis K (November 2013). "Mental health following traumatic physical injury: an integrative literature review". Injury. 44 (11): 1383–90. doi:10.1016/j.injury.2012.02.015. PMID 22409991.
- ^ Kassam-Adams N, Marsac ML, Hildenbrand A, Winston F (December 2013). "Posttraumatic stress following pediatric injury: update on diagnosis, risk factors, and intervention". JAMA Pediatrics. 167 (12): 1158–65. doi:10.1001/jamapediatrics.2013.2741. PMID 24100470.
- ^ Powers MB, Halpern JM, Ferenschak MP, Gillihan SJ, Foa EB (August 2010). "A meta-analytic review of prolonged exposure for posttraumatic stress disorder". Clinical Psychology Review. 30 (6): 635–41. doi:10.1016/j.cpr.2010.04.007. PMID 20546985.
- ^ Guideline Development Panel for the Treatment of PTSD in Adults (2017). Clinical Practice Guideline for the Treatment of Posttraumatic Stress Disorder (PTSD) in Adults (PDF). Washington, D.C.: American Psychological Association. pp. ES–2.
- ^ a b Lee CW, Cuijpers P (June 2013). "A meta-analysis of the contribution of eye movements in processing emotional memories". Journal of Behavior Therapy and Experimental Psychiatry (Submitted manuscript). 44 (2): 231–9. doi:10.1016/j.jbtep.2012.11.001. PMID 23266601.
- ^ Cahill SP, Foa EB (2004). Taylor S (ed.). Advances in the Treatment of Posttraumatic Stress Disorder: Cognitive-behavioral perspectives. New York: Springer. pp. 267–313.
- ^ Sloan DM, Marx BP (2019). Written exposure therapy for PTSD: A brief treatment approach for mental health professionals. Washington: American Psychological Association. doi:10.1037/0000139-001. ISBN 978-1-4338-3013-6. S2CID 239337813. Retrieved 13 February 2022.
- ^ VA/DOD Clinical Practice Guideline for the Management of Posttraumatic Stress Disorder and Acute Stress Disorder (PDF). United States Department of Veterans Affairs. 2017. pp. 46–47.
- ^ a b Suomi A, Evans L, Rodgers B, Taplin S, Cowlishaw S (December 2019). "Couple and family therapies for post-traumatic stress disorder (PTSD)". The Cochrane Database of Systematic Reviews. 2019 (12): CD011257. doi:10.1002/14651858.CD011257.pub2. PMC 6890534. PMID 31797352.
- ^ Seidler GH, Wagner FE (November 2006). "Comparing the efficacy of EMDR and trauma-focused cognitive-behavioral therapy in the treatment of PTSD: a meta-analytic study". Psychological Medicine. 36 (11): 1515–22. doi:10.1017/S0033291706007963. PMID 16740177. S2CID 39751799.
- ^ a b Moreno-Alcázar A, Treen D, Valiente-Gómez A, Sio-Eroles A, Pérez V, Amann BL, et al. (2017). "Efficacy of Eye Movement Desensitization and Reprocessing in Children and Adolescent with Post-traumatic Stress Disorder: A Meta-Analysis of Randomized Controlled Trials". Frontiers in Psychology. 8: 1750. doi:10.3389/fpsyg.2017.01750. PMC 5641384. PMID 29066991.
- ^ Rolfsnes ES, Idsoe T (April 2011). "School-based intervention programs for PTSD symptoms: a review and meta-analysis". Journal of Traumatic Stress. 24 (2): 155–65. doi:10.1002/jts.20622. PMID 21425191.
- ^ a b Forman-Hoffman V, Middleton JC, Feltner C, Gaynes BN, Weber RP, Bann C, et al. (17 May 2018). "Psychological and Pharmacological Treatments for Adults With Posttraumatic Stress Disorder: A Systematic Review Update". Agency for Healthcare Research and Quality. doi:10.23970/ahrqepccer207 (inactive 23 March 2024). PMID 30204376. Archived from the original on 10 July 2018. Retrieved 25 August 2020.
{{cite journal}}: CS1 maint: DOI inactive as of March 2024 (link) - ^ "Treatment of PTSD – PTSD: National Center for PTSD". U.S. Department of Veterans Affairs. 26 May 2016. Archived from the original on 1 December 2016.
- ^ "PTSD Treatment Options". Defense Centers of Excellence. 23 November 2016. Archived from the original on 30 November 2016.
- ^ "Cognitive Behavioral Therapy (CBT) for Treatment of PTSD". American Psychiatric Association. Archived from the original on 9 January 2018. Retrieved 8 January 2018.
- ^ "Treatment of PTSD – PTSD". National Center for PTSD. Retrieved 8 January 2018.
- ^ "Online CBT for post-traumatic stress disorder is as effective as face-to-face therapy". NIHR Evidence. 27 January 2023. doi:10.3310/nihrevidence_56507. S2CID 257844874.
- ^ Bisson JI, Ariti C, Cullen K, Kitchiner N, Lewis C, Roberts NP, et al. (June 2022). "Guided, internet based, cognitive behavioural therapy for post-traumatic stress disorder: pragmatic, multicentre, randomised controlled non-inferiority trial (RAPID)". BMJ. 377: e069405. doi:10.1136/bmj-2021-069405. PMC 9202033. PMID 35710124.
- ^ Simon N, Robertson L, Lewis C, Roberts NP, Bethell A, Dawson S, et al. (May 2021). "Internet-based cognitive and behavioural therapies for post-traumatic stress disorder (PTSD) in adults". The Cochrane Database of Systematic Reviews. 2021 (5): CD011710. doi:10.1002/14651858.CD011710.pub3. PMC 8136365. PMID 34015141.
- ^ Grohol JM (17 May 2016). "What Is Exposure Therapy?". Psychcentral.com. Archived from the original on 11 August 2010. Retrieved 14 July 2010.
- ^ McLean CP, Levy HC, Miller ML, Tolin DF (February 2022). "Exposure therapy for PTSD: A meta-analysis". Clinical Psychology Review. 91: 102115. doi:10.1016/j.cpr.2021.102115. PMID 34954460. S2CID 245394152.
- ^ Ursano RJ, Bell C, Eth S, Friedman M, Norwood A, Pfefferbaum B, et al. (November 2004). "Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder". The American Journal of Psychiatry. 161 (11 Suppl): 3–31. PMID 15617511.
- ^ Committee on Treatment of Posttraumatic Stress Disorder, Institute of Medicine: Treatment of Posttraumatic Stress Disorder: An Assessment of the Evidence. Washington, D.C.: National Academies Press. 2008. ISBN 978-0-309-10926-0.[page needed]
- ^ "Prolonged Exposure Therapy". PTSD: National Center for PTSD. U.S. Department of Veteran Affairs. 29 September 2009. Archived from the original on 14 November 2009. Retrieved 14 July 2010.
- ^ Karlin BE, Ruzek JI, Chard KM, Eftekhari A, Monson CM, Hembree EA, et al. (December 2010). "Dissemination of evidence-based psychological treatments for posttraumatic stress disorder in the Veterans Health Administration". Journal of Traumatic Stress. 23 (6): 663–73. doi:10.1002/jts.20588. PMID 21171126.
- ^ Mulick PS, Landes S, Kanter JW (2005). "Contextual Behavior Therapies in the Treatment of PTSD: A Review" (PDF). International Journal of Behavioral Consultation and Therapy. 1 (3): 223–228. CiteSeerX 10.1.1.625.4407. doi:10.1037/h0100747. Archived from the original (PDF) on 16 September 2012.
- ^ Hassija CM, Gray MJ (2007). "Behavioral Interventions for Trauma and Posttraumatic Stress Disorder". International Journal of Behavioral Consultation and Therapy. 3 (2): 166–175. doi:10.1037/h0100797.
- ^ Mulick PS, Naugle AE (2009). "Behavioral Activation in the Treatment of Comorbid Posttraumatic Stress Disorder and Major Depressive Disorder". International Journal of Behavioral Consultation and Therapy. 5 (2): 330–339. doi:10.1037/h0100892.
- ^ a b Shapiro F (April 1989). "Efficacy of the eye movement desensitization procedure in the treatment of traumatic memories". Journal of Traumatic Stress. 2 (2): 199–223. doi:10.1002/jts.2490020207.
- ^ Shapiro F, Maxfield L (August 2002). "Eye Movement Desensitization and Reprocessing (EMDR): information processing in the treatment of trauma". Journal of Clinical Psychology. 58 (8): 933–46. doi:10.1002/jclp.10068. PMID 12115716.
- ^ a b The Management of Post-Traumatic Stress Working Group (2010). "VA/DoD clinical practice guideline for management of post-traumatic stress". Department of Veterans Affairs, Department of Defense. p. Version 2.0. Archived from the original on 30 May 2013. Retrieved 2 June 2013.
- ^ a b c Gillies D, Taylor F, Gray C, O'Brien L, D'Abrew N (December 2012). "Psychological therapies for the treatment of post-traumatic stress disorder in children and adolescents". The Cochrane Database of Systematic Reviews. 12: CD006726. doi:10.1002/14651858.CD006726.pub2. hdl:1959.13/1311467. PMID 23235632.
- ^ a b Jeffries FW, Davis P (May 2013). "What is the role of eye movements in eye movement desensitization and reprocessing (EMDR) for post-traumatic stress disorder (PTSD)? a review". Behavioural and Cognitive Psychotherapy. 41 (3): 290–300. doi:10.1017/S1352465812000793. PMID 23102050. S2CID 33309479.
- ^ a b c d e f Jonas DE, Cusack K, Forneris CA (April 2013). Psychological and pharmacological treatments for adults with posttraumatic stress disorder (PTSD). Comparative Effectiveness Reviews No. 92. Rockville, MD: Agency for Healthcare Research and Quality. PMID 23658937. Archived from the original on 18 January 2017.
- ^ "Innovative treatment found to help post-traumatic stress sufferers". Murdoch University (Press release). 9 October 2014.
- ^ Brewin CR, Andrews B, Valentine JD (October 2000). "Meta-analysis of risk factors for posttraumatic stress disorder in trauma-exposed adults". Journal of Consulting and Clinical Psychology. 68 (5): 748–66. doi:10.1037/0022-006X.68.5.748. PMID 11068961. S2CID 13749007.
- ^ Ozer EJ, Best SR, Lipsey TL, Weiss DS (January 2003). "Predictors of posttraumatic stress disorder and symptoms in adults: a meta-analysis". Psychological Bulletin. 129 (1): 52–73. doi:10.1037/0033-2909.129.1.52. PMID 12555794.
- ^ Weissman MM, Markowitz JC, Klerman GL (2007). Clinician's Quick Guide to Interpersonal Psychotherapy. New York: Oxford University Press.[page needed]
- ^ Bleiberg KL, Markowitz JC (January 2005). "A pilot study of interpersonal psychotherapy for posttraumatic stress disorder". The American Journal of Psychiatry. 162 (1): 181–3. doi:10.1176/appi.ajp.162.1.181. PMID 15625219.
- ^ Krystal JH, Neumeister A (October 2009). "Noradrenergic and serotonergic mechanisms in the neurobiology of posttraumatic stress disorder and resilience". Brain Research. 1293: 13–23. doi:10.1016/j.brainres.2009.03.044. PMC 2761677. PMID 19332037.
- ^ a b Jeffreys M, Capehart B, Friedman MJ (2012). "Pharmacotherapy for posttraumatic stress disorder: review with clinical applications". Journal of Rehabilitation Research and Development. 49 (5): 703–715. doi:10.1682/JRRD.2011.09.0183. PMID 23015581.
While evidence-based, trauma-focused psychotherapy is the preferred treatment for PTSD, pharmacotherapy is also an important treatment option. First-line pharmacotherapy agents include selective serotonin reuptake inhibitors and the selective serotonin-norepinephrine reuptake inhibitor venlafaxine.
- ^ Williams T, Phillips NJ, Stein DJ, Ipser JC (March 2022). "Pharmacotherapy for post traumatic stress disorder (PTSD)". The Cochrane Database of Systematic Reviews. 2022 (3): CD002795. doi:10.1002/14651858.CD002795.pub3. PMC 8889888. PMID 35234292.
- ^ Puetz TW, Youngstedt SD, Herring MP (28 May 2015). Hashimoto K (ed.). "Effects of Pharmacotherapy on Combat-Related PTSD, Anxiety, and Depression: A Systematic Review and Meta-Regression Analysis". PLOS ONE. 10 (5): e0126529. Bibcode:2015PLoSO..1026529P. doi:10.1371/journal.pone.0126529. PMC 4447407. PMID 26020791.
The cumulative evidence summarized in this review indicates that pharmacotherapy significantly reduces PTSD, anxiety, and depressive symptom severity among combat veterans with PTSD. The magnitude of the overall effects of pharmacotherapy on PTSD (Δ = 0.38), anxiety (Δ = 0.42), and depressive symptoms (Δ = 0.52) were moderate...
- ^ Kapfhammer HP (June 2014). "Patient-reported outcomes in post-traumatic stress disorder. Part II: focus on pharmacological treatment". Dialogues in Clinical Neuroscience (in English, Spanish, and French). 16 (2): 227–237. doi:10.31887/DCNS.2014.16.2/hkapfhammer. PMC 4140515. PMID 25152660.
- ^ Jain S, Greenbaum MA, Rosen C (February 2012). "Concordance between psychotropic prescribing for veterans with PTSD and clinical practice guidelines". Psychiatric Services. 63 (2): 154–60. doi:10.1176/appi.ps.201100199. PMID 22302333.
- ^ Auxéméry Y (October 2012). "[Posttraumatic stress disorder (PTSD) as a consequence of the interaction between an individual genetic susceptibility, a traumatogenic event and a social context]". L'Encéphale (in French). 38 (5): 373–80. doi:10.1016/j.encep.2011.12.003. PMID 23062450.
- ^ Kapfhammer HP (December 2008). "[Therapeutic possibilities after traumatic experiences]". Psychiatria Danubina. 20 (4): 532–45. PMID 19011595.
- ^ Reist C (2005). Post-traumatic Stress Disorder. Epocrates.com.
- ^ Maxmen JS, Ward NG (2002). Psychotropic drugs: fast facts (3rd ed.). New York: W.W. Norton & Company. p. 349. ISBN 978-0-393-70301-6.
- ^ Martényi F (March 2005). "[Three paradigms in the treatment of posttraumatic stress disorder]". Neuropsychopharmacologia Hungarica. 7 (1): 11–21. PMID 16167463.
- ^ a b Veterans Affairs and Department of Defense clinical practice guideline for management of post-traumatic stress. VA/DoD. 2010.
- ^ Bandelow B, Zohar J, Hollander E, Kasper S, Möller HJ, Zohar J, et al. (2008). "World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders – first revision". The World Journal of Biological Psychiatry. 9 (4): 248–312. doi:10.1080/15622970802465807. PMID 18949648.
- ^ Green B (July 2014). "Prazosin in the treatment of PTSD". Journal of Psychiatric Practice. 20 (4): 253–9. doi:10.1097/01.pra.0000452561.98286.1e. PMID 25036580. S2CID 40069887.
- ^ Singh B, Hughes AJ, Mehta G, Erwin PJ, Parsaik AK (July 2016). "Efficacy of Prazosin in Posttraumatic Stress Disorder: A Systematic Review and Meta-Analysis". The Primary Care Companion for CNS Disorders. 18 (4). doi:10.4088/PCC.16r01943. PMID 27828694.
- ^ Griffin GD, Charron D, Al-Daccak R (November 2014). "Post-traumatic stress disorder: revisiting adrenergics, glucocorticoids, immune system effects and homeostasis". Clinical & Translational Immunology. 3 (11): e27. doi:10.1038/cti.2014.26. PMC 4255796. PMID 25505957.
- ^ Black N, Stockings E, Campbell G, Tran LT, Zagic D, Hall WD, et al. (December 2019). "Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis". The Lancet. Psychiatry. 6 (12): 995–1010. doi:10.1016/S2215-0366(19)30401-8. PMC 6949116. PMID 31672337.
- ^ O'Neil ME, Nugent SM, Morasco BJ, Freeman M, Low A, Kondo K, et al. (September 2017). "Benefits and Harms of Plant-Based Cannabis for Posttraumatic Stress Disorder: A Systematic Review". Annals of Internal Medicine. 167 (5): 332–340. doi:10.7326/M17-0477. PMID 28806794.
- ^ Betthauser K, Pilz J, Vollmer LE (August 2015). "Use and effects of cannabinoids in military veterans with posttraumatic stress disorder". American Journal of Health-System Pharmacy (Review). 72 (15): 1279–84. doi:10.2146/ajhp140523. PMID 26195653.
- ^ "Long-term Nabilone Use: A Review of the Clinical Effectiveness and Safety". CADTH Rapid Response Reports. October 2015. PMID 26561692.
- ^ Gregg K (13 July 2016). "Raimondo signs law allowing marijuana for treatment of PTSD". The Providence Journal. Archived from the original on 16 August 2016. Retrieved 18 August 2016.
- ^ Lawrence S, De Silva M, Henley R (January 2010). Lawrence S (ed.). "Sports and games for post-traumatic stress disorder (PTSD)". The Cochrane Database of Systematic Reviews. 2010 (1): CD007171. doi:10.1002/14651858.CD007171.pub2. PMC 7390394. PMID 20091620. Archived from the original on 1 February 2014.
- ^ Jankowski K. "PTSD and physical health". Information on trauma and PTSD for professionals, National Center for PTSD. U.S. Department of Veterans Affairs. Archived from the original on 30 July 2009. Retrieved 8 June 2013.
- ^ U.S. Department of Veterans Affairs. "Lifestyle Changes Recommended for PTSD Patients". Information on trauma and PTSD for veterans, general public and family from the National Center for PTSD. U.S. Department of Veterans Affairs. Archived from the original on 31 July 2009. Retrieved 8 June 2013.
- ^ a b Wethington HR, Hahn RA, Fuqua-Whitley DS, Sipe TA, Crosby AE, Johnson RL, et al. (September 2008). "The effectiveness of interventions to reduce psychological harm from traumatic events among children and adolescents: a systematic review". American Journal of Preventive Medicine. 35 (3): 287–313. doi:10.1016/j.amepre.2008.06.024. PMID 18692745. Archived from the original on 3 February 2014.
- ^ Fletcher KE, Barkley RA (2003). "7". In Mash EJ (ed.). Child psychopathology (2nd ed.). New York: Guilford Press. pp. 330–371. ISBN 978-1-57230-609-7.
- ^ "Marine Corps Offers Yoga, Massages to Marriages Strained by War". Fox News. Associated Press. 2 April 2008. Archived from the original on 5 April 2008. Retrieved 3 April 2008.
- ^ Sweeney H (6 November 2015). "Private Hospital Network to Help VA with Mental Health Care for Vets". Military.com. Archived from the original on 30 March 2017. Retrieved 29 March 2017.
- ^ Cullen K (2 May 2016). "Covering all the bases for veterans". The Boston Globe. Archived from the original on 30 March 2017. Retrieved 29 March 2017.
- ^ "FDA approves Apple Watch app NightWare to treat PTSD nightmares". Kyma. 11 November 2020.
- ^ a b c "Mortality and Burden of Disease Estimates for WHO Member States: Persons, all ages (2004)" (xls). World Health Organization. 2004. Retrieved 12 November 2009.
- ^ Brunet A, Akerib V, Birmes P (August 2007). "Don't throw out the baby with the bathwater (PTSD is not overdiagnosed)". Canadian Journal of Psychiatry. 52 (8): 501–2, discussion 503. doi:10.1177/070674370705200805. PMID 17955912.
- ^ Kilpatrick DG, Resnick HS, Milanak ME, Miller MW, Keyes KM, Friedman MJ (October 2013). "National estimates of exposure to traumatic events and PTSD prevalence using DSM-IV and DSM-5 criteria". Journal of Traumatic Stress. 26 (5): 537–47. doi:10.1002/jts.21848. PMC 4096796. PMID 24151000.
- ^ a b "Mortality and Burden of Disease Estimates for WHO Member States: Females, all ages (2004)" (xls). World Health Organization. 2004. Retrieved 12 November 2009.
- ^ a b "Mortality and Burden of Disease Estimates for WHO Member States: Males, all ages (2004)" (xls). World Health Organization. 2004. Retrieved 12 November 2009.
- ^ a b Koenen KC, Ratanatharathorn A, Ng L, McLaughlin KA, Bromet EJ, Stein DJ, et al. (October 2017). "Posttraumatic stress disorder in the World Mental Health Surveys". Psychological Medicine. 47 (13): 2260–2274. doi:10.1017/S0033291717000708. PMC 6034513. PMID 28385165.
- ^ Nuwer R (14 September 2023). "MDMA Therapy for PTSD Inches Closer to U.S. Approval". The New York Times. New York Times.
- ^ Sher L (August 2010). "Neurobiology of suicidal behavior in post-traumatic stress disorder". Expert Review of Neurotherapeutics. 10 (8): 1233–5. doi:10.1586/ern.10.114. PMID 20662745. S2CID 5900319.
- ^ Mintz S (2007). "The War's Costs". Digital History. Archived from the original on 7 September 2003.
- ^ Price JL. "Findings from the National Vietnam Veterans' Readjustment Study – Factsheet". United States Department of Veterans Affairs. National Center for PTSD. Archived from the original on 30 April 2009.
- ^ a b "Psychological Costs of War: Military Combat and Mental Health". Journalistsresource.org. 27 February 2012. Archived from the original on 2 February 2014. Retrieved 29 January 2014.
- ^ Spoont M, Arbisi P, Fu S, Greer N, Kehle-Forbes S, Meis L, et al. (January 2013). Screening for Post-Traumatic Stress Disorder (PTSD) in Primary Care: A Systematic Review. VA Evidence-based Synthesis Program Reports. Department of Veterans Affairs. PMID 23487872.
- ^ Meade BJ, Glenn MK, Wirth O (29 March 2013). "Mission Critical: Getting Vets With PTSD Back to Work". NIOSH: Workplace Safety and Health. Medscape & NIOSH. Archived from the original on 16 March 2016.
- ^ a b c d e f Lowell A, Suarez-Jimenez B, Helpman L, Zhu X, Durosky A, Hilburn A, et al. (March 2018). "9/11-related PTSD among highly exposed populations: a systematic review 15 years after the attack". Psychological Medicine. 48 (4): 537–553. doi:10.1017/S0033291717002033. PMC 5805615. PMID 28805168.
- ^ a b c d Perrin MA, DiGrande L, Wheeler K, Thorpe L, Farfel M, Brackbill R (September 2007). "Differences in PTSD prevalence and associated risk factors among World Trade Center disaster rescue and recovery workers". The American Journal of Psychiatry. 164 (9): 1385–94. doi:10.1176/appi.ajp.2007.06101645. PMID 17728424. S2CID 22476443.
- ^ a b c d e Stellman JM, Smith RP, Katz CL, Sharma V, Charney DS, Herbert R, et al. (September 2008). "Enduring mental health morbidity and social function impairment in world trade center rescue, recovery, and cleanup workers: the psychological dimension of an environmental health disaster". Environmental Health Perspectives. 116 (9): 1248–53. doi:10.1289/ehp.11164. PMC 2535630. PMID 18795171.
- ^ a b c d e Moghimi Y (2012). "Anthropological discourses on the globalization of posttraumatic stress disorder (PTSD) in post-conflict societies". Journal of Psychiatric Practice. 18 (1): 29–37. doi:10.1097/01.pra.0000410985.53970.3b. PMID 22261981. S2CID 36228060.
- ^ Pillen A (2016). "Language, Translation, Trauma". Annual Review of Anthropology. 45: 95–111. doi:10.1146/annurev-anthro-102215-100232.
- ^ Skerrett DM (2010). "Can the Sapir-Whorf hypothesis save the planet? lessons from cross-cultural psychology for critical language policy". Current Issues in Language Planning. 11 (4): 331–340. doi:10.1080/14664208.2010.534236. S2CID 144639205.
- ^ a b Kohrt BA, Hrushka DJ (2010). "Nepali concepts of psychological trauma: the role of idioms of distress, ethnopsychology and ethnophysiology in alleviating suffering and preventing stigma". Culture, Medicine and Psychiatry. 34 (2): 322–352. doi:10.1007/s11013-010-9170-2. PMC 3819627. PMID 20309724.
- ^ Pedersen D, Tremblay J, Errázuriz C, Gamarra J (July 2008). "The sequelae of political violence: assessing trauma, suffering and dislocation in the Peruvian highlands". Social Science & Medicine. 67 (2): 205–217. doi:10.1016/j.socscimed.2008.03.040. PMID 18423959.
- ^ a b Maedel A, Schauer E, Odenwald M, Elbert T (2010). Psychological Rehabilitation of Ex-combatants in Non-Western, Post-conflict Settings, Trauma Rehabilitation After War and Conflict. New York, NY: Springer. doi:10.1007/978-1-4419-5722-1_9.
- ^ Crocq MA, Crocq L (March 2000). "From shell shock and war neurosis to posttraumatic stress disorder: a history of psychotraumatology". Dialogues in Clinical Neuroscience. 2 (1): 47–55. doi:10.31887/DCNS.2000.2.1/macrocq. PMC 3181586. PMID 22033462. S2CID 24931891.
- ^ Regier DA, Kuhl EA, Kupfer DJ (June 2013). "The DSM-5: Classification and criteria changes". World Psychiatry. 12 (2): 92–98. doi:10.1002/wps.20050. PMC 3683251. PMID 23737408.
- ^ a b c Herbert JD, Forman EM (2010). "Cross-cultural perspectives on posttraumatic stress". Clinician's Guide to Posttraumatic Stress Disorder. pp. 235–261. doi:10.1002/9781118269961.ch10. ISBN 978-0-470-45095-6.
- ^ "VA Compensation Rate Table". Department of Veterans Affairs. Archived from the original on 3 November 2012. Retrieved 20 October 2012.
- ^ "Access VA Health Benefits". Department of Veterans Affairs. Archived from the original on 16 October 2012. Retrieved 20 October 2012.
- ^ "VA Vocational Rehabilitation". Department of Veterans Affairs. Archived from the original on 19 October 2012. Retrieved 20 October 2012.
- ^ "Vet Success". Department of Veterans Affairs + State Government Veterans Agencies. Archived from the original on 19 October 2012. Retrieved 20 October 2012.
- ^ "Independent Living Support for Veterans". Department of Veterans Affairs. Archived from the original on 24 October 2012. Retrieved 20 October 2012.
- ^ "Veterans Benefits". Veterans Benefits Administration. Archived from the original on 26 November 2012. Retrieved 30 November 2012.
- ^ Dixon L (28 February 2009). "Lance Corporal Johnson Beharry accuses Government of neglecting soldiers". The Times. London. Retrieved 29 August 2009. (subscription required)
- ^ "UK | Full interview: L/Cpl Johnson Beharry". BBC News. 28 February 2009. Archived from the original on 19 February 2014. Retrieved 29 August 2009.
- ^ "The Operational Stress Injury Social Support (OSISS) Program for Canadian Veterans". Archived from the original on 6 July 2011. See also "Evaluation of the OSISS Peer Support Network" (PDF). Dept. of National Defence and Veterans Affairs Canada. January 2005. Archived (PDF) from the original on 30 January 2014.
- ^ Heber A, Grenier S, Richardson D, Darte K (2006). "Combining Clinical Treatment and Peer Support: A Unique Approach to Overcoming Stigma and Delivering Care". Human Dimensions in Military Operations – Military Leaders' Strategies for Addressing Stress and Psychological Support. Neuilly-sur-Seine, France: Canadian Department Of National Defence. Archived from the original (PDF) on 7 October 2012. Retrieved 30 January 2014.
- ^ Richardson JD, Darte K, Grenier S, English A, Sharpe J (2008). "Operational Stress Injury Social Support: a Canadian innovation in professional peer support". Canadian Military Journal. 9 (1): 57–64. Archived from the original on 21 December 2013. Retrieved 30 January 2014.
- ^ "The New Veterans Charter for CF Veterans and their Families". Vac-Acc.Gc.Ca. 12 July 2006. Archived from the original on 19 June 2006. Retrieved 29 August 2009.
- ^ "Ancient Assyrian Soldiers Were Haunted by War, Too".
- ^ Shay J (2000). "Killing rage: physis or nomos—or both". War and Violence in Ancient Greece. Duckworth and the Classical Press of Wales. pp. 31–56. ISBN 0-7156-3046-6.
- ^ "Henry IV, Part I, Act II, Scene 3". Open Source Shakespeare. Archived from the original on 27 March 2014. Retrieved 30 January 2014.
- ^ Shay J (1994). Achilles in Vietnam: Combat Trauma and the Undoing of Character. Scribner. pp. 165–66.
- ^ Möbius S (2015). "Im Kugelhagel der Musketen" [In the hail of muskets]. Damals (in German). Vol. 47, no. 12. pp. 64–69.
- ^ Andreasen NC (19 February 2004). Brave New Brain: Conquering Mental Illness in the Era of the Genome. New York: Oxford University Press. p. 303. ISBN 978-0-19-516728-3.
- ^ Jones JA (2013). "From Nostalgia to Post-Traumatic Stress Disorder: A Mass Society Theory of Psychological Reactions to Combat". Inquiries Journal. 5 (2): 1–3. Archived from the original on 17 February 2014.
- ^ "World War One – A New Kind of War, Part II". www.ralphmag.org. Archived from the original on 3 March 2016., From 14 – 18 Understanding the Great War, by Stéphane Audoin-Rouzeau, Annette Becker[incomplete short citation]
- ^ a b Andreasen NC (October 2010). "Posttraumatic stress disorder: a history and a critique". Annals of the New York Academy of Sciences. 1208 (Psychiatric and Neurologic Aspects of War): 67–71. Bibcode:2010NYASA1208...67A. doi:10.1111/j.1749-6632.2010.05699.x. PMID 20955327. S2CID 42645212.
- ^ a b American Psychiatric Association (1952). Diagnostic and Statistical Manual. American Psychiatric Association Mental Hospital Service. p. 326.3. ISBN 978-0-89042-017-1.
- ^ "When trauma tips you over: PTSD Part 1". All in the Mind. Australian Broadcasting Commission. 9 October 2004. Archived from the original on 3 June 2008.
- ^ Holmstrom LL, Burgess AW (1978). The Victim of Rape: Institutional Reactions. Wiley-Interscience. ISBN 978-0-471-40785-0.
- ^ a b Shalev AY, Yehuda R, McFarlane AC (2000). International handbook of human response to trauma. New York: Kluwer Academic/Plenum Press. ISBN 978-0-306-46095-1. Archived from the original on 17 June 2007.[page needed]
- ^ Jones DR, Fischer JR (1 April 1982). "Emotional Effects on USAF Personnel of Recovering and Identifying Victims from Jonestown, Guyana" (PDF). Archived (PDF) from the original on 8 June 2019.
- ^ Scrignar CB (2 August 1999). "PTSD, the Traumatic Principle and Lawsuits". Psychiatric Times. Retrieved 25 June 2018.
- ^ "International Statistical Classification of Diseases and Related Health Problems 10th Revision Version for 2007". World Health Organization. 2007. Archived from the original on 5 December 2014. Retrieved 3 October 2011.
- ^ "Civilian PTSD Symptoms and Risk for Involvement in the Criminal Justice System". Journal of the Academy of Psychiatry and the Law. 40 (4): 522–529. 1 December 2012. ISSN 1093-6793. Retrieved 29 November 2014.
- ^ "Search results: 'post-traumatic stress disorder' in the title of a journal article". U.S. National Library of Medicine. Archived from the original on 14 May 2016. Retrieved 21 January 2015.
- ^ "PTSD". TheFreeDictionary.com. Farlex, Inc. Retrieved 21 January 2015.
- ^ Righy C, Rosa RG, da Silva RT, Kochhann R, Migliavaca CB, Robinson CC, et al. (June 2019). "Prevalence of post-traumatic stress disorder symptoms in adult critical care survivors: a systematic review and meta-analysis". Critical Care. 23 (1): 213. doi:10.1186/s13054-019-2489-3. PMC 6560853. PMID 31186070.
- ^ Thompson M (2011). "The Disappearing 'Disorder': Why PTSD is becoming PTS". Time. Retrieved 3 October 2018.
- ^ Peters M (19 May 2017). "George Carlin: Euphemism Fighter Supreme". McSweeny's. Retrieved 3 April 2019.
- ^ Fodor KE, Unterhitzenberger J, Chou CY, Kartal D, Leistner S, Milosavljevic M, et al. (2014). "Is traumatic stress research global? A bibliometric analysis". European Journal of Psychotraumatology. 5 (1): 23269. doi:10.3402/ejpt.v5.23269. PMC 3930940. PMID 24563730.
- ^ a b Ojo JO, Greenberg MB, Leary P, Mouzon B, Bachmeier C, Mullan M, et al. (December 2014). "Neurobehavioral, neuropathological and biochemical profiles in a novel mouse model of co-morbid post-traumatic stress disorder and mild traumatic brain injury". Frontiers in Behavioral Neuroscience. 8: 213. doi:10.3389/fnbeh.2014.00213. PMC 4067099. PMID 25002839.
- ^ Poulos AM, Reger M, Mehta N, Zhuravka I, Sterlace SS, Gannam C, et al. (August 2014). "Amnesia for early life stress does not preclude the adult development of posttraumatic stress disorder symptoms in rats". Biological Psychiatry. 76 (4): 306–14. doi:10.1016/j.biopsych.2013.10.007. PMC 3984614. PMID 24231200.
- ^ Piraccini E, Munakomi S, Chang KV (2020). "Stellate Ganglion Blocks". StatPearls. StatPearls Publishing LLC. PMID 29939575.
- ^ Hill MN, Patel S (October 2013). "Translational evidence for the involvement of the endocannabinoid system in stress-related psychiatric illnesses". Biology of Mood & Anxiety Disorders. 3 (1): 19. doi:10.1186/2045-5380-3-19. PMC 3817535. PMID 24286185.
- ^ Feldwisch-Drentrup H (July 2002). "New clues to why a French drug trial went horribly wrong". Science. Retrieved 11 December 2019.
- ^ Mitchell JM, Bogenschutz M, Lilienstein A, Harrison C, Kleiman S, Parker-Guilbert K, et al. (June 2021). "MDMA-assisted therapy for severe PTSD: a randomized, double-blind, placebo-controlled phase 3 study". Nature Medicine. 27 (6): 1025–1033. doi:10.1038/s41591-021-01336-3. PMC 8205851. PMID 33972795.
- ^ Singleton SP, Wang JB, Mithoefer M, Hanlon C, George MS, Mithoefer A, et al. (2023). "Altered brain activity and functional connectivity after MDMA-assisted therapy for post-traumatic stress disorder". Frontiers in Psychiatry. 13: 947622. doi:10.3389/fpsyt.2022.947622. PMC 9879604. PMID 36713926.
- ^ Muthukumaraswamy SD, Forsyth A, Lumley T (September 2021). "Blinding and expectancy confounds in psychedelic randomized controlled trials". Expert Review of Clinical Pharmacology. 14 (9): 1133–1152. doi:10.1080/17512433.2021.1933434. PMID 34038314. S2CID 235215630.
- ^ Burke MJ, Blumberger DM (October 2021). "Caution at psychiatry's psychedelic frontier". Nature Medicine. 27 (10): 1687–1688. doi:10.1038/s41591-021-01524-1. PMID 34635858. S2CID 238635462.
- ^ Halvorsen JØ, Naudet F, Cristea IA (October 2021). "Challenges with benchmarking of MDMA-assisted psychotherapy" (PDF). Nature Medicine. 27 (10): 1689–1690. doi:10.1038/s41591-021-01525-0. PMID 34635857. S2CID 238636360.
- ^ a b Straud CL, Siev J, Messer S, Zalta AK (October 2019). "Examining military population and trauma type as moderators of treatment outcome for first-line psychotherapies for PTSD: A meta-analysis". Journal of Anxiety Disorders. 67: 102133. doi:10.1016/j.janxdis.2019.102133. PMC 6739153. PMID 31472332.
- ^ Hamblen JL, Norman SB, Sonis JH, Phelps AJ, Bisson JI, Nunes VD, et al. (September 2019). "A guide to guidelines for the treatment of posttraumatic stress disorder in adults: An update" (PDF). Psychotherapy. 56 (3): 359–373. doi:10.1037/pst0000231. PMID 31282712. S2CID 195829939.
- ^ Kline AC, Cooper AA, Rytwinksi NK, Feeny NC (February 2018). "Long-term efficacy of psychotherapy for posttraumatic stress disorder: A meta-analysis of randomized controlled trials". Clinical Psychology Review. 59: 30–40. doi:10.1016/j.cpr.2017.10.009. PMC 5741501. PMID 29169664.
- ^ Goetter EM, Bui E, Ojserkis RA, Zakarian RJ, Brendel RW, Simon NM (October 2015). "A Systematic Review of Dropout From Psychotherapy for Posttraumatic Stress Disorder Among Iraq and Afghanistan Combat Veterans". Journal of Traumatic Stress. 28 (5): 401–9. doi:10.1002/jts.22038. PMID 26375387. S2CID 25316702.
- ^ Merz J, Schwarzer G, Gerger H (June 2019). "Comparative Efficacy and Acceptability of Pharmacological, Psychotherapeutic, and Combination Treatments in Adults With Posttraumatic Stress Disorder: A Network Meta-analysis". JAMA Psychiatry. 76 (9): 904–913. doi:10.1001/jamapsychiatry.2019.0951. PMC 6563588. PMID 31188399.
- ^ Forman-Hoffman V, Middleton JC, Feltner C, Gaynes BN, Weber RP, Bann C, et al. (2018). Psychological and Pharmacological Treatments for Adults With Posttraumatic Stress Disorder: A Systematic Review Update. AHRQ Comparative Effectiveness Reviews. Rockville, MD: Agency for Healthcare Research and Quality (US). PMID 30204376.
- ^ Althobaiti S, Kazantzis N, Ofori-Asenso R, Romero L, Fisher J, Mills KE, et al. (March 2020). "Efficacy of interpersonal psychotherapy for post-traumatic stress disorder: A systematic review and meta-analysis". Journal of Affective Disorders. 264: 286–294. doi:10.1016/j.jad.2019.12.021. PMID 32056763. S2CID 211111940.
![]() This article incorporates text from a free content work. Licensed under CC BY-SA 3.0 IGO. Text taken from A Lifeline to learning: leveraging mobile technology to support education for refugees, UNESCO, UNESCO. UNESCO.
This article incorporates text from a free content work. Licensed under CC BY-SA 3.0 IGO. Text taken from A Lifeline to learning: leveraging mobile technology to support education for refugees, UNESCO, UNESCO. UNESCO.
External links
- Post-traumatic stress disorder at Curlie
- Post traumatic stress disorder information from The National Child Traumatic Stress Network
- Information resources from The University of Queensland School of Medicine
- APA practice parameters for assessment and treatment for PTSD (Updated 2017)
- Resources for professionals from the VA National PTSD Center
 Psychiatry portal
Psychiatry portal