| |
| Names | |
|---|---|
| Other names
sodium metagermanate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.031.535 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Na2GeO3 | |
| Molar mass | 166.62 g/mol |
| Appearance | white solid |
| Odor | odorless |
| Density | 3.31 g/cm3 |
| Melting point | 1,060 °C (1,940 °F; 1,330 K) |
| 14.4 g/100 mL (0 °C) 23.8 g/100 mL (25 °C) | |
Refractive index (nD)
|
1.59 |
| Structure | |
| orthorhombic | |
| Related compounds | |
Related compounds
|
Sodium silicate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
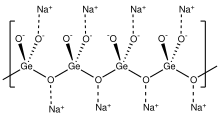
Sodium germanate is an inorganic compound with the chemical formula Na2GeO3. It exists as a colorless solid. Sodium germanate is primarily used for the synthesis of other germanium compounds.
Preparation and reactions[edit]
Sodium germanate can be prepared by the fusion of germanium oxide with sodium hydroxide at high temperatures:
- 2 NaOH + GeO2 → Na2GeO3 + H2O
An intermediate in this reaction is the protonated derivative NaHGeO3, which is a water-soluble salt.
Structure[edit]
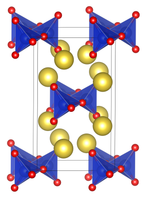
it is structurally analogous to sodium metasilicate, Na2SiO3, consisting of polymeric GeO32− anions made up of vertex sharing {GeO4} tetrahedra.[1][2]
See also[edit]
References[edit]
- ^ Cruickshank, D. W. J.; Kálmán, A.; Stephens, J. S. (1978). "A Reinvestigation of Sodium Metagermanate". Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry. 34 (4): 1333–1334. Bibcode:1978AcCrB..34.1333C. doi:10.1107/S0567740878005488.
- ^ C. C. Addison, Inorganic Chemistry of the Main-Group Elements, vol 1, 1973, The chemical Society, ISBN 9780851867526