| |
| Names | |
|---|---|
| Preferred IUPAC name
(9R)-1-[(2R,3S,4R,5R)-5-(6-Amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)oxolan-2-yl]-3,5,9-trihydroxy-3,5,10,14,19-pentaoxo-8,8-dimethyl-2,4,6-trioxa-18-thia-11,15-diaza-3λ5,5λ5-diphosphahenicosan-21-oic acid | |
| Identifiers | |
| |
| ChemSpider | |
| ECHA InfoCard | 100.007.596 |
| MeSH | Malonyl+CoA |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C24H38N7O19P3S | |
| Molar mass | 853.582 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
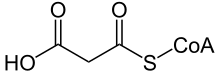
Malonyl-CoA is a coenzyme A derivative of malonic acid.
Functions[edit]
It plays a key role in chain elongation in fatty acid biosynthesis and polyketide biosynthesis.
Cytosolic fatty acid biosynthesis[edit]
Malonyl-CoA provides 2-carbon units to fatty acids and commits them to fatty acid chain synthesis.
Malonyl-CoA is formed by carboxylating acetyl-CoA using the enzyme acetyl-CoA carboxylase. One molecule of acetyl-CoA joins with a molecule of bicarbonate,[1] requiring energy rendered from ATP.
Malonyl-CoA is utilised in fatty acid biosynthesis by the enzyme malonyl coenzyme A:acyl carrier protein transacylase (MCAT). MCAT serves to transfer malonate from malonyl-CoA to the terminal thiol of holo-acyl carrier protein (ACP).
Mitochondrial fatty acid synthesis[edit]
Malonyl-CoA is formed in the first step of mitochondrial fatty acid synthesis (mtFASII) from malonic acid by malonyl-CoA synthetase (ACSF3).[2][3]
Polyketide biosynthesis[edit]
MCAT is also involved in bacterial polyketide biosynthesis. The enzyme MCAT together with an acyl carrier protein (ACP), and a polyketide synthase (PKS) and chain-length factor heterodimer, constitutes the minimal PKS of type II polyketides.
Regulation[edit]
Malonyl-CoA is a highly regulated molecule in fatty acid synthesis; as such, it inhibits the rate-limiting step in beta-oxidation of fatty acids. Malonyl-CoA inhibits fatty acids from associating with carnitine by regulating the enzyme carnitine acyltransferase, thereby preventing them from entering the mitochondria, where fatty acid oxidation and degradation occur.
Related diseases[edit]
Malonyl-CoA plays a special role in the mitochondrial clearance of toxic malonic acid in the metabolic disorder combined malonic and methylmalonic aciduria (CMAMMA).[4] In CMAMMA due to ACSF3, malonyl-CoA synthetase is decreased, which can generate malonyl-CoA from malonic acid, which can then be converted to acetyl-CoA by malonyl-CoA decarboxylase.[2][4] In contrast, in CMAMMA due to malonyl-CoA decarboxylase deficiency, malonyl-CoA decarboxylase is decreased, which converts malonyl-CoA to acetyl-CoA.[4]
See also[edit]
References[edit]
- ^ Nelson D, Cox M (2008). Lehninger principles of biochemistry (5th ed.). p. 806.
- ^ a b Witkowski, Andrzej; Thweatt, Jennifer; Smith, Stuart (September 2011). "Mammalian ACSF3 Protein Is a Malonyl-CoA Synthetase That Supplies the Chain Extender Units for Mitochondrial Fatty Acid Synthesis". Journal of Biological Chemistry. 286 (39): 33729–33736. doi:10.1074/jbc.M111.291591. ISSN 0021-9258. PMC 3190830. PMID 21846720.
- ^ Bowman, Caitlyn E.; Rodriguez, Susana; Selen Alpergin, Ebru S.; Acoba, Michelle G.; Zhao, Liang; Hartung, Thomas; Claypool, Steven M.; Watkins, Paul A.; Wolfgang, Michael J. (2017). "The Mammalian Malonyl-CoA Synthetase ACSF3 Is Required for Mitochondrial Protein Malonylation and Metabolic Efficiency". Cell Chemical Biology. 24 (6): 673–684.e4. doi:10.1016/j.chembiol.2017.04.009. PMC 5482780. PMID 28479296.
- ^ a b c Bowman, Caitlyn E.; Wolfgang, Michael J. (January 2019). "Role of the malonyl-CoA synthetase ACSF3 in mitochondrial metabolism". Advances in Biological Regulation. 71: 34–40. doi:10.1016/j.jbior.2018.09.002. PMC 6347522. PMID 30201289.
![{\displaystyle \mathrm {Malonic\ acid+CoA+ATP\ {\xrightarrow[{ACSF3}]{Malonyl{-}CoA\ Synthetase}}\ Malonyl{-}CoA\ {\xrightarrow[{MLYCD}]{Malonyl-CoA\ Decarboxylase}}\ Acetyl{-}CoA} }](https://wikimedia.org/api/rest_v1/media/math/render/svg/f3389fec9ec81ec307389ddc12b767a2fd82069f)