| |

| |
| Names | |
|---|---|
| IUPAC name
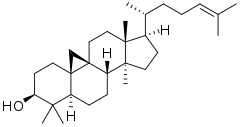
9,19-Cyclo-9β-lanost-24-en-3β-ol
| |
| Systematic IUPAC name
(1R,3aS,3bS,5aR,7S,9aR,10aS,12aR)-3a,6,6,12a-Tetramethyl-1-[(2R)-6-methylhept-5-en-2-yl]tetradecahydro-2H,10H-cyclopenta[a]cyclopropa[e]phenanthren-7-ol | |
| Other names
9β,19-cyclo-24-lanosten-3β-ol,
(3β)-9,19-Cyclolanost-24-en-3-ol | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C30H50O | |
| Molar mass | 426.72 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cycloartenol is an important triterpenoid often found in plants. It belongs to the sterol class of steroids. It is the starting point for the synthesis of almost all plant steroids,[1] making them chemically distinct from the steroids of fungi and animals, which are, instead, produced from lanosterol.
Synthesis[edit]
The biosynthesis of cycloartenol starts from the triterpenoid squalene. It is the first precursor in the biosynthesis of other stanols and sterols, referred to as phytostanols and phytosterols in photosynthetic organisms and plants. The identities and distribution of phytostanols and phytosterols is characteristic of a plant species.
References[edit]
- ^ Schaller, Hubert (May 2003). "The role of sterols in plant growth and development". Progress in Lipid Research. 42 (3): 163–175. doi:10.1016/S0163-7827(02)00047-4. PMID 12689617.