 | |
| Clinical data | |
|---|---|
| Trade names | Amdaquine, Amobin, Camoquin, others[1] |
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.001.518 |
| Chemical and physical data | |
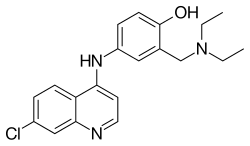
| Formula | C20H22ClN3O |
| Molar mass | 355.87 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Amodiaquine (ADQ) is a medication used to treat malaria, including Plasmodium falciparum malaria when uncomplicated.[2][3] It is recommended to be given with artesunate to reduce the risk of resistance.[2] Due to the risk of rare but serious side effects, it is not generally recommended to prevent malaria.[2] Though, the World Health Organization (WHO) in 2013 recommended use for seasonal preventive in children at high risk in combination with sulfadoxine and pyrimethamine.[4]
Amodiaquine is a 4-aminoquinoline compound related to chloroquine.[2] The side effects of amodiaquine are generally minor to moderate and are similar to those of chloroquine.[3] Rarely liver problems or low blood cell levels may occur.[2] When taken in excess headaches, trouble seeing, seizures, and cardiac arrest may occur.[2] The WHO recommends its use for pregnant women during the second and third trimester as well as during lactation, but reports that evidence for use in the first trimester is still insufficient.[5]
Amodiaquine was first made in 1948.[6] It is on the World Health Organization's List of Essential Medicines.[7][8] While not available in the United States,[9] it is widely available in Africa.[2][10]
Medical uses[edit]
Amodiaquine has become an important drug in the combination therapy for malaria treatment in Africa.[11] It is often used in combination with artesunate as a by mouth artemisinin-based combination therapy (ACT) for uncomplicated P. falciparum malaria.[12] Amodiaquine has also been found to work against chloroquine-resistant P. falciparum strains of malaria, though there is geographic variation in its activity against chloroquine-resistant strains.[13][14]
It is also used in combination with sulfadoxine/pyrimethamine.[15][16]
Interactions[edit]
There have been reports of increased liver toxicity in people with HIV/AIDS on zidovudine or efavirenz when treated with amodiaquine-containing ACT regimens, therefore it is recommended that these people avoid amodiaquine.[12]
Pharmacokinetics and pharmacogenetics[edit]
It is bioactivated hepatically to its primary metabolite, N-desethylamodiaquine, by the cytochrome p450 enzyme CYP2C8. Among amodiaquine users, several rare but serious side effects have been reported and linked to variants in the CYP2C8 alleles. CYP2C8*1 is characterized as the wild-type allele, which shows an acceptable safety profile, while CYP2C8*2, *3 and *4 all show a range of "poor metabolizer" phenotypes. People who are poor metabolizers of amodiaquine display lower treatment efficacy against malaria, as well as increased toxicity.[17] Several studies have been conducted to determine the prevalence of CYP2C8 alleles amongst malaria patients in East Africa, and have tentatively shown the variant alleles have significant prevalence in that population.[18] About 3.6% of the population studied showed high risk for a poor reaction to or reduced treatment outcomes when treated with amodiaquine. This information is useful in developing programs of pharmacovigilance in East Africa, and have important clinical considerations for prescribing antimalarial medications in regions with high CYP2C8 variant frequency.
See also[edit]
- Structurally similar is FGI-104
References[edit]
- ^ "Amodiaquine". drugs.com. Archived from the original on 27 November 2016. Retrieved 27 November 2016.
- ^ a b c d e f g Nair A, Abrahamsson B, Barends DM, Groot DW, Kopp S, Polli JE, et al. (December 2012). "Biowaiver monographs for immediate release solid oral dosage forms: amodiaquine hydrochloride". Journal of Pharmaceutical Sciences. 101 (12): 4390–4401. doi:10.1002/jps.23312. PMID 22949374.
- ^ a b Olliaro P, Mussano P (2003). "Amodiaquine for treating malaria". The Cochrane Database of Systematic Reviews (2): CD000016. doi:10.1002/14651858.CD000016. PMC 6532704. PMID 12804382.
- ^ Seasonal malaria chemoprevention with sulfadoxine–pyrimethamine plus amodiaquine in children: a field guide (PDF). Geneva: The World Health Organization. August 2013. ISBN 978-92-4-150473-7.
- ^ "WHO Guidelines for malaria". www.who.int. Retrieved 2024-01-22.
- ^ Ahmad I, Ahmad T, Usmanghani K (1992). "Amodiaquine hydrochloride". Profiles of Drug Substances, Excipients and Related Methodology. Analytical Profiles of Drug Substances and Excipients. Vol. 21. Academic Press. p. 45. doi:10.1016/S0099-5428(08)60388-3. ISBN 978-0-08-086116-6. Archived from the original on 2017-09-08.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "Amodiaquine". Livertox. Archived from the original on 27 November 2016. Retrieved 27 November 2016.
- ^ Centers for Disease Control (CDC) (April 1985). "Revised recommendations for preventing malaria in travelers to areas with chloroquine-resistant Plasmodium falciparum". MMWR. Morbidity and Mortality Weekly Report. 34 (14): 185–90, 195. PMID 3156271.
- ^ Kerb R, Fux R, Mörike K, Kremsner PG, Gil JP, Gleiter CH, Schwab M (December 2009). "Pharmacogenetics of antimalarial drugs: effect on metabolism and transport". The Lancet. Infectious Diseases. 9 (12): 760–774. doi:10.1016/S1473-3099(09)70320-2. PMID 19926036.
- ^ a b World Health Organization (2015). Guidelines for the treatment of malaria (3rd ed.). World Health Organization. hdl:10665/162441. ISBN 978-92-4-154912-7.
- ^ "Amodiaquine". PubChem. U.S. National Library of Medicine. Archived from the original on 2016-10-29. Retrieved 2016-10-28.
- ^ Bennett, John Eugene; Dolin, Raphael; Blaser, Martin J., eds. (2020). "41 - Antimalarial Drugs". Mandell, Douglas, and Bennett's principles and practice of infectious diseases (Ninth ed.). Philadelphia, PA: Elsevier. pp. 519–534. ISBN 978-0-323-48255-4.
- ^ Staedke SG, Kamya MR, Dorsey G, Gasasira A, Ndeezi G, Charlebois ED, Rosenthal PJ (August 2001). "Amodiaquine, sulfadoxine/pyrimethamine, and combination therapy for treatment of uncomplicated falciparum malaria in Kampala, Uganda: a randomised trial". Lancet. 358 (9279): 368–374. doi:10.1016/S0140-6736(01)05557-X. PMID 11502317. S2CID 42745422.
- ^ World Health Organization (2013). Seasonal malaria chemoprevention with sulfadoxine–pyrimethamine plus amodiaquine in children: a field guide. World Health Organization. hdl:10665/85726. ISBN 978-92-4-150473-7.
- ^ Elyazar IR, Hay SI, Baird JK (April 2011). "Malaria distribution, prevalence, drug resistance and control in Indonesia". Advances in Parasitology. 74 (74): 41–175. doi:10.1016/B978-0-12-385897-9.00002-1. ISBN 978-0-12-385897-9. PMC 3075886. PMID 21295677.
- ^ Roederer MW, McLeod H, Juliano JJ (November 2011). "Can pharmacogenomics improve malaria drug policy?". Bulletin of the World Health Organization. 89 (11): 838–845. doi:10.2471/BLT.11.087320. PMC 3209725. PMID 22084530.
External links[edit]
- "Amodiaquine". Drug Information Portal. U.S. National Library of Medicine.