| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.000.850 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1078 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2Cl4F2 | |
| Molar mass | 203.82 g·mol−1 |
| Density | 1.65 g/mL[1] |
| Melting point | 40.6 °C (105.1 °F; 313.8 K)[2] |
| Boiling point | 91 °C (196 °F; 364 K)[2] |
| Related compounds | |
Related compounds
|
CFC-112 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
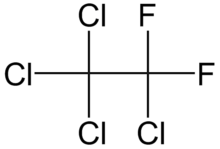
Tetrachloro-1,1-difluoroethane or 1,1,1,2-tetrachloro-2,2-difluoroethane, Freon 112a, R-112a, or CFC-112a is an asymmetric chlorofluorocarbon isomer of tetrachloro-1,1-difluoroethane with formula CClF2CCl3. It contains ethane substituted by four chlorine atoms and two fluorine atoms. With a boiling point of 91.5°C it is the freon with second highest boiling point.
Tetrachlorodifluoroethane as made is a mixture of the symmetrical and asymmetric isomers.[3]
Preparation[edit]
Tetrachloro-1,1-difluoroethane can be prepared in 40% yield by reacting 1,1,2-trichloro-1,2,2-trifluoroethane (freon 113) with aluminium chloride at 60°C.[2]
It can also be made in a reaction with hydrogen fluoride with hexachloroethane or tetrachloroethane with extra chlorine. This reaction occurs with an aluminium fluoride catalyst at 400°C. unsymmetrical trichlorotrifluoroethane (CCl2FCClF2) is also produced along with other chlorofluorocarbons. Separation of the symmetrical and unsymmetrical isomer is difficult.[4]
Properties[edit]
Tetrachloro-1,1-difluoroethane is non-combustible.
It has a critical pressure of 4.83 MPa and a critical temperature of 279.2°. At the critical point the density is 0.754 g/cc.[5]
Tetrachloro-1,1-difluoroethane in liquid form is miscible with perfluorocarbons.[6]
Reactions[edit]
Tetrachloro-1,1-difluoroethane reacts with zinc in ethanol at 60°C to yield unsymmetrical dichlordifluoroethylene (CCl2=CF2).[7]
Use[edit]
Tetrachlorodifluoroethane (mixture of isomers) has been used as a veterinary medicine to treat parasites (Fasciola hepatica).[8]
Atmosphere[edit]
Tetrachloro-1,1-difluoroethane was first detected in air collected from Cape Grim, Tasmania in the Cape Grim Air Archive, and later from air bubbles in snow from Greenland. The substance made its first appearance around 1965, and increased in level until around 2000.[9] In 2000 Earth's atmosphere contained 0.08 parts per trillion of Freon 112a.[10] Level slightly declined to 0.07 ppt by 2012.[9] Estimated lifetime in the stratosphere is 44 years.[10] By 2014 3,600 tons of Freon 112a had been put into the atmosphere.[9] As of 2023, levels have been rising in the Earth's atmosphere.[11]
As a greenhouse gas its radiative efficiency is 0.25 Wm−2ppb−1.[10]
References[edit]
- ^ "CDC - NIOSH Pocket Guide to Chemical Hazards - 1,1,1,2-Tetrachloro-2,2-difluoroethane". www.cdc.gov.
- ^ a b c Miller, William T.; Fager, Edward W.; Griswald, Paul H. (February 1950). "The Rearrangement of Chlorofluorocarbons by Aluminum Chloride 1". Journal of the American Chemical Society. 72 (2): 705–707. doi:10.1021/ja01158a013.
- ^ Gallagher, C. H.; Boray, J. C.; Koch, J. H. (June 1965). "Toxicity of Samples of Tetrachlorodifluoroethane". Australian Veterinary Journal. 41 (6): 167–172. doi:10.1111/j.1751-0813.1965.tb01814.x. PMID 14337687.
- ^ Vecchio, M; Groppelli, G; Tatlow, J. C. (1 July 1974). "Studies on a vapour-phase process for the manufacture of chlorofluoroethanes". Journal of Fluorine Chemistry. 4 (2): 117–139. doi:10.1016/S0022-1139(00)82507-5.
- ^ Bruno, Thomas J. (1990). Spectroscopic Library for Alternative Refrigerant Analysis. U.S. Department of Commerce, National Institute of Standards and Technology. p. 9.
- ^ Fowler, R.; Buford III, W.; Hamilton, Jr., J.; Sweet, R.; Weber, C.; Kasper, J.; Litant, I. (March 1947). "Synthesis of Fluorocarbons". Industrial & Engineering Chemistry. 39 (3): 292–298. doi:10.1021/ie50447a612.
- ^ Barr (Jr.), John T.; Gibson, J. D. (1950). The Preparation and Reactions of Substituted Polychloro 1,1-difluoroethanes. Carbide and Carbon Chemicals Division, Union Carbide and Carbon Corporation, K-25 Laboratory Division. p. 19.
- ^ McKellar, Quintin A.; Kinabo, Ludovick D. B. (1 July 1991). "The pharmacology of flukicidal drugs". British Veterinary Journal. 147 (4): 306–321. doi:10.1016/0007-1935(91)90003-6. ISSN 0007-1935. PMID 1913127.
- ^ a b c Laube, Johannes C.; Newland, Mike J.; Hogan, Christopher; Brenninkmeijer, Carl A. M.; Fraser, Paul J.; Martinerie, Patricia; Oram, David E.; Reeves, Claire E.; Röckmann, Thomas; Schwander, Jakob; Witrant, Emmanuel; Sturges, William T. (April 2014). "Newly detected ozone-depleting substances in the atmosphere" (PDF). Nature Geoscience. 7 (4): 266–269. Bibcode:2014NatGe...7..266L. doi:10.1038/ngeo2109. S2CID 140544959.
- ^ a b c Davis, Maxine E.; Bernard, François; McGillen, Max R.; Fleming, Eric L.; Burkholder, James B. (1 July 2016). "UV and infrared absorption spectra, atmospheric lifetimes, and ozone depletion and global warming potentials for CCl2FCCl2F (CFC-112), CCl3CClF2 (CFC-112a), CCl3CF3 (CFC-113a), and CCl2FCF3; (CFC-114a)". Atmospheric Chemistry and Physics. 16 (12): 8043–8052. Bibcode:2016ACP....16.8043D. doi:10.5194/acp-16-8043-2016. hdl:1983/df193a7b-14de-427c-a539-238701f9e3b3. S2CID 102078043.
- ^ Western, Luke M.; et al. (3 April 2023). "Global increase of ozone-depleting chlorofluorocarbons from 2010 to 2020". Nature Geoscience. 16 (4): 309–313. Bibcode:2023NatGe..16..309W. doi:10.1038/s41561-023-01147-w. hdl:1983/9e103fef-e61c-49c7-a1a3-902540ec1d7c. S2CID 257941769.
Extra reading[edit]
- Sládek, Petr; Navrátil, Oldřich; Linhart, Petr (1992). "Extraction of Selected Lanthanoids and Scandium with Bis(2-ethylhexyl)hydrogenphosphate in 1,1,2,2-Tetrachlorodifluoroethane". Collection of Czechoslovak Chemical Communications. 57 (8): 1647–1654. doi:10.1135/cccc19921647.
- Sládek, Petr; Navrátil, Oldřich; Linhart, Petr (1992). "Extraction of Ce, Pm, Eu, Tm and Sc Using Di-n-butylhydrogenphosphate in 1,1,2,2-Tetrachlorodifluoroethane". Collection of Czechoslovak Chemical Communications. 57 (8): 1639–1646. doi:10.1135/cccc19921639.
- Kakáč, B.; Hudlický, M. (1965). "Organic compounds of fluorine. VII. Spectrophotometric study in the series of halofluoroethanes". Collection of Czechoslovak Chemical Communications. 30 (3): 745–751. doi:10.1135/cccc19650745.