| Transient receptor potential (TRP) ion channel | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | TRP | ||||||||
| Pfam | PF06011 | ||||||||
| InterPro | IPR013555 | ||||||||
| OPM superfamily | 8 | ||||||||
| OPM protein | 3j5p | ||||||||
| Membranome | 605 | ||||||||
| |||||||||
Transient receptor potential channels (TRP channels) are a group of ion channels located mostly on the plasma membrane of numerous animal cell types. Most of these are grouped into two broad groups: Group 1 includes TRPC ( "C" for canonical), TRPV ("V" for vanilloid), TRPVL ("VL" for vanilloid-like), TRPM ("M" for melastatin), TRPS ("S" for soromelastatin), TRPN ("N" for mechanoreceptor potential C), and TRPA ("A" for ankyrin). Group 2 consists of TRPP ("P" for polycystic) and TRPML ("ML" for mucolipin).[1][2] Other less-well categorized TRP channels exist, including yeast channels and a number of Group 1 and Group 2 channels present in non-animals.[2][3][4] Many of these channels mediate a variety of sensations such as pain, temperature, different kinds of taste, pressure, and vision. In the body, some TRP channels are thought to behave like microscopic thermometers and used in animals to sense hot or cold.[5] Some TRP channels are activated by molecules found in spices like garlic (allicin), chili pepper (capsaicin), wasabi (allyl isothiocyanate); others are activated by menthol, camphor, peppermint, and cooling agents; yet others are activated by molecules found in cannabis (i.e., THC, CBD and CBN) or stevia. Some act as sensors of osmotic pressure, volume, stretch, and vibration. Most of the channels are activated or inhibited by signaling lipids and contribute to a family of lipid-gated ion channels.[6][7]
These ion channels have a relatively non-selective permeability to cations, including sodium, calcium and magnesium.
TRP channels were initially discovered in the so-called "transient receptor potential" mutant (trp-mutant) strain of the fruit fly Drosophila, hence their name (see History of Drosophila TRP channels below). Later, TRP channels were found in vertebrates where they are ubiquitously expressed in many cell types and tissues. Most TRP channels are composed of 6 membrane-spanning helices with intracellular N- and C-termini. Mammalian TRP channels are activated and regulated by a wide variety of stimuli and are expressed throughout the body.
Families[edit]
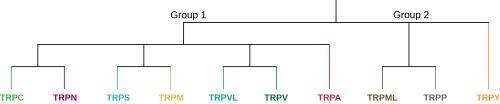
In the animal TRP superfamily there are currently 9 proposed families split into two groups, each family containing a number of subfamilies.[2] Group one consists of TRPC, TRPV, TRPVL, TRPA, TRPM, TRPS, and TRPN, while group two contains TRPP and TRPML. There is an additional family labeled TRPY that is not always included in either of these groups. All of these sub-families are similar in that they are molecular sensing, non-selective cation channels that have six transmembrane segments, however, each sub-family is very unique and shares little structural homology with one another. This uniqueness gives rise to the various sensory perception and regulation functions that TRP channels have throughout the body. Group one and group two vary in that both TRPP and TRPML of group two have a much longer extracellular loop between the S1 and S2 transmembrane segments. Another differentiating characteristic is that all the group one sub-families either contain an N-terminal intracellular ankyrin repeat sequence, a C-terminal TRP domain sequence, or both—whereas both group two sub-families have neither.[8] Below are members of the sub-families and a brief description of each:
TRPA[edit]
| Family | Sub-Family | Known Taxa[9][10][11] |
|---|---|---|
| TRPA | TRPA1 | Vertebrates, arthropods, and molluscs |
| TRPA-like | Choanoflagellates, cnidarians, nematodes, arthropods (only crustaceans and myriapods), molluscs, and echinoderms | |
| TRPA5 | Arthropods (only crustaceans and insects) | |
| painless | ||
| pyrexia | ||
| waterwitch | ||
| HsTRPA | Specific to hymenopteran insects |
TRPA, A for "ankyrin", is named for the large amount of ankyrin repeats found near the N-terminus.[12] TRPA is primarily found in afferent nociceptive nerve fibers and is associated with the amplification of pain signaling as well as cold pain hypersensitivity. These channels have been shown to be both mechanical receptors for pain and chemosensors activated by various chemical species, including isothiocyanates (pungent chemicals in substances such as mustard oil and wasabi), cannabinoids, general and local analgesics, and cinnamaldehyde.[13]
While TRPA1 is expressed in a wide variety of animals, a variety of other TRPA channels exist outside of vertebrates. TRPA5, painless, pyrexia, and waterwitch are distinct phylogenetic branches within the TRPA clade, and are only evidenced to be expressed in crustaceans and insects,[8] while HsTRPA arose as a Hymenoptera-specific duplication of waterwitch.[14] Like TRPA1 and other TRP channels, these function as ion channels in a number of sensory systems. TRPA- or TRPA1-like channels also exists in a variety of species as a phylogenetically distinct clade, but these are less well understood.[10]
TRPC[edit]
| Family | Sub-Family | Known Taxa[11][15][16] |
|---|---|---|
| TRPC | TRPC1 | Vertebrates |
| TRPC2 | ||
| TRPC3 | ||
| TRPC4 | ||
| TRPC5 | ||
| TRPC6 | ||
| TRPC7 | ||
| TRP | Arthropods | |
| TRPgamma | ||
| TRPL | ||
| Unknown | Choanoflagellates, cnidarians, xenacoelomorphs, lophotrochozoans, and nematodes |
TRPC, C for "canonical", is named for being the most closely related to Drosophila TRP, the namesake of TRP channels. The phylogeny of TRPC channels has not been resolved in detail, but they are present across animal taxa. There are actually only six TRPC channels expressed in humans because TRPC2 is found to be expressed solely in mice and is considered a pseudo-gene in humans; this is partly due to the role of TRPC2 in detecting pheromones, which mice have an increased ability compared to humans. Mutations in TRPC channels have been associated with respiratory diseases along with focal segmental glomerulosclerosis in the kidneys.[13] All TRPC channels are activated either by phospholipase C (PLC) or diacyglycerol (DAG).
TRPML[edit]
| Family | Sub-Family | Known Taxa[11][17] |
|---|---|---|
| TRPML | Unknown | Cnidarians, basal vertebrates, tunicates, cephalochordates, hemichordates, echinoderms, arthropods, and nematodes |
| TRPML1 | Specific to jawed vertebrates | |
| TRPML2 | ||
| TRPML3 |
TRPML, ML for "mucolipin", gets its name from the neurodevelopmental disorder mucolipidosis IV. Mucolipidosis IV was first discovered in 1974 by E.R. Berman who noticed abnormalities in the eyes of an infant.[18] These abnormalities soon became associated with mutations to the MCOLN1 gene which encodes for the TRPML1 ion channel. TRPML is still not highly characterized. The three known vertebrate copies are restricted to jawed vertebrates, with some exceptions (e.g. Xenopus tropicalis).[17]
TRPM[edit]
| Family | Sub-Family | Known Taxa |
|---|---|---|
| TRPM | Alpha/α (inc. TRPM1, 3, 6, and 7) | Choanoflagellates and animals (except tardigrades) |
| Beta/β (inc. TRPM2, 4, 5, and 8) |
TRPM, M for "melastatin", was found during a comparative genetic analysis between benign nevi and malignant nevi (melanoma).[12] Mutations within TRPM channels have been associated with hypomagnesemia with secondary hypocalcemia. TRPM channels have also become known for their cold-sensing mechanisms, such is the case with TRPM8.[13] Comparative studies have shown that the functional domains and critical amino acids of TRPM channels are highly conserved across species.[19][10][20]
Phylogenetics has shown that TRPM channels are split into two major clades, αTRPM and βTRPM.[10][16] αTRPMs include vertebrate TRPM1, TRPM3, and the "chanzymes" TRPM6 and TRPM7, as well as the only insect TRPM channel, among others. βTRPMs include, but are not limited to, vertebrate TRPM2, TRPM4, TRPM5, and TRPM8 (the cold and menthol sensor). Two additional major clades have been described: TRPMc, which is present only in a variety of arthropods,[16] and a basal clade,[11][10] which has since been proposed to be a distinct and separate TRP channel family (TRPS).[16]
TRPN[edit]
| Family | Sub-Family | Known Taxa[21][11] |
|---|---|---|
| TRPN | TRPN/nompC | Placozoans, cnidarians, nematodes, arthropods, molluscs, annelids, and vertebrates (excluding amniotes) |
TRPN was originally described in Drosophila melanogaster and Caenorhabditis elegans as nompC, a mechanically gated ion channel.[22][21] Only a single TRPN, N for "no mechanoreceptor potential C," or "nompC", is known to be broadly expressed in animals (although some Cnidarians have more), and is notably only a pseudogene in amniote vertebrates.[21][11] Despite TRPA being named for ankyrin repeats, TRPN channels are thought to have the most of any TRP channel, typically around 28, which are highly conserved across taxa [21] Since its discovery, Drosophila nompC has been implicated in mechanosensation (including mechanical stimulation of the cuticle and sound detection) and cold nociception.[23]
TRPP[edit]
| Family | Sub-Family[11][24][25][2] | Known Taxa[26][25] |
|---|---|---|
| TRPP | PKD1-like | Animals (excluding arthropods) |
| PKD2-like | Animals | |
| Brividos | Insects |
TRPP, P for "polycistin", is named for polycystic kidney disease, which is associated with these channels.[12] These channels are also referred to as PKD (polycistic kidney disease) ion channels.
PKD2-like genes (examples include TRPP2, TRPP3, and TRPP5) encode canonical TRP channels. PKD1-like genes encode much larger proteins with 11 transmembrane segments, which do not have all the features of other TRP channels. However, 6 of the transmebrane segments of PKD1-like proteins have substantial sequence homology with TRP channels, indicating they may simply have diversified greatly from other closely related proteins.[26]
Insects have a third sub-family of TRPP, called brividos, which participate in cold sensing.[25][2]
TRPS[edit]
TRPS, S for Soromelastatin, was named as it forms a sister group to TRPM. TRPS is broadly present in animals, but notably absent in vertebrates and insects (among others).[16] TRPS has not yet been well described functionally, though it is known that the C. elegans TRPS, known as CED-11, is a calcium channel which participates in apoptosis.[27]
TRPV[edit]
| Family | Sub-Family | Known Taxa [11][28] |
|---|---|---|
| TRPV | Nanchung | Placozoans, cnidarians, nematodes, annelids, molluscs, and arthropods (possibly excluding arachnids) |
| Inactive | ||
| TRPV1 | Specific to vertebrates | |
| TRPV2 | ||
| TRPV3 | ||
| TRPV4 | ||
| TRPV5 | ||
| TRPV6 |
TRPV, V for "vanilloid", was originally discovered in Caenorhabditis elegans, and is named for the vanilloid chemicals that activate some of these channels.[24][29] These channels have been made famous for their association with molecules such as capsaicin (a TRPV1 agonist).[13] In addition to the 6 known vertebrate paralogues, 2 major clades are known outside of the deterostomes: nanchung and Iav. Mechanistic studies of these latter clades have been largely restricted to Drosophila, but phylogenetic analyses has placed a number of other genes from Placozoa, Annelida, Cnidaria, Mollusca, and other arthropods within them.[11][30][31] TRPV channels have also been described in protists.[11]
TRPVL[edit]
TRPVL has been proposed to be a sister clade to TRPV, and is limited to the cnidarians Nematostella vectensis and Hydra magnipapillata, and the annelid Capitella teleta.[11] Little is known concerning these channels.
TRPY[edit]
TRPY, Y for "yeast", is highly localized to the yeast vacuole, which is the functional equivalent of a lysosome in a mammalian cell, and acts as a mechanosensor for vacuolar osmotic pressure. Patch clamp techniques and hyperosmotic stimulation have illustrated that TRPY plays a role in intracellular calcium release.[32] Phylogenetic analysis has shown that TRPY1 does not form a part with the other metazoan TRP groups one and two, and is suggested to have evolved after the divergence of metazoans and fungi.[8] Others have indicated that TRPY are more closely related to TRPP.[33]
Structure[edit]
TRP channels are composed of 6 membrane-spanning helices (S1-S6) with intracellular N- and C-termini. Mammalian TRP channels are activated and regulated by a wide variety of stimuli including many post-transcriptional mechanisms like phosphorylation, G-protein receptor coupling, ligand-gating, and ubiquitination. The receptors are found in almost all cell types and are largely localized in cell and organelle membranes, modulating ion entry.
Most TRP channels form homo- or heterotetramers when completely functional. The ion selectivity filter, pore, is formed by the complex combination of p-loops in the tetrameric protein, which are situated in the extracellular domain between the S5 and S6 transmembrane segments. As with most cation channels, TRP channels have negatively charged residues within the pore to attract the positively charged ions.[34]
Group 1 Characteristics[edit]
Each channel in this group is structurally unique, which adds to the diversity of functions that TRP channels possess, however, there are some commonalities that distinguish this group from others. Starting from the intracellular N-terminus there are varying lengths of ankryin repeats (except in TRPM) that aid with membrane anchoring and other protein interactions. Shortly following S6 on the C-terminal end, there is a highly conserved TRP domain (except in TRPA) which is involved with gating modulation and channel multimerization. Other C-terminal modifications such as alpha-kinase domains in TRPM7 and M8 have been seen as well in this group.[8][13][12]
Group 2 Characteristics[edit]
Group two most distinguishable trait is the long extracellular span between the S1 and S2 transmembrane segments. Members of group two are also lacking in ankryin repeats and a TRP domain. They have been shown, however, to have endoplasmic reticulum (ER) retention sequences towards on the C-terminal end illustrating possible interactions with the ER.[8][13][12]
Function[edit]
TRP channels modulate ion entry driving forces and Ca2+ and Mg2+ transport machinery in the plasma membrane, where most of them are located. TRPs have important interactions with other proteins and often form signaling complexes, the exact pathways of which are unknown.[35] TRP channels were initially discovered in the trp mutant strain of the fruit fly Drosophila[36] which displayed transient elevation of potential in response to light stimuli and were so named transient receptor potential channels.[37] TRPML channels function as intracellular calcium release channels and thus serve an important role in organelle regulation.[35] Importantly, many of these channels mediate a variety of sensations like the sensations of pain, temperature, different kinds of taste, pressure, and vision. In the body, some TRP channels are thought to behave like microscopic thermometers and are used in animals to sense hot or cold. TRPs act as sensors of osmotic pressure, volume, stretch, and vibration. TRPs have been seen to have complex multidimensional roles in sensory signaling. Many TRPs function as intracellular calcium release channels.
Pain and temperature sensation[edit]
TRP ion channels convert energy into action potentials in somatosensory nociceptors.[38] Thermo-TRP channels have a C-terminal domain that is responsible for thermosensation and have a specific interchangeable region that allows them to sense temperature stimuli that is tied to ligand regulatory processes.[39] Although most TRP channels are modulated by changes in temperature, some have a crucial role in temperature sensation. There are at least 6 different Thermo-TRP channels and each plays a different role. For instance, TRPM8 relates to mechanisms of sensing cold, TRPV1 and TRPM3 contribute to heat and inflammation sensations, and TRPA1 facilitates many signaling pathways like sensory transduction, nociception, inflammation and oxidative stress.[38]
Taste[edit]
TRPM5 is involved in taste signaling of sweet, bitter and umami tastes by modulating the signal pathway in type II taste receptor cells.[40] TRPM5 is activated by the sweet glycosides found in the stevia plant.
Several other TRP channels play a significant role in chemosensation through sensory nerve endings in the mouth that are independent from taste buds. TRPA1 responds to mustard oil (allyl isothiocyanate), wasabi, and cinnamon, TRPA1 and TRPV1 responds to garlic (allicin), TRPV1 responds to chilli pepper (capsaicin), TRPM8 is activated by menthol, camphor, peppermint, and cooling agents; TRPV2 is activated by molecules (THC, CBD and CBN) found in marijuana.
TRP-like channels in insect vision[edit]

The trp-mutant fruit flies, which lack a functional copy of trp gene, are characterized by a transient response to light, unlike wild-type flies that demonstrate a sustained photoreceptor cell activity in response to light.[36] A distantly related isoform of TRP channel, TRP-like channel (TRPL), was later identified in Drosophila photoreceptors, where it is expressed at approximately 10- to 20-fold lower levels than TRP protein. A mutant fly, trpl, was subsequently isolated. Apart from structural differences, the TRP and TRPL channels differ in cation permeability and pharmacological properties.
TRP/TRPL channels are solely responsible for depolarization of insect photoreceptor plasma membrane in response to light. When these channels open, they allow sodium and calcium to enter the cell down the concentration gradient, which depolarizes the membrane. Variations in light intensity affect the total number of open TRP/TRPL channels, and, therefore, the degree of membrane depolarization. These graded voltage responses propagate to photoreceptor synapses with second-order retinal neurons and further to the brain.
It is important to note that the mechanism of insect photoreception is dramatically different from that in mammals. Excitation of rhodopsin in mammalian photoreceptors leads to the hyperpolarization of the receptor membrane but not to depolarization as in the insect eye. In Drosophila and, it is presumed, other insects, a phospholipase C (PLC)-mediated signaling cascade links photoexcitation of rhodopsin to the opening of the TRP/TRPL channels. Although numerous activators of these channels such as phosphatidylinositol-4,5-bisphosphate (PIP2) and polyunsaturated fatty acids (PUFAs) were known for years, a key factor mediating chemical coupling between PLC and TRP/TRPL channels remained a mystery until recently. It was found that breakdown of a lipid product of PLC cascade, diacylglycerol (DAG), by the enzyme diacylglycerol lipase, generates PUFAs that can activate TRP channels, thus initiating membrane depolarization in response to light.[41] This mechanism of TRP channel activation may be well-preserved among other cell types where these channels perform various functions.
Clinical significance[edit]
Mutations in TRPs have been linked to neurodegenerative disorders, skeletal dysplasia, kidney disorders,[35] and may play an important role in cancer. TRPs may make important therapeutic targets. There is significant clinical significance to TRPV1, TRPV2, TRPV3 and TRPM8’s role as thermoreceptors, and TRPV4 and TRPA1’s role as mechanoreceptors; reduction of chronic pain may be possible by targeting ion channels involved in thermal, chemical, and mechanical sensation to reduce their sensitivity to stimuli.[42] For instance the use of TRPV1 agonists would potentially inhibit nociception at TRPV1, particularly in pancreatic tissue where TRPV1 is highly expressed.[43] The TRPV1 agonist capsaicin, found in chili peppers, has been indicated to relieve neuropathic pain.[35] TRPV1 agonists inhibit nociception at TRPV1
Role in cancer[edit]
Altered expression of TRP proteins often leads to tumorigenesis, as reported for TRPV1, TRPV6, TRPC1, TRPC6, TRPM4, TRPM5, and TRPM8.[43] TRPV1 and TRPV2 have been implicated in breast cancer. TRPV1 expression in aggregates found at endoplasmic reticulum or Golgi apparatus and/or surrounding these structures in breast cancer patients confer worse survival.[44]
TRPM family of ion channels are particularly associated with prostate cancer where TRPM2 (and its long noncoding RNA TRPM2-AS), TRPM4, and TRPM8 are overexpressed in prostate cancer associated with more aggressive outcomes.[45] TRPM3 has been shown to promote growth and autophagy in clear cell renal cell carcinoma,[46] TRPM4 is overexpressed in diffuse large B-cell lymphoma associated with poorer survival,[47] while TRPM5 has oncogenic properties in melanoma.[48]
TRP channels take center stage in modulating chemotherapy resistance in breast cancer.[49] Some TRP channels such as TRPA1 and TRPC5 are tightly associated with drug resistance during cancer treatment; TRPC5-mediated high Ca2+ influx activates the transcription factor NFATC3 (Nuclear Factor of Activated T Cells, Cytoplasmic 3), which triggers p-glycoprotein (p-gp) transcription. The overexpression of p-gp is widely recognized as a major factor in chemoresistance in cancer cells, as it functions as an active efflux pump that can remove various foreign substances, including chemotherapeutic agents, from within the cell.[49]

- TRPC5 overexpression activates the transcription factor NFATC3 Ca2+ signaling pathway, leading to p-gp overexpression. Moreover, the overexpressed p-gp expels chemotherapeutic drugs such as doxorubicin triggering chemoresistance.
- Chemoresistant breast cancer cells overexpressing TRPC5 transfer channel units to chemo sensitive recipient cells via extracellular vesicles (EV), leading to the development of TRPC5-mediated chemoresistance in these cells.[49]
Contrarily, other TRP channels, such as TRPV1 and TRPV2, have been demonstrated to potentiate the anti-tumorigenic effects of certain chemotherapeutic agents and TRPV2 is a potential biomarker and therapeutic target in triple negative breast cancer.[49]
Role in inflammatory responses[edit]
In addition to TLR4 mediated pathways, certain members of the family of the transient receptor potential ion channels recognize LPS. LPS-mediated activation of TRPA1 was shown in mice[50] and Drosophila melanogaster flies.[51] At higher concentrations, LPS activates other members of the sensory TRP channel family as well, such as TRPV1, TRPM3 and to some extent TRPM8.[52] LPS is recognized by TRPV4 on epithelial cells. TRPV4 activation by LPS was necessary and sufficient to induce nitric oxide production with a bactericidal effect.[53]
History of Drosophila TRP channels[edit]
The original TRP-mutant in Drosophila was first described by Cosens and Manning in 1969 as "a mutant strain of D. melanogaster which, though behaving phototactically positive in a T-maze under low ambient light, is visually impaired and behaves as though blind". It also showed an abnormal electroretinogram response of photoreceptors to light which was transient rather than sustained as in the "wild type".[36] It was investigated subsequently by Baruch Minke, a post-doc in the group of William Pak, and named TRP according to its behavior in the ERG.[54] The identity of the mutated protein was unknown until it was cloned by Craig Montell, a post-doctoral researcher in Gerald Rubin's research group, in 1989, who noted its predicted structural relationship to channels known at the time[37] and Roger Hardie and Baruch Minke who provided evidence in 1992 that it is an ion channel that opens in response to light stimulation.[55] The TRPL channel was cloned and characterized in 1992 by the research group of Leonard Kelly.[56] In 2013, Montell and his research group found that the TRPL (TRP-like) cation channel was a direct target for tastants in gustatory receptor neurons and could be reversibly down-regulated.[57]
See also[edit]
References[edit]
- ^ Islam MS, ed. (January 2011). Transient Receptor Potential Channels. Advances in Experimental Medicine and Biology. Vol. 704. Berlin: Springer. p. 700. ISBN 978-94-007-0264-6.
- ^ a b c d e Himmel NJ, Cox DN (August 2020). "Transient receptor potential channels: current perspectives on evolution, structure, function and nomenclature". Proceedings. Biological Sciences. 287 (1933): 20201309. doi:10.1098/rspb.2020.1309. PMC 7482286. PMID 32842926.
- ^ Arias-Darraz L, Cabezas D, Colenso CK, Alegría-Arcos M, Bravo-Moraga F, Varas-Concha I, et al. (January 2015). "A transient receptor potential ion channel in Chlamydomonas shares key features with sensory transduction-associated TRP channels in mammals". The Plant Cell. 27 (1): 177–88. doi:10.1105/tpc.114.131862. PMC 4330573. PMID 25595824.
- ^ Lindström JB, Pierce NT, Latz MI (October 2017). "Role of TRP Channels in Dinoflagellate Mechanotransduction". The Biological Bulletin. 233 (2): 151–167. doi:10.1086/695421. PMID 29373067. S2CID 3388001.
- ^ Vriens J, Nilius B, Voets T (September 2014). "Peripheral thermosensation in mammals". Nature Reviews. Neuroscience. 15 (9): 573–89. doi:10.1038/nrn3784. PMID 25053448. S2CID 27149948.
- ^ Robinson CV, Rohacs T, Hansen SB (September 2019). "Tools for Understanding Nanoscale Lipid Regulation of Ion Channels". Trends in Biochemical Sciences. 44 (9): 795–806. doi:10.1016/j.tibs.2019.04.001. PMC 6729126. PMID 31060927.
- ^ Hansen SB (May 2015). "Lipid agonism: The PIP2 paradigm of ligand-gated ion channels". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1851 (5): 620–8. doi:10.1016/j.bbalip.2015.01.011. PMC 4540326. PMID 25633344.
- ^ a b c d e Kadowaki T (October 2015). "Evolutionary dynamics of metazoan TRP channels". Pflügers Archiv. 467 (10): 2043–53. doi:10.1007/s00424-015-1705-5. PMID 25823501. S2CID 9190224.
- ^ Kang K, Pulver SR, Panzano VC, Chang EC, Griffith LC, Theobald DL, Garrity PA (March 2010). "Analysis of Drosophila TRPA1 reveals an ancient origin for human chemical nociception". Nature. 464 (7288): 597–600. Bibcode:2010Natur.464..597K. doi:10.1038/nature08848. PMC 2845738. PMID 20237474.
- ^ a b c d e Himmel NJ, Letcher JM, Sakurai A, Gray TR, Benson MN, Cox DN (November 2019). "Drosophila menthol sensitivity and the Precambrian origins of transient receptor potential-dependent chemosensation". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 374 (1785): 20190369. doi:10.1098/rstb.2019.0369. PMC 6790378. PMID 31544603.
- ^ a b c d e f g h i j k Peng G, Shi X, Kadowaki T (March 2015). "Evolution of TRP channels inferred by their classification in diverse animal species". Molecular Phylogenetics and Evolution. 84: 145–57. doi:10.1016/j.ympev.2014.06.016. PMID 24981559.
- ^ a b c d e Moran MM, McAlexander MA, Bíró T, Szallasi A (August 2011). "Transient receptor potential channels as therapeutic targets". Nature Reviews. Drug Discovery. 10 (8): 601–20. doi:10.1038/nrd3456. PMID 21804597. S2CID 8809131.
- ^ a b c d e f Szallasi A (2015-04-09). TRP channels as therapeutic targets : from basic science to clinical use. Szallasi, Arpad, 1958-, McAlexander, M. Allen. Amsterdam [Netherlands]. ISBN 9780124200791. OCLC 912315205.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Kohno K, Sokabe T, Tominaga M, Kadowaki T (September 2010). "Honey bee thermal/chemical sensor, AmHsTRPA, reveals neofunctionalization and loss of transient receptor potential channel genes". The Journal of Neuroscience. 30 (37): 12219–29. doi:10.1523/JNEUROSCI.2001-10.2010. PMC 6633439. PMID 20844118.
- ^ French AS, Meisner S, Liu H, Weckström M, Torkkeli PH (2015). "Transcriptome analysis and RNA interference of cockroach phototransduction indicate three opsins and suggest a major role for TRPL channels". Frontiers in Physiology. 6: 207. doi:10.3389/fphys.2015.00207. PMC 4513288. PMID 26257659.
- ^ a b c d e Himmel NJ, Gray TR, Cox DN (July 2020). "Phylogenetics Identifies Two Eumetazoan TRPM Clades and an Eighth TRP Family, TRP Soromelastatin (TRPS)". Molecular Biology and Evolution. 37 (7): 2034–2044. doi:10.1093/molbev/msaa065. PMC 7306681. PMID 32159767.
- ^ a b García-Añoveros J, Wiwatpanit T (2014). "TRPML2 and Mucolipin Evolution". Mammalian Transient Receptor Potential (TRP) Cation Channels. Handbook of Experimental Pharmacology. Vol. 222. pp. 647–58. doi:10.1007/978-3-642-54215-2_25. ISBN 978-3-642-54214-5. PMID 24756724.
- ^ Berman ER, Livni N, Shapira E, Merin S, Levij IS (April 1974). "Congenital corneal clouding with abnormal systemic storage bodies: a new variant of mucolipidosis". The Journal of Pediatrics. 84 (4): 519–26. doi:10.1016/s0022-3476(74)80671-2. PMID 4365943.
- ^ Mederos y Schnitzler M, Wäring J, Gudermann T, Chubanov V (May 2008). "Evolutionary determinants of divergent calcium selectivity of TRPM channels". FASEB Journal. 22 (5): 1540–51. doi:10.1096/fj.07-9694com. PMID 18073331. S2CID 25474094.
- ^ Iordanov I, Tóth B, Szollosi A, Csanády L (April 2019). "Enzyme activity and selectivity filter stability of ancient TRPM2 channels were simultaneously lost in early vertebrates". eLife. 8. doi:10.7554/eLife.44556. PMC 6461439. PMID 30938679.
- ^ a b c d Schüler A, Schmitz G, Reft A, Özbek S, Thurm U, Bornberg-Bauer E (June 2015). "The Rise and Fall of TRP-N, an Ancient Family of Mechanogated Ion Channels, in Metazoa". Genome Biology and Evolution. 7 (6): 1713–27. doi:10.1093/gbe/evv091. PMC 4494053. PMID 26100409.
- ^ Walker RG, Willingham AT, Zuker CS (March 2000). "A Drosophila mechanosensory transduction channel". Science. 287 (5461): 2229–34. Bibcode:2000Sci...287.2229W. doi:10.1126/science.287.5461.2229. PMID 10744543.
- ^ Himmel N, Patel A, Cox D (March 2017). "Invertebrate Nociception". The Oxford Research Encyclopedia of Neuroscience. doi:10.1093/acrefore/9780190264086.013.166. ISBN 9780190264086.
- ^ a b Montell C (July 2001). "Physiology, phylogeny, and functions of the TRP superfamily of cation channels". Science's STKE. 2001 (90): re1. doi:10.1126/stke.2001.90.re1. PMID 11752662. S2CID 37074808.
- ^ a b c Gallio M, Ofstad TA, Macpherson LJ, Wang JW, Zuker CS (February 2011). "The coding of temperature in the Drosophila brain". Cell. 144 (4): 614–24. doi:10.1016/j.cell.2011.01.028. PMC 3336488. PMID 21335241.
- ^ a b Bezares-Calderón LA, Berger J, Jasek S, Verasztó C, Mendes S, Gühmann M, et al. (December 2018). "Neural circuitry of a polycystin-mediated hydrodynamic startle response for predator avoidance". eLife. 7. doi:10.7554/eLife.36262. PMC 6294549. PMID 30547885.
- ^ Driscoll K, Stanfield GM, Droste R, Horvitz HR (August 2017). "Presumptive TRP channel CED-11 promotes cell volume decrease and facilitates degradation of apoptotic cells in Caenorhabditis elegans". Proceedings of the National Academy of Sciences of the United States of America. 114 (33): 8806–8811. Bibcode:2017PNAS..114.8806D. doi:10.1073/pnas.1705084114. PMC 5565440. PMID 28760991.
- ^ Cattaneo AM, Bengtsson JM, Montagné N, Jacquin-Joly E, Rota-Stabelli O, Salvagnin U, et al. (2016). "TRPA5, an Ankyrin Subfamily Insect TRP Channel, is Expressed in Antennae of Cydia pomonella (Lepidoptera: Tortricidae) in Multiple Splice Variants". Journal of Insect Science. 16 (1): 83. doi:10.1093/jisesa/iew072. PMC 5026476. PMID 27638948.
- ^ Colbert HA, Smith TL, Bargmann CI (November 1997). "OSM-9, a novel protein with structural similarity to channels, is required for olfaction, mechanosensation, and olfactory adaptation in Caenorhabditis elegans". The Journal of Neuroscience. 17 (21): 8259–69. doi:10.1523/JNEUROSCI.17-21-08259.1997. PMC 6573730. PMID 9334401.
- ^ Gong Z, Son W, Chung YD, Kim J, Shin DW, McClung CA, et al. (October 2004). "Two interdependent TRPV channel subunits, inactive and Nanchung, mediate hearing in Drosophila". The Journal of Neuroscience. 24 (41): 9059–66. doi:10.1523/JNEUROSCI.1645-04.2004. PMC 6730075. PMID 15483124.
- ^ Kim J, Chung YD, Park DY, Choi S, Shin DW, Soh H, et al. (July 2003). "A TRPV family ion channel required for hearing in Drosophila". Nature. 424 (6944): 81–4. Bibcode:2003Natur.424...81K. doi:10.1038/nature01733. PMID 12819662. S2CID 4426696.
- ^ Dong XP, Wang X, Xu H (April 2010). "TRP channels of intracellular membranes". Journal of Neurochemistry. 113 (2): 313–28. doi:10.1111/j.1471-4159.2010.06626.x. PMC 2905631. PMID 20132470.
- ^ Palovcak E, Delemotte L, Klein ML, Carnevale V (July 2015). "Comparative sequence analysis suggests a conserved gating mechanism for TRP channels". The Journal of General Physiology. 146 (1): 37–50. doi:10.1085/jgp.201411329. PMC 4485022. PMID 26078053.
- ^ Hille B (2001). Ion channels of excitable membranes (3rd ed.). Sunderland, Mass.: Sinauer. ISBN 978-0878933211. OCLC 46858498.
- ^ a b c d Winston KR, Lutz W (March 1988). "Linear accelerator as a neurosurgical tool for stereotactic radiosurgery". Neurosurgery. 22 (3): 454–64. doi:10.1097/00006123-198803000-00002. PMID 3129667.
- ^ a b c Cosens DJ, Manning A (October 1969). "Abnormal electroretinogram from a Drosophila mutant". Nature. 224 (5216): 285–7. Bibcode:1969Natur.224..285C. doi:10.1038/224285a0. PMID 5344615. S2CID 4200329.
- ^ a b Montell C, Rubin GM (April 1989). "Molecular characterization of the Drosophila trp locus: a putative integral membrane protein required for phototransduction". Neuron. 2 (4): 1313–23. doi:10.1016/0896-6273(89)90069-x. PMID 2516726. S2CID 8908180.
- ^ a b Eccles R (1989). "Nasal physiology and disease with reference to asthma". Agents and Actions. Supplements. 28: 249–61. PMID 2683630.
- ^ Brauchi S, Orta G, Salazar M, Rosenmann E, Latorre R (May 2006). "A hot-sensing cold receptor: C-terminal domain determines thermosensation in transient receptor potential channels". The Journal of Neuroscience. 26 (18): 4835–40. doi:10.1523/JNEUROSCI.5080-05.2006. PMC 6674176. PMID 16672657.
- ^ Philippaert K, Pironet A, Mesuere M, Sones W, Vermeiren L, Kerselaers S, et al. (March 2017). "Steviol glycosides enhance pancreatic beta-cell function and taste sensation by potentiation of TRPM5 channel activity". Nature Communications. 8: 14733. Bibcode:2017NatCo...814733P. doi:10.1038/ncomms14733. PMC 5380970. PMID 28361903.
- ^ Leung HT, Tseng-Crank J, Kim E, Mahapatra C, Shino S, Zhou Y, et al. (June 2008). "DAG lipase activity is necessary for TRP channel regulation in Drosophila photoreceptors". Neuron. 58 (6): 884–96. doi:10.1016/j.neuron.2008.05.001. PMC 2459341. PMID 18579079.
- ^ Levine JD, Alessandri-Haber N (August 2007). "TRP channels: targets for the relief of pain". Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease. 1772 (8): 989–1003. doi:10.1016/j.bbadis.2007.01.008. PMID 17321113.
- ^ a b Prevarskaya N, Zhang L, Barritt G (August 2007). "TRP channels in cancer". Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease. 1772 (8): 937–46. doi:10.1016/j.bbadis.2007.05.006. PMID 17616360.
- ^ Lozano C, Córdova C, Marchant I, Zúñiga R, Ochova P, Ramírez-Barrantes R, et al. (15 October 2018). "Intracellular aggregated TRPV1 is associated with lower survival in breast cancer patients". Breast Cancer: Targets and Therapy. 10: 161–168. doi:10.2147/BCTT.S170208. PMC 6197232. PMID 30410392.
- ^ Wong KK, Banham AH, Yaacob NS, Nur Husna SM (February 2019). "The oncogenic roles of TRPM ion channels in cancer". Journal of Cellular Physiology. 234 (9): 14556–14573. doi:10.1002/jcp.28168. PMID 30710353. S2CID 73432591.
- ^ Hall DP, Cost NG, Hegde S, Kellner E, Mikhaylova O, Stratton Y, et al. (November 2014). "TRPM3 and miR-204 establish a regulatory circuit that controls oncogenic autophagy in clear cell renal cell carcinoma". Cancer Cell. 26 (5): 738–53. doi:10.1016/j.ccell.2014.09.015. PMC 4269832. PMID 25517751.
- ^ Loo SK, Ch'ng ES, Md Salleh MS, Banham AH, Pedersen LM, Møller MB, et al. (July 2017). "TRPM4 expression is associated with activated B cell subtype and poor survival in diffuse large B cell lymphoma". Histopathology. 71 (1): 98–111. doi:10.1111/his.13204. PMID 28248435. S2CID 4767956.
- ^ Palmer RK, Atwal K, Bakaj I, Carlucci-Derbyshire S, Buber MT, Cerne R, et al. (December 2010). "Triphenylphosphine oxide is a potent and selective inhibitor of the transient receptor potential melastatin-5 ion channel". Assay and Drug Development Technologies. 8 (6): 703–13. doi:10.1089/adt.2010.0334. PMID 21158685.
- ^ a b c d Soussi, M; Hasselsweiller, A; Gkika, D (12 September 2023). "TRP Channels: The Neglected Culprits in Breast Cancer Chemotherapy Resistance?". Membranes. 13 (9): 788. doi:10.3390/membranes13090788. PMC 10536409. PMID 37755210.
 This article incorporates text available under the CC BY 4.0 license.
This article incorporates text available under the CC BY 4.0 license.
- ^ Meseguer V, Alpizar YA, Luis E, Tajada S, Denlinger B, Fajardo O, et al. (20 January 2014). "TRPA1 channels mediate acute neurogenic inflammation and pain produced by bacterial endotoxins". Nature Communications. 5: 3125. Bibcode:2014NatCo...5.3125M. doi:10.1038/ncomms4125. PMC 3905718. PMID 24445575.
- ^ Soldano A, Alpizar YA, Boonen B, Franco L, López-Requena A, Liu G, et al. (June 2016). "Gustatory-mediated avoidance of bacterial lipopolysaccharides via TRPA1 activation in Drosophila". eLife. 5. doi:10.7554/eLife.13133. PMC 4907694. PMID 27296646.
- ^ Boonen B, Alpizar YA, Sanchez A, López-Requena A, Voets T, Talavera K (July 2018). "Differential effects of lipopolysaccharide on mouse sensory TRP channels". Cell Calcium. 73: 72–81. doi:10.1016/j.ceca.2018.04.004. PMID 29689522. S2CID 13681499.
- ^ Alpizar YA, Boonen B, Sanchez A, Jung C, López-Requena A, Naert R, et al. (October 2017). "TRPV4 activation triggers protective responses to bacterial lipopolysaccharides in airway epithelial cells". Nature Communications. 8 (1): 1059. Bibcode:2017NatCo...8.1059A. doi:10.1038/s41467-017-01201-3. PMC 5651912. PMID 29057902.
- ^ Minke B, Wu C, Pak WL (November 1975). "Induction of photoreceptor voltage noise in the dark in Drosophila mutant". Nature. 258 (5530): 84–7. Bibcode:1975Natur.258...84M. doi:10.1038/258084a0. PMID 810728. S2CID 4206531.
- ^ Hardie RC, Minke B (April 1992). "The trp gene is essential for a light-activated Ca2+ channel in Drosophila photoreceptors". Neuron. 8 (4): 643–51. doi:10.1016/0896-6273(92)90086-S. PMID 1314617. S2CID 34820827.
- ^ Phillips AM, Bull A, Kelly LE (April 1992). "Identification of a Drosophila gene encoding a calmodulin-binding protein with homology to the trp phototransduction gene". Neuron. 8 (4): 631–42. doi:10.1016/0896-6273(92)90085-R. PMID 1314616. S2CID 21130927.
- ^ Zhang, Yali V.; Raghuwanshi, Rakesh P.; Shen, Wei L.; Montell, Craig (October 2013). "Food experience-induced taste desensitization modulated by the Drosophila TRPL channel". Nature Neuroscience. 16 (10): 1468–1476. doi:10.1038/nn.3513. ISSN 1546-1726. PMC 3785572. PMID 24013593.
External links[edit]
- Transient+Receptor+Potential+Channels at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- "Transient Receptor Potential Channels". IUPHAR Database of Receptors and Ion Channels. International Union of Basic and Clinical Pharmacology.
- Clapham DE, DeCaen P, Carvacho I, Chaudhuri D, Doerner JF, Julius D, et al. "Transient Receptor Potential channels". IUPHAR/BPS Guide to Pharmacology.
- "TRIP Database". a manually curated database of protein-protein interactions for mammalian TRP channels.