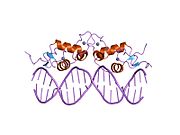
The glucocorticoid receptor (GR or GCR) also known as NR3C1 (nuclear receptor subfamily 3, group C, member 1) is the receptor to which cortisol and other glucocorticoids bind.
The GR is expressed in almost every cell in the body and regulates genes controlling the development, metabolism, and immune response. Because the receptor gene is expressed in several forms, it has many different (pleiotropic) effects in different parts of the body.
When glucocorticoids bind to GR, its primary mechanism of action is the regulation of gene transcription.[5][6] The unbound receptor resides in the cytosol of the cell. After the receptor is bound to glucocorticoid, the receptor-glucocorticoid complex can take either of two paths. The activated GR complex up-regulates the expression of anti-inflammatory proteins in the nucleus or represses the expression of pro-inflammatory proteins in the cytosol (by preventing the translocation of other transcription factors from the cytosol into the nucleus).
In humans, the GR protein is encoded by NR3C1 gene which is located on chromosome 5 (5q31).[7][8]
Structure[edit]
Like the other steroid receptors,[9] the glucocorticoid receptor is modular in structure[10] and contains the following domains (labeled A - F):
- A/B - N-terminal regulatory domain
- C - DNA-binding domain (DBD)
- D - hinge region
- E - ligand-binding domain (LBD)
- F - C-terminal domain
Ligand binding and response[edit]
In the absence of hormone, the glucocorticoid receptor (GR) resides in the cytosol complexed with a variety of proteins including heat shock protein 90 (hsp90), the heat shock protein 70 (hsp70) and the protein FKBP4 (FK506-binding protein 4).[11] The endogenous glucocorticoid hormone cortisol diffuses through the cell membrane into the cytoplasm and binds to the glucocorticoid receptor (GR) resulting in release of the heat shock proteins. The resulting activated form GR has two principal mechanisms of action, transactivation and transrepression,[12][13] described below.
Transactivation[edit]
A direct mechanism of action involves homodimerization of the receptor, translocation via active transport into the nucleus, and binding to specific DNA response elements activating gene transcription. This mechanism of action is referred to as transactivation. The biological response depends on the cell type.[citation needed]
Transrepression[edit]
In the absence of activated GR, other transcription factors such as NF-κB or AP-1 themselves are able to transactivate target genes.[14] However activated GR can complex with these other transcription factors and prevent them from binding their target genes and hence repress the expression of genes that are normally upregulated by NF-κB or AP-1. This indirect mechanism of action is referred to as transrepression.[citation needed] GR transrepression via NF-κB and AP-1 is restricted only to certain cell types, and is not considered the universal mechanism for IκBα repression. [15][16]
Clinical significance[edit]
The GR is abnormal in familial glucocorticoid resistance.[17]
In central nervous system structures, the glucocorticoid receptor is gaining interest as a novel representative of neuroendocrine integration, functioning as a major component of endocrine influence - specifically the stress response - upon the brain. The receptor is now implicated in both short and long-term adaptations seen in response to stressors and may be critical to the understanding of psychological disorders, including some or all subtypes of depression and post-traumatic stress disorder (PTSD).[18] Indeed, long-standing observations such as the mood dysregulations typical of Cushing's disease demonstrate the role of corticosteroids in regulating psychologic state; recent advances have demonstrated interactions with norepinephrine and serotonin at the neural level.[19][20]
In preeclampsia (a hypertensive disorder commonly occurring in pregnant women), the level of a miRNA sequence possibly targeting this protein is elevated in the blood of the mother. Rather, the placenta elevates the level of exosomes containing this miRNA, which can result in inhibition of translation of molecule. Clinical significance of this information is not yet clarified.[21]
Agonists and antagonists[edit]
Dexamethasone and other corticosteroids are agonists, while mifepristone and ketoconazole are antagonists of the GR. Anabolic steroids also prevent cortisol from binding to the glucocorticoid receptor.
Interactions[edit]
Glucocorticoid receptor has been shown to interact with:
- BAG1,[22][23]
- CEBPB,[24]
- CREBBP,[25]
- DAP3,[26]
- DAXX,[27]
- HSP90AA1,[26][28][29][30][31][32][33]
- HNRPU,[34]
- MED1,[35][36]
- MED14,[36]
- Mineralocorticoid receptor,[37]
- NRIP1,[35][38][39]
- NCOR1,[40][41]
- NCOA1,[35][42]
- NCOA2,[35][43]
- NCOA3,[35][44]
- POU2F1,[45][46]
- RANBP9,[47]
- RELA,[47][48][49]
- SMAD3,[50][51]
- SMARCD1,[44]
- SMARCA4[44][52]
- STAT3,[53][54]
- STAT5B,[55]
- Thioredoxin,[56]
- TRIM28,[57] and
- YWHAH.[58]
See also[edit]
References[edit]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000113580 - Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024431 - Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Lu NZ, Wardell SE, Burnstein KL, Defranco D, Fuller PJ, Giguere V, et al. (December 2006). "International Union of Pharmacology. LXV. The pharmacology and classification of the nuclear receptor superfamily: glucocorticoid, mineralocorticoid, progesterone, and androgen receptors". Pharmacological Reviews. 58 (4): 782–797. doi:10.1124/pr.58.4.9. PMID 17132855. S2CID 28626145. [Free full text]
- ^ Rhen T, Cidlowski JA (October 2005). "Antiinflammatory action of glucocorticoids--new mechanisms for old drugs". The New England Journal of Medicine. 353 (16): 1711–1723. doi:10.1056/NEJMra050541. PMID 16236742. S2CID 5744727.
- ^ Hollenberg SM, Weinberger C, Ong ES, Cerelli G, Oro A, Lebo R, et al. (December 1985). "Primary structure and expression of a functional human glucocorticoid receptor cDNA". Nature. 318 (6047): 635–641. Bibcode:1985Natur.318..635H. doi:10.1038/318635a0. PMC 6165583. PMID 2867473.
- ^ Francke U, Foellmer BE (May 1989). "The glucocorticoid receptor gene is in 5q31-q32 [corrected]". Genomics. 4 (4): 610–612. doi:10.1016/0888-7543(89)90287-5. PMID 2744768.
- ^ Kumar R, Thompson EB (May 1999). "The structure of the nuclear hormone receptors". Steroids. 64 (5): 310–319. doi:10.1016/S0039-128X(99)00014-8. PMID 10406480. S2CID 18333397.
- ^ Kumar R, Thompson EB (April 2005). "Gene regulation by the glucocorticoid receptor: structure:function relationship". The Journal of Steroid Biochemistry and Molecular Biology. 94 (5): 383–394. doi:10.1016/j.jsbmb.2004.12.046. PMID 15876404. S2CID 25315991.
- ^ Pratt WB, Morishima Y, Murphy M, Harrell M (2006). "Chaperoning of glucocorticoid receptors". Molecular Chaperones in Health and Disease. Handbook of Experimental Pharmacology. Vol. 172. pp. 111–138. doi:10.1007/3-540-29717-0_5. ISBN 978-3-540-25875-9. PMID 16610357.
- ^ Buckingham JC (January 2006). "Glucocorticoids: exemplars of multi-tasking". British Journal of Pharmacology. 147 (Supplement 1): S258–S268. doi:10.1038/sj.bjp.0706456. PMC 1760726. PMID 16402112.
- ^ Hayashi R, Wada H, Ito K, Adcock IM (October 2004). "Effects of glucocorticoids on gene transcription". European Journal of Pharmacology. 500 (1–3): 51–62. doi:10.1016/j.ejphar.2004.07.011. PMID 15464020.
- ^ Ray A, Prefontaine KE (January 1994). "Physical association and functional antagonism between the p65 subunit of transcription factor NF-kappa B and the glucocorticoid receptor". Proceedings of the National Academy of Sciences of the United States of America. 91 (2): 752–756. Bibcode:1994PNAS...91..752R. doi:10.1073/pnas.91.2.752. PMC 43027. PMID 8290595.
- ^ Coutinho AE, Chapman KE (March 2011). "The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights". Molecular and Cellular Endocrinology. 335 (1): 2–13. doi:10.1016/j.mce.2010.04.005. PMC 3047790. PMID 20398732.
- ^ Heck S, Bender K, Kullmann M, Göttlicher M, Herrlich P, Cato AC (August 1997). "I kappaB alpha-independent downregulation of NF-kappaB activity by glucocorticoid receptor". The EMBO Journal. 16 (15): 4698–4707. doi:10.1093/emboj/16.15.4698. PMC 1170096. PMID 9303314.
- ^ Mendonca BB, Leite MV, de Castro M, Kino T, Elias LL, Bachega TA, et al. (April 2002). "Female pseudohermaphroditism caused by a novel homozygous missense mutation of the GR gene". The Journal of Clinical Endocrinology and Metabolism. 87 (4): 1805–1809. doi:10.1210/jcem.87.4.8379. PMID 11932321.
- ^ Maletic V, Robinson M, Oakes T, Iyengar S, Ball SG, Russell J (December 2007). "Neurobiology of depression: an integrated view of key findings". International Journal of Clinical Practice. 61 (12): 2030–2040. doi:10.1111/j.1742-1241.2007.01602.x. PMC 2228409. PMID 17944926. [Free full text]
- ^ Savitz J, Lucki I, Drevets WC (May 2009). "5-HT(1A) receptor function in major depressive disorder". Progress in Neurobiology. 88 (1): 17–31. doi:10.1016/j.pneurobio.2009.01.009. PMC 2736801. PMID 19428959. [Free full text]
- ^ Schechter DS, Moser DA, Paoloni-Giacobino A, Stenz L, Gex-Fabry M, Aue T, et al. (2015). "Methylation of NR3C1 is related to maternal PTSD, parenting stress and maternal medial prefrontal cortical activity in response to child separation among mothers with histories of violence exposure". Front Psychol. 6: 690. doi:10.3389/fpsyg.2015.00690. PMC 4447998. PMID 26074844.
- ^ Salomon C, Guanzon D, Scholz-Romero K, Longo S, Correa P, Illanes SE, Rice GE (September 2017). "Placental Exosomes as Early Biomarker of Preeclampsia: Potential Role of Exosomal MicroRNAs Across Gestation". The Journal of Clinical Endocrinology and Metabolism. 102 (9): 3182–3194. doi:10.1210/jc.2017-00672. PMID 28531338.
- ^ Kullmann M, Schneikert J, Moll J, Heck S, Zeiner M, Gehring U, Cato AC (June 1998). "RAP46 is a negative regulator of glucocorticoid receptor action and hormone-induced apoptosis". The Journal of Biological Chemistry. 273 (23): 14620–14625. doi:10.1074/jbc.273.23.14620. PMID 9603979.
- ^ Schneikert J, Hübner S, Langer G, Petri T, Jäättelä M, Reed J, Cato AC (December 2000). "Hsp70-RAP46 interaction in downregulation of DNA binding by glucocorticoid receptor". The EMBO Journal. 19 (23): 6508–6516. doi:10.1093/emboj/19.23.6508. PMC 305849. PMID 11101523.
- ^ Boruk M, Savory JG, Haché RJ (November 1998). "AF-2-dependent potentiation of CCAAT enhancer binding protein beta-mediated transcriptional activation by glucocorticoid receptor". Molecular Endocrinology. 12 (11): 1749–1763. doi:10.1210/mend.12.11.0191. PMID 9817600.
- ^ Almlöf T, Wallberg AE, Gustafsson JA, Wright AP (June 1998). "Role of important hydrophobic amino acids in the interaction between the glucocorticoid receptor tau 1-core activation domain and target factors". Biochemistry. 37 (26): 9586–9594. doi:10.1021/bi973029x. PMID 9649342.
- ^ a b Hulkko SM, Wakui H, Zilliacus J (August 2000). "The pro-apoptotic protein death-associated protein 3 (DAP3) interacts with the glucocorticoid receptor and affects the receptor function". The Biochemical Journal. 349. 349 (3): 885–893. doi:10.1042/bj3490885. PMC 1221218. PMID 10903152.
- ^ Lin DY, Lai MZ, Ann DK, Shih HM (May 2003). "Promyelocytic leukemia protein (PML) functions as a glucocorticoid receptor co-activator by sequestering Daxx to the PML oncogenic domains (PODs) to enhance its transactivation potential". The Journal of Biological Chemistry. 278 (18): 15958–15965. doi:10.1074/jbc.M300387200. PMID 12595526.
- ^ Jibard N, Meng X, Leclerc P, Rajkowski K, Fortin D, Schweizer-Groyer G, et al. (March 1999). "Delimitation of two regions in the 90-kDa heat shock protein (Hsp90) able to interact with the glucocorticosteroid receptor (GR)". Experimental Cell Research. 247 (2): 461–474. doi:10.1006/excr.1998.4375. PMID 10066374.
- ^ Kanelakis KC, Shewach DS, Pratt WB (September 2002). "Nucleotide binding states of hsp70 and hsp90 during sequential steps in the process of glucocorticoid receptor.hsp90 heterocomplex assembly". The Journal of Biological Chemistry. 277 (37): 33698–33703. doi:10.1074/jbc.M204164200. PMID 12093808.
- ^ Hecht K, Carlstedt-Duke J, Stierna P, Gustafsson J, Brönnegârd M, Wikström AC (October 1997). "Evidence that the beta-isoform of the human glucocorticoid receptor does not act as a physiologically significant repressor". The Journal of Biological Chemistry. 272 (42): 26659–26664. doi:10.1074/jbc.272.42.26659. PMID 9334248.
- ^ de Castro M, Elliot S, Kino T, Bamberger C, Karl M, Webster E, Chrousos GP (September 1996). "The non-ligand binding beta-isoform of the human glucocorticoid receptor (hGR beta): tissue levels, mechanism of action, and potential physiologic role". Molecular Medicine. 2 (5): 597–607. doi:10.1007/BF03401643. PMC 2230188. PMID 8898375.
- ^ van den Berg JD, Smets LA, van Rooij H (February 1996). "Agonist-free transformation of the glucocorticoid receptor in human B-lymphoma cells". The Journal of Steroid Biochemistry and Molecular Biology. 57 (3–4): 239–249. doi:10.1016/0960-0760(95)00271-5. PMID 8645634. S2CID 20582144.
- ^ Stancato LF, Silverstein AM, Gitler C, Groner B, Pratt WB (April 1996). "Use of the thiol-specific derivatizing agent N-iodoacetyl-3-[125I]iodotyrosine to demonstrate conformational differences between the unbound and hsp90-bound glucocorticoid receptor hormone binding domain". The Journal of Biological Chemistry. 271 (15): 8831–8836. doi:10.1074/jbc.271.15.8831. PMID 8621522.
- ^ Eggert M, Michel J, Schneider S, Bornfleth H, Baniahmad A, Fackelmayer FO, et al. (November 1997). "The glucocorticoid receptor is associated with the RNA-binding nuclear matrix protein hnRNP U". The Journal of Biological Chemistry. 272 (45): 28471–28478. doi:10.1074/jbc.272.45.28471. PMID 9353307.
- ^ a b c d e Zilliacus J, Holter E, Wakui H, Tazawa H, Treuter E, Gustafsson JA (April 2001). "Regulation of glucocorticoid receptor activity by 14--3-3-dependent intracellular relocalization of the corepressor RIP140". Molecular Endocrinology. 15 (4): 501–511. doi:10.1210/mend.15.4.0624. PMID 11266503.
- ^ a b Hittelman AB, Burakov D, Iñiguez-Lluhí JA, Freedman LP, Garabedian MJ (October 1999). "Differential regulation of glucocorticoid receptor transcriptional activation via AF-1-associated proteins". The EMBO Journal. 18 (19): 5380–5388. doi:10.1093/emboj/18.19.5380. PMC 1171607. PMID 10508170.
- ^ Savory JG, Préfontaine GG, Lamprecht C, Liao M, Walther RF, Lefebvre YA, Haché RJ (February 2001). "Glucocorticoid receptor homodimers and glucocorticoid-mineralocorticoid receptor heterodimers form in the cytoplasm through alternative dimerization interfaces". Molecular and Cellular Biology. 21 (3): 781–793. doi:10.1128/MCB.21.3.781-793.2001. PMC 86670. PMID 11154266.
- ^ Tazawa H, Osman W, Shoji Y, Treuter E, Gustafsson JA, Zilliacus J (June 2003). "Regulation of subnuclear localization is associated with a mechanism for nuclear receptor corepression by RIP140". Molecular and Cellular Biology. 23 (12): 4187–4198. doi:10.1128/MCB.23.12.4187-4198.2003. PMC 156128. PMID 12773562.
- ^ Subramaniam N, Treuter E, Okret S (June 1999). "Receptor interacting protein RIP140 inhibits both positive and negative gene regulation by glucocorticoids". The Journal of Biological Chemistry. 274 (25): 18121–18127. doi:10.1074/jbc.274.25.18121. PMID 10364267.
- ^ Stevens A, Garside H, Berry A, Waters C, White A, Ray D (May 2003). "Dissociation of steroid receptor coactivator 1 and nuclear receptor corepressor recruitment to the human glucocorticoid receptor by modification of the ligand-receptor interface: the role of tyrosine 735". Molecular Endocrinology. 17 (5): 845–859. doi:10.1210/me.2002-0320. PMID 12569182.
- ^ Schulz M, Eggert M, Baniahmad A, Dostert A, Heinzel T, Renkawitz R (July 2002). "RU486-induced glucocorticoid receptor agonism is controlled by the receptor N terminus and by corepressor binding". The Journal of Biological Chemistry. 277 (29): 26238–26243. doi:10.1074/jbc.M203268200. PMID 12011091.
- ^ Kucera T, Waltner-Law M, Scott DK, Prasad R, Granner DK (July 2002). "A point mutation of the AF2 transactivation domain of the glucocorticoid receptor disrupts its interaction with steroid receptor coactivator 1". The Journal of Biological Chemistry. 277 (29): 26098–26102. doi:10.1074/jbc.M204013200. PMID 12118039.
- ^ Bledsoe RK, Montana VG, Stanley TB, Delves CJ, Apolito CJ, McKee DD, et al. (July 2002). "Crystal structure of the glucocorticoid receptor ligand binding domain reveals a novel mode of receptor dimerization and coactivator recognition". Cell. 110 (1): 93–105. doi:10.1016/S0092-8674(02)00817-6. PMID 12151000. S2CID 6955342.
- ^ a b c Hsiao PW, Fryer CJ, Trotter KW, Wang W, Archer TK (September 2003). "BAF60a mediates critical interactions between nuclear receptors and the BRG1 chromatin-remodeling complex for transactivation". Molecular and Cellular Biology. 23 (17): 6210–6220. doi:10.1128/MCB.23.17.6210-6220.2003. PMC 180928. PMID 12917342.
- ^ Préfontaine GG, Walther R, Giffin W, Lemieux ME, Pope L, Haché RJ (September 1999). "Selective binding of steroid hormone receptors to octamer transcription factors determines transcriptional synergism at the mouse mammary tumor virus promoter". The Journal of Biological Chemistry. 274 (38): 26713–26719. doi:10.1074/jbc.274.38.26713. PMID 10480874.
- ^ Préfontaine GG, Lemieux ME, Giffin W, Schild-Poulter C, Pope L, LaCasse E, et al. (June 1998). "Recruitment of octamer transcription factors to DNA by glucocorticoid receptor". Molecular and Cellular Biology. 18 (6): 3416–3430. doi:10.1128/MCB.18.6.3416. PMC 108923. PMID 9584182.
- ^ a b Rao MA, Cheng H, Quayle AN, Nishitani H, Nelson CC, Rennie PS (December 2002). "RanBPM, a nuclear protein that interacts with and regulates transcriptional activity of androgen receptor and glucocorticoid receptor". The Journal of Biological Chemistry. 277 (50): 48020–48027. doi:10.1074/jbc.M209741200. PMID 12361945.
- ^ Nissen RM, Yamamoto KR (September 2000). "The glucocorticoid receptor inhibits NFkappaB by interfering with serine-2 phosphorylation of the RNA polymerase II carboxy-terminal domain". Genes & Development. 14 (18): 2314–2329. doi:10.1101/gad.827900. PMC 316928. PMID 10995388.
- ^ Caldenhoven E, Liden J, Wissink S, Van de Stolpe A, Raaijmakers J, Koenderman L, et al. (April 1995). "Negative cross-talk between RelA and the glucocorticoid receptor: a possible mechanism for the antiinflammatory action of glucocorticoids". Molecular Endocrinology. 9 (4): 401–412. doi:10.1210/mend.9.4.7659084. PMID 7659084. S2CID 28680611.
- ^ Li G, Wang S, Gelehrter TD (October 2003). "Identification of glucocorticoid receptor domains involved in transrepression of transforming growth factor-beta action". The Journal of Biological Chemistry. 278 (43): 41779–41788. CiteSeerX 10.1.1.631.7318. doi:10.1074/jbc.M305350200. PMID 12902338. S2CID 950035.
- ^ Song CZ, Tian X, Gelehrter TD (October 1999). "Glucocorticoid receptor inhibits transforming growth factor-beta signaling by directly targeting the transcriptional activation function of Smad3". Proceedings of the National Academy of Sciences of the United States of America. 96 (21): 11776–11781. Bibcode:1999PNAS...9611776S. doi:10.1073/pnas.96.21.11776. PMC 18362. PMID 10518526.
- ^ Wallberg AE, Neely KE, Hassan AH, Gustafsson JA, Workman JL, Wright AP (March 2000). "Recruitment of the SWI-SNF chromatin remodeling complex as a mechanism of gene activation by the glucocorticoid receptor tau1 activation domain". Molecular and Cellular Biology. 20 (6): 2004–2013. doi:10.1128/MCB.20.6.2004-2013.2000. PMC 110817. PMID 10688647.
- ^ Lerner L, Henriksen MA, Zhang X, Darnell JE (October 2003). "STAT3-dependent enhanceosome assembly and disassembly: synergy with GR for full transcriptional increase of the alpha 2-macroglobulin gene". Genes & Development. 17 (20): 2564–2577. doi:10.1101/gad.1135003. PMC 218150. PMID 14522952.
- ^ Zhang Z, Jones S, Hagood JS, Fuentes NL, Fuller GM (December 1997). "STAT3 acts as a co-activator of glucocorticoid receptor signaling". The Journal of Biological Chemistry. 272 (49): 30607–30610. doi:10.1074/jbc.272.49.30607. PMID 9388192.
- ^ Stöcklin E, Wissler M, Gouilleux F, Groner B (October 1996). "Functional interactions between Stat5 and the glucocorticoid receptor" (PDF). Nature. 383 (6602): 726–728. Bibcode:1996Natur.383..726S. doi:10.1038/383726a0. PMID 8878484. S2CID 4356272.
- ^ Makino Y, Yoshikawa N, Okamoto K, Hirota K, Yodoi J, Makino I, Tanaka H (January 1999). "Direct association with thioredoxin allows redox regulation of glucocorticoid receptor function". The Journal of Biological Chemistry. 274 (5): 3182–3188. doi:10.1074/jbc.274.5.3182. PMID 9915858.
- ^ Chang CJ, Chen YL, Lee SC (October 1998). "Coactivator TIF1beta interacts with transcription factor C/EBPbeta and glucocorticoid receptor to induce alpha1-acid glycoprotein gene expression". Molecular and Cellular Biology. 18 (10): 5880–5887. doi:10.1128/mcb.18.10.5880. PMC 109174. PMID 9742105.
- ^ Wakui H, Wright AP, Gustafsson J, Zilliacus J (March 1997). "Interaction of the ligand-activated glucocorticoid receptor with the 14-3-3 eta protein". The Journal of Biological Chemistry. 272 (13): 8153–8156. doi:10.1074/jbc.272.13.8153. PMID 9079630.
Further reading[edit]
- Adcock IM, Ito K (June 2000). "Molecular mechanisms of corticosteroid actions". Monaldi Archives for Chest Disease = Archivio Monaldi per le Malattie del Torace. 55 (3): 256–266. PMID 10948677.
- Chikanza IC (June 2002). "Mechanisms of corticosteroid resistance in rheumatoid arthritis: a putative role for the corticosteroid receptor beta isoform". Annals of the New York Academy of Sciences. 966 (1): 39–48. Bibcode:2002NYASA.966...39C. doi:10.1111/j.1749-6632.2002.tb04200.x. PMID 12114257. S2CID 85100496.
- Neeck G, Kluter A, Dotzlaw H, Eggert M (June 2002). "Involvement of the glucocorticoid receptor in the pathogenesis of rheumatoid arthritis". Annals of the New York Academy of Sciences. 966 (1): 491–495. Bibcode:2002NYASA.966..491N. doi:10.1111/j.1749-6632.2002.tb04252.x. PMID 12114309. S2CID 5106644.
- Yudt MR, Cidlowski JA (August 2002). "The glucocorticoid receptor: coding a diversity of proteins and responses through a single gene". Molecular Endocrinology. 16 (8): 1719–1726. doi:10.1210/me.2002-0106. PMID 12145329.
- Torrego A, Pujols L, Picado C (September 2002). "[Response to glucocorticoid treatment in asthma. The role of alpha and beta isoforms of the glucocorticoid receptor]". Archivos de Bronconeumologia. 38 (9): 436–440. doi:10.1016/S0300-2896(02)75258-7. PMID 12237016.
- Bray PJ, Cotton RG (June 2003). "Variations of the human glucocorticoid receptor gene (NR3C1): pathological and in vitro mutations and polymorphisms". Human Mutation. 21 (6): 557–568. doi:10.1002/humu.10213. PMID 12754700. S2CID 26191891.
- Kino T, Pavlakis GN (April 2004). "Partner molecules of accessory protein Vpr of the human immunodeficiency virus type 1". DNA and Cell Biology. 23 (4): 193–205. doi:10.1089/104454904773819789. PMID 15142377.
- Lu NZ, Cidlowski JA (June 2004). "The origin and functions of multiple human glucocorticoid receptor isoforms". Annals of the New York Academy of Sciences. 1024 (1): 102–123. Bibcode:2004NYASA1024..102L. doi:10.1196/annals.1321.008. PMID 15265776. S2CID 36368837.
- Kino T, Chrousos GP (June 2004). "Human immunodeficiency virus type-1 accessory protein Vpr: a causative agent of the AIDS-related insulin resistance/lipodystrophy syndrome?". Annals of the New York Academy of Sciences. 1024 (1): 153–167. Bibcode:2004NYASA1024..153K. doi:10.1196/annals.1321.013. PMID 15265780. S2CID 23655886.
- Andersen JL, Planelles V (January 2005). "The role of Vpr in HIV-1 pathogenesis". Current HIV Research. 3 (1): 43–51. doi:10.2174/1570162052772988. PMID 15638722.
- Le Rouzic E, Benichou S (February 2005). "The Vpr protein from HIV-1: distinct roles along the viral life cycle". Retrovirology. 2 (1): 11. doi:10.1186/1742-4690-2-11. PMC 554975. PMID 15725353.
- Muthumani K, Choo AY, Premkumar A, Hwang DS, Thieu KP, Desai BM, Weiner DB (August 2005). "Human immunodeficiency virus type 1 (HIV-1) Vpr-regulated cell death: insights into mechanism". Cell Death and Differentiation. 12 (Suppl 1): 962–970. doi:10.1038/sj.cdd.4401583. PMID 15832179.
- Zhou J, Cidlowski JA (2005). "The human glucocorticoid receptor: one gene, multiple proteins and diverse responses". Steroids. 70 (5–7): 407–417. doi:10.1016/j.steroids.2005.02.006. PMID 15862824. S2CID 24272404.
- Chrousos GP, Kino T (October 2005). "Intracellular glucocorticoid signaling: a formerly simple system turns stochastic". Science's STKE. 2005 (304): pe48. doi:10.1126/stke.3042005pe48. PMID 16204701. S2CID 23148406.
- Plotkin LL, Labutin AL, Lebedev LV, Khanukov LA, Zelikson OS (1975). "[Balloon probe for the removal of emboli and thrombi]". Meditsinskaya Tekhnika (3): 42–43. PMID 1152650.
- Subramaniam M, Colvard D, Keeting PE, Rasmussen K, Riggs BL, Spelsberg TC (December 1992). "Glucocorticoid regulation of alkaline phosphatase, osteocalcin, and proto-oncogenes in normal human osteoblast-like cells". Journal of Cellular Biochemistry. 50 (4): 411–424. doi:10.1002/jcb.240500410. PMID 1469072. S2CID 21381419.
- Scherrer LC, Pratt WB (March 1992). "Association of the transformed glucocorticoid receptor with a cytoskeletal protein complex". The Journal of Steroid Biochemistry and Molecular Biology. 41 (3–8): 719–721. doi:10.1016/0960-0760(92)90411-B. hdl:2027.42/30199. PMID 1562545. S2CID 43672040.
- Cadepond F, Gasc JM, Delahaye F, Jibard N, Schweizer-Groyer G, Segard-Maurel I, et al. (July 1992). "Hormonal regulation of the nuclear localization signals of the human glucocorticosteroid receptor". Experimental Cell Research. 201 (1): 99–108. doi:10.1016/0014-4827(92)90352-9. PMID 1612132.
- Hurley DM, Accili D, Stratakis CA, Karl M, Vamvakopoulos N, Rorer E, et al. (February 1991). "Point mutation causing a single amino acid substitution in the hormone binding domain of the glucocorticoid receptor in familial glucocorticoid resistance". The Journal of Clinical Investigation. 87 (2): 680–686. doi:10.1172/JCI115046. PMC 296359. PMID 1704018.
- Encío IJ, Detera-Wadleigh SD (April 1991). "The genomic structure of the human glucocorticoid receptor". The Journal of Biological Chemistry. 266 (11): 7182–7188. doi:10.1016/S0021-9258(20)89627-6. PMID 1707881.
External links[edit]
- Human Protein Reference Database Archived 2006-03-01 at the Wayback Machine
- Glucocorticoid+receptors at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- FactorBook GR
- Overview of all the structural information available in the PDB for UniProt: P04150 (Glucocorticoid receptor) at the PDBe-KB.